Abstract
Brown rust (Puccinia melanocephala) and orange rust (P. kuehnii) cause important yield loss in global sugarcane production. Due to the difficulties of distinguishing between the two diseases to the naked eye, it is essential to use molecular techniques for an accurate rust diagnosis. A major gene, Bru1, which confers resistance to a broad spectrum of P. melanocephala strains in different parts of the world, has been described, and molecular markers closely associated with this allele have been developed. The aims of the present study were: (1) to optimise a PCR-based method to diagnose and characterise the causal agent of both rusts in Tucumán; (2) to determine the usefulness of the Bru1 gene in the Sugarcane Breeding Program of “Estación Experimental Agroindustrial Obispo Colombres” (EEAOC) by studying its association with resistant and susceptible phenotypes; and (3) to assess the frequency of the Bru1 allele in the sugarcane germplasm of the EEAOC. Conditions for both rust diagnoses were optimised. Out of 129 genotypes evaluated to study the usefulness of the Bru1 allele, 49 were found to be resistant to brown rust, but only eight of these resistant genotypes were positive for the Bru1 allele. Also frequency of appearance of the Bru1 allele was analysed in 191 sugarcane accessions of the EEAOC germplasm, and its presence was detected in only 7 % of the genotypes evaluated. In conclusion, although results showed that Bru1 markers enable positive selection of this character, additional source(s) of resistance are available in the EEAOC Sugarcane Breeding Program.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The rusts are fungal diseases caused by obligate pathogens affecting primarily plant foliage. These diseases are characterised by the initial appearance of small elongated yellowish spots, visible on both sides of the leaves. These spots decrease photosynthetic capacity, increasing their length gradually, becoming brown, orange-brown or red-brown over time. Puccinia melanocephala is the causal agent of brown rust, an important sugarcane disease in many production areas (Raid and Comstock 2000). Brown rust has an important economic impact, having been associated with significant yield losses (>50 %) (Hoy and Hollier 2009). Puccinia kuehnii Butler is the causal agent of orange rust, a less widespread disease than brown rust. The occurrence of this pathogen in the Western hemisphere was confirmed only recently in USA (Comstock et al. 2008) and since then, it has been reported in many countries in Central and South America (Chavarría et al. 2009; Barbasso et al. 2010), but not in Argentina (Funes et al. 2011). Differences in urediniospore coloration and the apical thickening of P. kuehnii urediniospores are the only morphological features distinguishing the two pathogens, although they do not constitute a definitive diagnosis method. Since the identification of the two sugarcane rust pathogens in the field is very difficult by simple observation, and due to the fact that orange rust is an important potential threat to the Argentinean sugarcane industry, an accurate, fast and robust diagnostic method was developed to allow a precise and sensitive discrimination between P. kuehnii and P. melanocephala.
The most effective method of control of these diseases is the use of resistant varieties. However, resistance durability is affected by a number of factors, including pathogen genetics, plant growth stage, weather conditions, plant nutrition and soil characteristics (Anderson and Dean 1986; Anderson et al. 1990, 1991; Raid and Comstock 2000). Due to this, shifts in cultivar reactions from resistance to susceptibility have been observed (Hoy and Hollier 2009). Nevertheless, a brown rust-resistant sugarcane cultivar, R570, one of the most widely cultivated varieties for the last 20 years on the islands of Reunion and Mauritius, has never lost its resistance to brown rust although it has been challenged with various rust isolates collected from all over the world (Asnaghi et al. 2001). The brown rust resistance of this cultivar was studied and results showed a monogenic and dominant control involving a single copy of the resistance allele (Daugrois et al. 1996). This major resistance gene, Bru1, is the first well-characterised Mendelian trait described in the complex genomic context of sugarcane (Asnaghi et al. 2004). Costet et al. (2012) reported that brown rust resistance in modern sugarcane cultivars relies essentially on Bru1. Le Cunff et al. (2008) found that the Bru1 gene is included in an unknown size insertion that is specific to the Bru1-bearing haplotype, being absent in other hom(e)ologous haplotypes. This insertion induces a reduction in recombination, resulting in strong linkage disequilibrium (LD) in the Bru1 region and a complete LD between two flanking molecular markers, R12H16 and 9020-F4. These markers represent good diagnostic markers for the presence of Bru1, since their presence predicts a resistant behaviour of any modern cultivar. In addition, the absence of these markers in a resistant cultivar indicates the absence of Bru1, suggesting an alternative source of resistance (Costet et al. 2012).
