Abstract
Background
Right ventricular (RV) performance in patients of pulmonary hypertension (PH) requires optimal assessment. The objective of this study is to develop phase analysis using 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) imaging as a feasible tool for evaluation of RV dyssynchrony in PH.
Methods and Results
Fifty-four PH patients with well-characterized hemodynamic parameters were enrolled. All subjects performed FDG-PET imaging for RV phase analysis and RV function evaluation. Two-dimensional echocardiography with speckle tracking analysis was conducted to obtain RV time to peak systolic strain (PSST) as a comparison. The median contraction delay difference between RV middle free wall and septum measured by PET phase analysis (RVPDPET) was 20.12° (interquartile range, 4.99°-30.10°). The median difference of PSST between RV middle free wall and middle septal wall (RVPDEcho) measured by echocardiography was 43.98° (interquartile range, 6.25°-72.00°). RVPDPET was well correlated with RVPDEcho (r = 0.685, P < .001). RV phase standard deviation (RVSD) and histogram bandwidth (RVBW) derived from PET phase histogram were significantly correlated with cardiac index, RV ejection fraction, 6-minute walking distance, and serum N-terminal pro B-type natriuretic peptide (NT-proBNP) (RVSD: r = −0.532, P < .001; r = −0.551, P < .001; r = −0.544, P < .001; r = 0.404, P < .01; respectively, RVBW: r = −0.492, P < .001; r = −0.466, P < .001; r = −0.544, P < .001; r = 0.349, P = .01, respectively), while there were no significant correlations between RVSD and RVBW with hemodynamic parameters (right atrial pressure, right ventricular systolic pressure, right ventricular end-diastolic pressure, mean pulmonary artery pressure, and total pulmonary resistance).
Conclusions
Contraction delays between RV free wall and septum in PH measured by phase analysis and speckle tracking echocardiography were well correlated. RV dyssynchrony measured by phase analysis of FDG-PET was significantly related to RV dysfunction. Phase analysis of FDG-PET is feasible to evaluate RV mechanical dyssynchrony in patients of PH.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In pulmonary hypertension (PH), pulmonary vascular proliferation progressively increases pulmonary artery pressure and right ventricular (RV) afterload, subsequently leading to right heart failure.1 RV function as a major determinant of clinical outcomes in PH patients is of great clinical importance and requires optimal assessment.2 To better understand the performance of RV, modern imaging modalities focus on evaluation of not only ventricular systolic function but also right heart metabolism and mechanical synchronicity.
At present, RV mechanical dyssynchrony in PH is predominantly evaluated by echocardiography and has been shown to relate to inefficient RV contraction, impaired left ventricle (LV) diastolic filling, and pump function.3 However, echocardiographic techniques, especially three-dimensional tissue Doppler imaging or speckle tracking, require high standardized expertise for post-processing and optimization of observer variability in clinical practice.4 Lately, automated and reproducible phase analysis using 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) has been suggested as a suitable option for LV dyssynchrony measurement.5 Moreover, recent studies employing FDG-PET showed its value for assessment of RV systolic function and myocardial glucose metabolism in PH, suggesting a potential of FDG-PET imaging in comprehensive evaluation of RV function in PH.6 However, the utility of FDG-PET imaging in assessment of RV mechanical synchrony has not been studied. Thus, this study was aimed to validate the feasibility of phase analysis on FDG-PET imaging to evaluate RV mechanical synchrony in patients with PH in a head-to-head comparison with speckle tracking echocardiography and to explore the relationship of RV dyssynchrony and clinical severity.
Methods
Study Population
A total of fifty-four consecutive patients with PH were enrolled in this study at Fu Wai Hospital from Jan 2011 to Dec 2013. Diagnosis of PH was defined as an increase in mean pulmonary artery pressure (mPAP) ≥ 25 mmHg at rest assessed by right heart catheterization according to guidelines for the diagnosis and treatment of PH.7 Of the fifty-four patients, twenty-six patients had idiopathic pulmonary arterial hypertension, one had familial pulmonary arterial hypertension, ten had PH associated with congenital heart disease, eight had PH associated with connective tissue disease, six had chronic thromboembolic PH, and three had hereditary hemorrhagic telangiectasia. Patients with diabetes mellitus or glucose intolerance, coronary artery disease, cardiomyopathy, primary valvular disease, or systemic arterial hypertension were excluded. FDG-PET imaging and speckle tracking echocardiography were performed in all subjects at intervals within 3 days. The study was approved by the Institutional Ethics Committee of Fu Wai Hospital. All patients enrolled in the study provided written informed consent.
FDG-PET Imaging
After a minimum 8-hour overnight fast, all patients were given 50 g oral glucose load 30 minutes before the injection of 185 MBq 18F-FDG. Data were acquired 60 minutes later using PET-CT (Truepoint Biography 64, Siemens Healthcare). CT transmission scanning (140 kV, 35 mA) was performed for attenuation correction. The acquisition time was 10 minutes for emission (three-dimensional mode). The cardiac cycle was divided into eight equal intervals. Attenuation-corrected data were reconstructed using an iterative algorithm (OSEM, 4 iterations, 8 subsets). The matrix was 128 × 128 pixels and the reconstruction zoom was 2.0. Short-axis slices of 3 mm thickness were obtained in PET images.
Gated 18F-FDG PET images were used to assess RV function as in our previous study.6 The parameters including right ventricular end-diastolic volume (RVEDV), right ventricular end-systolic volume (RVESV), and right ventricular ejection fraction (RVEF) were automatically calculated by QGS (version 3.1, Cedars-Sinai Medical Center, Los Angeles, CA, USA), and manual correction was processed in case of inadequate anatomical delineation.
In PET image processing, reconstructed transaxial images were reoriented into short-axis images using the Emory Cardiac Toolbox (version 3, Emory University, Atlanta, GA, USA). An interactive tool, as illustrated in Figure 1 was used to specify parameters characterizing RV location and size from the short-axis images. These RV parameters were input into a 3D sampling algorithm, which searched the maximal count circumferential profiles for each cardiac frame.8 The 3D samples were then processed by a RV phase analysis tool based on the first-harmonic Fourier approximation to calculate the onset of contraction for each sample, which is similar to the LV phase analysis technique published by Chen et al.9
PET image processing. (A) An interactive tool to identify RV parameters, including the apex, base, and circle enclosing the RV myocardium; (B) the phase polar map of the RV. The mean phases in the red and blue regions on (B) were used to calculate contraction delay between RV middle free wall and middle septal wall
The phase values of all RV samples were rendered on the RV polar map, which was then segmented to calculate the regional mean phases in RV middle free wall and RV middle septal wall, respectively. Thereafter, the contraction delay between the middle RV free wall and septal wall was calculated as the difference between their mean phases (RVPDPET). The RV phase standard deviation (RVSD) and histogram bandwidth (RVBW) were measured for assessing the RV dyssynchrony, which is similar to assessment of LV dyssynchrony described previously.9
Echocardiographic Examination and Speckle Tracking Analysis
Transthoracic echocardiographic examinations were performed on all subjects using iE33 (S5-1 transducer, Philips Medical Systems, Best, The Netherlands) by experienced cardiologists, blinded to FDG-PET imaging and clinical details. Two-dimensional grayscale images were acquired from apical four-chamber view in three consecutive cardiac cycles with a frame rate over 70 Hz. Speckle tracking analysis was performed on QLAB Quantification Software 8.1 (Philips Healthcare, Netherlands). Time to peak systolic strain (PSST) was analyzed in RV middle free wall and middle septal wall corresponding to the PET slices. The contraction delay difference between RV free wall and septum was defined as the difference of PSST between RV middle free wall and middle septal wall (RVPDEcho). PSST for each patient was standardized by R-R interval. Average values of three cardiac cycles were used in statistical analysis.