The work presented here aims at (1) optimising a PCR-based method in order to diagnose and characterise the causal agent population of both brown and orange rust in Tucumán; (2) determining the usefulness of applying molecular markers for the Bru1 gene in the Sugarcane Breeding Program of “Estación Experimental Agroindustrial Obispo Colombres” (EEAOC) by studying their association with resistant and susceptible phenotypes; and (3) assessing the frequency of the Bru1 allele in the sugarcane germplasm of the EEAOC.
Materials and Methods
Detection of P. kuehnii and P. melanocephala by PCR
To evaluate the presence of the causal agents of brown and orange rust, 30 young leaf samples (first and second sheets with visible ligules) exhibiting typical symptoms of rust were collected from plants at different production areas of Tucumán, Argentina (Cerco Represa, Las Talitas, Santa Ana, Río Seco, La Banda, Mercedes and Fronterita). Plant material with different numbers of uredinial lesions was collected in order to determine the optimal conditions for nucleic acid extraction for rust detection and diagnosis.
Total RNA and DNA were extracted essentially using the CTAB (cetyl trimethyl ammonium bromide) technique described by Aljanabi et al. (1999), optimised for samples with rust pustules.
Amplification of ribosomal DNA (rDNA) was performed by PCR, by using five different pairs of primers: two general ones for fungi (ITS1F/ITS4, NL1/NL4), a pair of species-specific primers to differentiate between P. melanocephala and P. kuehnii by the size of the band amplified (PkPmF: AAGAGTGCACTTAATTGTGGCTC/PkPmR: TCCCACCTGATTTGAGGTCT), one specific for P. melanocephala (Pm1F: AATTGTGGCTCGAACCATCTTC/Pm1R: TTGCTACTTTCCTTGATGCTC) and one specific for P. kuehnii (PkPmF: AAGAGTGCACTTAATTGTGGCTC/Pk1R: CAGGTAACACCTTCCTTGATGTG) (Virtudazo et al. 2001; Glynn et al. 2010). Different concentrations of MgCl2, primers and nucleic acids (mould), as well as annealing temperature of the primers, were analysed. Positive and negative controls were included for P. melanocephala and P. kuehnii in all cases. The latter control was kindly provided by Centro de Tecnología Canaviera (CTC), Brazil. PCR products were separated on 1.5 % agarose gels, stained with GelRed and visualised on a UV transilluminator. Some amplified bands were removed, purified from agarose gel using the QIAGEN Gel Extraction Kit and sequenced in both directions at the “Instituto Nacional de Tecnología Agropecuaria” Laboratory (Castelar, Argentina) sequencing service. All DNA sequences obtained were compared with available sequences in the public data base GenBank.
Genotyping for the Presence of Bru1 Using Molecular Markers
To determine the usefulness of Bru1 in our local rust condition, 129 sugarcane accessions, frequently used as parents in the EEAOC Breeding Program (Table 1), were analysed in order to study the existence of this gene by applying molecular diagnostic markers R12H16 and 9020-F4-RsaI. Out of the 129 sugarcane cultivars tested, most were HOCP varieties (46 %) obtained in crossings performed at Canal Point and then selected at Houma, and TUC varieties (40 %) obtained and selected within the EEAOC Breeding Program.
Then, to determine frequency of appearance of Bru1 gene in the EEAOC germplasm collection, 191 sugarcane accessions (Table 2) were studied to detect the presence of the Bru1 gene, by using diagnostic markers.
To genotype for the presence of Bru1 gene, three young leaves from each genotype were collected and stored at −70 °C until further processed. Before DNA extraction, plant tissue was kept frozen by using liquid nitrogen and then it was ground to a fine powder with a mortar and pestle. Genomic DNA was extracted following the CTAB method essentially as described by Aljanabi et al. (1999). Two different molecular diagnostic markers, R12H16 and 9020-F4, associated with Bru1 and strongly associated with brown rust resistance (Costet et al. 2012) were used in this study to test for the presence of the Bru1 gene. All PCRs were carried out using the same setup: 20 μl final volume containing 50 ng template DNA, 0.4 μM of each primer; 0.4 mM of each dNTP, 2.5 mM MgCl2, 0.5 units Taq Polymerase (Invitrogen LifeTech) with 1X PCR buffer provided with the enzyme. Cycling was performed in My Cycler thermocycler (Bio-Rad) as follows: 4 min denaturation at 94 °C followed by 35 cycles of 94 °C for 30 s, 55.5 °C for 45 s, 72 °C for 72 s and final elongation for 8 min at 72 °C. All amplifications were repeated at least three times. Fifteen microlitres of the 9020-F4 PCR products was digested with restriction enzyme Rsa I to visualise the presence of a specific band associated with the Bru1 allele. The R12H16 PCR marker corresponds to a specific fragment whose presence is associated with the Bru1 gene. R12H16 PCR products and 9020-F4-RsaI restriction fragments were visualised following electrophoresis on a 1.5 and 3 % agarose gel, respectively, and stained with GelRed.
Field Evaluation of Brown Rust Resistance
In order to determine the usefulness of the Bru1 gene in our local condition, field testing for rust susceptibility and resistance for 129 sugarcane cultivars were determined by using natural infection on plant cane in an unreplicated design consisting of individual 3-m-long row plots. The trial was planted in Cerco Represa (Tucumán, Argentina) in July 2011 and evaluated for brown rust appearance in February 2012, i.e., in a high-pressure inoculum locality in the most favourable period for rust development. Brown rust reaction was scored in each plot on a 0 (totally resistant) to 9 (very susceptible) scale, according to Amorin et al. (1987), by two independent observers. This scale is based on visual assessment of the disease symptoms. A score 0–2 indicates resistant plants with no pustules of the fungus, though some necrotic or chlorotic spots may appear on the leaves. A score of 3–4 indicates moderate resistant plants with very rare pustule lesions on old leaves but with no sporulating pustules. From grade 5 to 9, plants are considered as susceptible with the density of sporulating lesions increasing, as well as their appearance on younger leaves and extensive leaf necrosis.
The LCP 85-384 variety, which is very susceptible to brown rust, was planted at the border surrounding the trial, as well as repeatedly inside the plots in order to maximise infection.
Results
PCR Diagnosis of P. kuehnii and P. melanocephala
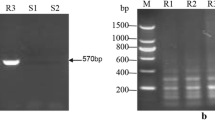
Good-quality nucleic acid was obtained by using samples with a reduced number of pustules and one additional round of phenol treatment in the extraction procedure. All 30 sugarcane leaf samples that exhibited rust symptoms were found to be positive for P. melanocephala generating a 670-base pairs (bp) product with primer set ITS1F/ITS4, a 608-bp product with the pair NL1/NL4, a 585-bp product with PkPmF/PkPmR and finally a 480-bp product with Pm1F/Pm1R (Fig. 1). No band was observed at 527 bp when using the primer pair specific for P. kuehnii in any of the samples.
Amplification products with primers PkPmF/PkPmR: lanes 1–3, negative controls; lanes 4 and 5, P. melanocephala positive controls; lanes 6 and 7, P. kuehnii positive controls. Amplification products with PkPmF/Pk1R primers: lanes 8–10, negative controls; lanes 11 and 12, P. melanocephala samples and lanes 13 and 14, P. kuehnii positive controls. Amplification products with Pm1F/Pm1R primers: lane 15, negative control; lanes 16 and 17, P. kuehnii samples and lanes 18 and 19, P. melanocephala positive controls. M: ladder molecular Cien Marker, Promega (100–1000 pb)
Comparisons between sequences obtained and sequences deposited in the GenBank database confirmed brown rust’s presence in samples tested.
Frequency of Brown Rust Resistance and Bru1 Markers Within the Breeding Population of EEAOC
The reaction to brown rust for 129 sugarcane parents frequently used in the EEAOC Breeding Program was evaluated in the field under natural infestation conditions. Rust severity was scored on a 0 (most resistant) to 9 (most susceptible) scale, according to Amorin et al. (1987). Genotypes were considered as resistant, collecting a score of 2 or lower and as susceptible when the score was equal to or higher than 3. Out of 129 evaluated genotypes, 49 (38 %) were found to be resistant to brown rust. Both molecular markers, R12H16 and 9020-F4-RsaI, associated with the Bru1 gene were detected in 8 out of the 49 resistant genotypes tested (16.3 %). Both markers were present in all positive samples, suggesting a high linkage grade between them, and were absent in all the 80 susceptible accessions. Interestingly, 83.7 % of all clones considered to be resistant to rust, as seen from our field trial, did not contain Bru1 as inferred from analyses with the two diagnostic markers. This result clearly indicates that resistance to brown rust in EEAOC Breeding Program depends mostly on one or more alternative sources of resistance.