Statistical Analysis
Normally distributed continuous data were expressed as mean ± standard deviation (SD) and non-normally distributed continuous data were expressed as median with the first and the third quartiles. Categorical data were expressed as frequency with percentage (%). Differences between two groups were analyzed by the unpaired Student t test for continuous variables and the Fisher exact test for categorical variables. Correlations between the two parameters were determined by linear regression analysis. All statistical analyses were performed with SPSS 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient Characteristics
Table 1 shows baseline characteristics of total 54 patients. The mean age was 30.5 ± 9.3 years old. 43 (79.6%) patients were female. Most patients had heart failure with 1.9% (1/54) of patients in New York Heart Association class I, 38.9% in (21/54) class II, 55.6% (30/54) in class III, and 3.7% (2/54) in class IV. The mean RVEF was 31.5% ± 10.9% and the mean LVEF was 60.6% ± 14.5%.
The Correlation of FDG-PET and Speckle Tracking Echocardiography in Measurement of Contraction Delay Between RV Free Wall and Septum
The contraction delay between RV free wall and septum measured by FDG-PET imaging and speckle tracking echocardiography were compared to validate the accuracy of phase analysis of FDG-PET images.
In the 54 patients, FDG-PET imaging and echocardiography concordantly found that the contraction onset of the middle RV free wall was later than that of middle RV septal wall in 41 patients; meanwhile, these two methods concordantly found that the contraction onset of the middle RV free wall was earlier than that of middle RV septal wall in nine patients, achieving an overall agreement rate of 92.6% (50/54).
For a clearer comparison, RVPDEcho units of ms were converted to RVPDPET units of degrees using R-R interval. The median RVPDPET was 20.12° (interquartile range, 4.99°-30.10°) and the median RVPDEcho was 43.98° (interquartile range, 6.25-72.00°). RVPDPET was well correlated with RVPDEcho (r = 0.685, P < .001, Figure 2).
The Correlation of RV Dyssynchrony and Clinical Severity
The quantitative parameters for RV mechanical dyssynchrony, RVSD and RVBW, were derived from the phase histogram. The mean phase RVSD was 39.1° ± 12.0° and the mean phase RVBW was 123.9° ± 51.1°.
Figure 3 shows correlations between RVSD, RVBW, and RV systolic function, hemodynamic parameters, 6-minute walking distance (6MWD), and NT-proBNP. Both RVSD and RVBW had significantly negative correlations with cardiac index, RVEF, and 6MWD (RVSD: r = −0.532, P < .001; r = −0.551, P < .001; r = −0.544, P < .001; respectively, RVBW: r = −0.492, P < .001; r = −0.466, P < .001; r = −0.544, P < .001; respectively), and positively correlated with NT-proBNP (RVSD: r = 0.404, P < .01, RVBW: r = 0.349, P = .01).
Table 2 shows correlation coefficients between RV dyssynchrony parameters and hemodynamic parameters. There were no significant correlations between RVSD and RVBW with hemodynamic parameters (right atrial pressure, right ventricular systolic pressure, right ventricular end-diastolic pressure, mean pulmonary artery pressure, and total pulmonary resistance).
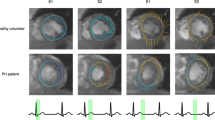
Representative images of RV function evaluation, PET phase analysis, and echocardiographic speckle tracking analysis of a PH patient was shown in Figure 4.
Representative images of RV function evaluation (A) and phase analysis (B) on FDG-PET and echocardiographic speckle tracking analysis (C) of a 31-year-old female patient with PH. RV dysfunction (RVEF = 34%) and RV dyssynchrony (RVSD = 21.2°, RVBW = 71°) were shown. The phase analysis and speckle tracking analysis concordantly found that contraction onset of the middle RV free wall (MAL, indicated by the red arrow) was later than that of middle RV septal wall (MIS, indicated by the blue arrow), RVPDPET was 22.5° and RVPDEcho was 42.7°
Discussion
To our knowledge, this is the first study to evaluate RV dyssynchrony of PH using FDG-PET imaging and explore the relationship between the RV dyssynchrony parameters measured by phase analysis of FDG-PET and speckle tracking echocardiographic measurement as well as clinical severity.