Screening for Rust Resistance Gene Bru1 Using Molecular Markers in the EEAOC Sugarcane Germplasm
One hundred and ninety-one sugarcane accessions from EEAOC sugarcane germplasm were analysed to study the frequency of appearance of the Bru1 gene, through molecular diagnostic marker R12H16 and 9020-F4-RsaI. Only 7 % (14/191) of the genotypes evaluated, namely Co 290, Co 419, Co 421, CP52-68, CP53-17, POJ2878, R570, TUC01-45, TUC02-38, TUC02-41, TUC03-32, TUC03-36, TUC04-4 and TUC94-58, were found to contain the Bru1 gene allele.
Discussion and Conclusions
A rapid and robust nucleic acid extraction protocol was optimised for the detection of fungi causing the disease sugarcane rust. This allowed a simple and quick detection process, especially when assessing a large number of plant samples, since it was not necessary to separate plant tissue from fungus spores.
Protocol optimisation for detecting P. melanocephala and P. kuehnii by PCR assays allowed the development of a robust and specific detection protocol for both rust fungi. It is also important to highlight that the presence of P. kuehnii in commercial sugarcane fields in Argentina was not detected during this study. Therefore, the detection protocol for orange rust will allow for continuous monitoring of crops, as well as in imported plant materials, helping to prevent the outbreak of the pathogen in Argentina. Moreover, it is always important to have access to precise diagnostic methods in order to help to design management strategies of potential diseases, whose consequences are still unknown.
The great usefulness of the molecular markers associated with the Bru1 gene was clearly demonstrated, since all genotypes having these markers were resistant to brown rust in our local conditions. However, it must be pointed out that 83.7 % of all clones considered as resistant were negative to the presence of Bru1, indicating that the main resistance to brown rust in our Breeding Program depends mostly on alternative resistance sources.
Screening for rust resistance gene Bru1 in 191 accessions of the EEAOC sugarcane germplasm showed that this gene was only detected in 7 % of the clones evaluated, including clones in the last evaluation steps of our breeding program, as well as in ancestors of CP varieties (from Canal Point, USA). The EEAOC Breeding Program constantly exchanges breeding parents with those in the USA (Canal Point, Louisiana and Houma) due to similar climate and growth conditions. As a consequence, these breeding programs share a narrow gene pool (Perera et al. 2012). The low frequency of Bru1-positive genotypes found is in agreement with the results obtained by Glynn et al. (2012); they found that the frequency of Bru1 in parental clones used in Louisiana sugarcane crossing programs was 6 %.
The alternative source(s) of resistance available in our germplasm could be important as it can help widen the narrowed genetic base of brown rust resistance in other parts of the world (Costet et al. 2012).
References
Aljanabi, S.M., L. Forget, and A. Dookun. 1999. An improved and rapid protocol for the isolation of polysaccharide-and polyphenol-free sugarcane DNA. Plant Molecular Biology Reporter 17: 1–8.
Amorin, L., A.B. Filho, A. Sanguino, C.O. Nogueira Cardoso, V.A. de Moraes, and C.R. Fernandes. 1987. Metodología de avaliacao da ferrugem da cana de acucar (Puccinia melanocephala). Boletín Técnico Copersucar 39: 13–16.
Anderson, D.L., and J.L. Dean. 1986. Relationship of rust severity and plant nutrients in sugarcane. Phytopathology 76: 581–585.
Anderson, D.L., R.N. Raid, M.S. Irey, and L.J. Henderson. 1990. Association of sugarcane rust severity with soil factors. Plant Disease 74: 683–686.
Anderson, D.L., L.J. Henderson, R.N. Raid, and M.S. Irey. 1991. Sugar cane rust severity and leaf nutrient status. Sugar Cane 3: 5–10.
Asnaghi, C., A. D’Hont, J.C. Glaszmann, and P. Rott. 2001. Resistance of sugarcane cultivar R570 to Puccinia melanocephala from different geographic locations. Plant Disease 85: 282–286.
Asnaghi, C., D. Roques, S. Ruffel, C. Kaye, J.-Y. Hoarau, H. Télismart, J.C. Girard, L.M. Raboin, A.M. Risterucci, L. Grivet, and A. D’Hont. 2004. Targeted mapping of a sugarcane rust resistance gene (Bru1) using bulked segregant analysis and AFLP markers. Theoretical and Applied Genetics 108: 759–764.
Barbasso, D., H. Jordao, W. Maccheroni, J. Boldini, and A. Sanguine. 2010. First report of Puccinia kuehnii causal agent of orange rust of sugarcane, in Brazil. Plant Disease 94: 1170.
Chavarría, E., F. Subirós, J. Vega, G. Ralda, N.C. Glynn, J.C. Comstock, and L.A. Castlebury. 2009. First report of orange rust of sugarcane caused by Puccinia kuehnii in Costa Rica and Nicaragua. Plant Disease 93: 425.