The phase analysis technique has been well established to measure LV mechanical dyssynchrony from gated SPECT myocardial perfusion imaging (MPI).9 It has been demonstrated to show good temporal resolution,10 high reproducibility,11 and good robustness with camera types,12 tracer dose,13 heart rate,14 and perfusion defects.15 The LV dyssynchrony parameters have been shown to correlate well with those measured by echocardiography.16,17 In a clinical study with 30 heart failure patients, there was a significant correlation of the parameters measured from FDG-PET and SPECT MPI (bandwidth: r = 0.88, P < .001; phase SD: r = 0.88, P < .001), resulting in an excellent clinical agreement of 93% to measure LV mechanical dyssynchrony.5 Nevertheless, there is no study to measure RV mechanical dyssynchrony from gated SPECT MPI or FDG-PET, mainly because RV in general has low counts and phase analysis is a count-based method. This study included FDG-PET images of patients with PH, where RV has comparable count levels to LV due to increased afterload and hypertrophic myocardium; therefore, the phase analysis technique developed for LV dyssynchrony measurement can be extended to RV dyssynchrony measurement. Echocardiography has been used to evaluate RV dyssynchrony in clinical practice18-21; however, there are limitations due to its reproducibility. Therefore, as an alternative technique, RV phase analysis using FDG-PET may add clinical values to the management of patients with PH.
We compared RV mechanical dyssynchrony measured from gated FDG-PET with that measured from speckle tracking echocardiography. Noteworthy, both speckle tracking and tissue Doppler echocardiography techniques are being clinically used to measure mechanical dyssynchrony; however, compared to tissue Doppler technique, speckle tracking has superior reproducibility,22 and it can measure the strain pattern caused by active wall thickening/thinning.22 In order to compare LV mechanical dyssynchrony measured by phase analysis from gated SPECT with that by speckle tracking echocardiography in,22 the middle LV wall was divided into six segments; it was found that the phase standard deviation (SD) correlated well with the SD of the times to peak systolic radial strains of two opposing LV walls measured by speckle tracking (r > 0.7, P < .001). In our study, the difference between mean phases of middle RV free wall and middle RV septum (RVPDPET) was validated by the difference between the times to peak systolic strains of middle RV free wall and middle RV septum (RVPDEcho), and a correlation of 0.685 was achieved (P < .001), indicating a good correlation to measure RV mechanical dyssynchrony by phase analysis and speckle tracking.
We further compared RV mechanical dyssynchrony with clinical severity indices to explore the clinical relevance of RV dyssynchrony in PH patients. Our data showed that RV mechanical dyssynchrony seemed to be more related to RV function in comparison to pulmonary vascular hemodynamics. Patients with worse RV systolic function, cardiac index may be related to worse RV systolic synchronicity. Noteworthy, RVSD and RVBW were well correlated with NT-proBNP and 6MWD, two well-studied RV dysfunction markers in PH. In response to ventricular pressure and volume overload,23 NT-proBNP can be used to detect RV systolic dysfunction in PH,24 while 6MWD reflecting exercise capacity has a strong and independent association with mortality.25 Although the underlying mechanism of RV mechanical dyssynchrony remains not fully elucidated, previous studies have showed that chronic RV pressure overload would cause RV dilation and increased RV wall stress, leading to prolonged contraction duration and RV dyssynchrony.26,27 RV dyssynchrony impairs RV performance, leads to a paradoxical septum shift, and subsequently influences LV filling. Therefore, RV dyssynchrony can be interpreted as a sign of RV failure3 and its correlations with RV functional parameters, NT-proBNP and 6MWD can be explained.
Although the relationships between RV dyssynchrony and pulmonary vascular hemodynamics were not significant in current study, several reasons possibly explained this discrepancy from other reports.28,29 Firstly, subjects enrolled in this study included PH patients with heterogeneous etiology who may present different RV compensatory capability. Patients with higher pulmonary artery pressure may be more capable to cope with the increased RV afterload and develop less severe RV failure compared to patients with lower pulmonary artery pressure. Moreover, approximately 70.4% patients in current study had moderate to severe tricuspid regurgitation, which would compromise RV afterload to a certain extent. In these cases, RV function as well as RV dyssynchrony may not necessarily link to pulmonary vascular pressure.