Comstock, J.C., S.G. Sood, N.C. Glynn, J. Jr, J.McKemy Shine, and L.A. Castlebury. 2008. First report of Puccinia kuehnii, causal agent of orange rust of sugarcane, in the United States and Western Hemisphere. Plant Disease 92: 175.
Costet, L., L. Le Cunff, S. Royaert, L.-M. Raboin, C. Hervouet, L. Toubi, H. Telismart, O. Garsmeur, Y. Rousselle, J. Pauquet, S. Nibouche, J.-C. Glaszmann, J.-Y. Hoarau, and A. D’Hont. 2012. Haplotype structure around Bru1 reveals a narrow genetic basis for brown rust resistance in modern sugarcane cultivars. Theoretical and Applied Genetics 125: 825–836.
Daugrois, J.H., L. Grivet, D. Roques, J.Y. Hoarau, H. Lombard, J.C. Glaszmann, and A. D’Hont. 1996. A putative major gene for rust resistance linked with a RFLP marker in sugarcane cultivar ‘R570’. Theoretical and Applied Genetics 92: 1059–1064.
Funes, C., A.M. Rago, M.F. Perera, S.G. Pérez Gómez, R. Fernández de Ulivarri, and M.I. Cuenya. 2011. Roya naranja de la caña de azúcar: Situación actual en el continente americano. Avance Agroindustrial 32: 13–17.
Glynn, N.C., L.J. Dixon, L.A. Castlebury, L.J. Szabo, and J.C. Comstock. 2010. PCR assays for the sugarcane rust pathogens Puccinia kuehnii and P. melanocephala and detection of a SNP associated with geographical distribution in P. kuehnii. Plant Pathology 59: 703–711.
Glynn, N.C., J.C. Comstock, and K. McCorkle. 2012. Resistance to sugarcane brown rust: Opportunities and challenges in using the Bru1 resistance gene. In: Abstracts of the 10th pathology workshop of the International Society of Sugarcane Technologists, Nanning, China, 7–11 May 2012. Guangxi Academy of Agricultural Sciences and Guangxi University State Key Laboratory for Conservation and Utilisation of Subtropical Agrobioresources, p 2.
Hoy, J.W., and C.A. Hollier. 2009. Effect of brown rust on yield of sugarcane in Louisiana. Plant Disease 93: 1171–1174.
Hutchinson, P. B., and J. Daniels. 1971. A rating scale for sugarcane characteristics. In Proc. XIVth ISSCT Congress, Baton Rouge, Louisiana, USA. ISSCT, Mauritius
Le Cunff, L., O. Garsmeur, L.M. Raboin, J. Pauquet, H. Telismart, A. Selvi, L. Grivet, R. Philippe, D. Begum, M. Deu, L. Costet, R. Wing, J.C. Glaszmann, and A. D’Hont. 2008. Diploid/polyploid syntenic shuttle mapping and haplotype-specific chromosome walking toward a rust resistance gene (Bru1) in highly polyploid sugarcane (2n * 12x * 115). Genetics 180: 649–660.
Perera, M.F., M.E. Arias, D. Costilla, A.C. Luque, M.B. García, C. Díaz Romero, J. Racedo, S. Ostengo, M.P. Filippone, M.I. Cuenya, and A.P. Castagnaro. 2012. Genetic diversity assessment and genotype identification in sugarcane based on DNA markers and morphological traits. Euphytica 185: 491–510.
Raid, R.N., and J.C. Comstock. 2000. Common rust. In A guide to sugarcane diseases, ed. P. Rott, R.A. Bailey, J.C. Comstock, B.J. Croft, and A.S. Saumtally, 85–89. Montpellier, France: CIRAD and ISSCT.
Virtudazo, E.V., H. Nakamura, and M. Kakishima. 2001. Phylogenetic analysis of sugarcane rusts based on sequences of ITS, 5.8 S rDNA and D1/D2 regions of LSU rDNA. Journal of General Plant Pathology 67: 28–36.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Racedo, J., Perera, M.F., Bertani, R. et al. Molecular Diagnostic of Both Brown and Orange Sugarcane Rust and Evaluation of Sugarcane Brown Rust Resistance in Tucuman, Argentina, Using Molecular Markers Associated with Bru1 a Broad-Range Resistance Allele. Sugar Tech 18, 414–419 (2016). https://doi.org/10.1007/s12355-015-0404-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-015-0404-z