Additionally, applicability of FDG-PET imaging in PH has been increasingly explored. FDG-PET imaging can quantify RV myocardial glucose metabolism in PH. Several studies have reported that PH patients had an increased glucose uptake in the RV and it correlated with the severity of RV pressure overload and RV systolic dysfunction30,31 and may be a predictor for clinical worsening.32 Moreover, data from our previous study showed RV volume and ejection fraction assessed by FDG-PET imaging were in moderate to high correlation with cardiac magnetic resonance imaging, suggesting FDG-PET imaging can be an available method for measurement of RV volume and function.6 This study showed the feasibility of FDG-PET imaging to evaluate RV mechanical dyssynchrony. Therefore, employing FDG-PET imaging in PH patients allows for the simultaneous assessment of RV function, glucose metabolism, and mechanical dyssynchrony without additional acquisition. This enables comprehensive evaluation of RV performance and indicates a potential usefulness of FDG-PET imaging as a biomarker in PH patients.
Limitations
Firstly, no control group was included in this study because of low counts in the thin RV wall in normal subjects. Secondly, the current findings were based on a relatively small subset of PH patients with heterogeneous etiology. Given the lack of a standardization of imaging techniques to evaluate ventricular dyssynchrony, employing two-dimensional speckle tracking echocardiography as reference may not be optimal. Data to be collected in a large cohort of patients at baseline and during follow-up would further examine the feasibility and applicability of RV dyssynchrony evaluation on FDG-PET imaging, and a subgroup comparison of different PH patients can be analyzed to determine the clinical relevance.
New Knowledge Gained
This study to our knowledge firstly studied the feasibility of applying phase analysis on FDG-PET imaging to RV mechanical dyssynchrony measurement. In patients with PH, RV mechanical dyssynchrony evaluated by phase analysis on FDG-PET was well correlated with echocardiographic dyssynchrony parameters and was significantly related to RV dysfunction.
Conclusions
In patients with PH, phase analysis on gated FDG-PET imaging could effectively evaluate RV mechanical dyssynchrony and has good correlation with speckle tracking echocardiography. The RV dyssynchrony parameters measured by phase analysis correlated well with RV functional and clinical parameters, but not with pulmonary vascular hemodynamics. With the new phase analysis tool, FDG-PET imaging can provide a method for the comprehensive assessment of RV function, glucose metabolism, and mechanical dyssynchrony for patients with PH.
Abbreviations
- PH:
-
Pulmonary hypertension
- RV:
-
Right ventricle/ventricular
- LV:
-
Left ventricle/ventricular
- PET:
-
Positron emission tomography
- FDG:
-
Fluorodeoxyglucose
- RVPD:
-
Contraction delay difference between RV free wall and septum
- RVSD:
-
RV phase standard deviation
- RVBW:
-
RV phase histogram bandwidth
- PSST:
-
Time to peak systolic strain
References
Rasmussen JT, Thenappan T, Benditt DG, Weir EK, Pritzker MR. Is cardiac resynchronization therapy for right ventricular failure in pulmonary arterial hypertension of benefit? Pulm Circ 2014;4:552-9.
Ryan JJ, Archer SL. The right ventricle in pulmonary arterial hypertension: Disorders of metabolism, angiogenesis and adrenergic signaling in right ventricular failure. Circ Res 2014;115:176-88.
Vonk Noordegraaf A, Haddad F, Bogaard HJ, Hassoun PM. Noninvasive imaging in the assessment of the cardiopulmonary vascular unit. Circulation 2015;131:899-913.
Boogers MJ, Chen J, Veltman CE, van Bommel RJ, Mooyaart EA, Al-Younis I, et al. Left ventricular diastolic dyssynchrony assessed with phase analysis of gated myocardial perfusion SPECT: A comparison with tissue Doppler imaging. Eur J Nucl Med Mol Imaging 2011;38:2031-9.
Pazhenkottil AP, Buechel RR, Nkoulou R, Ghadri JR, Herzog BA, Husmann L, et al. Left ventricular dyssynchrony assessment by phase analysis from gated PET-FDG scans. J Nucl Cardiol 2011;18:920-5.
Wang L, Zhang Y, Yan C, He J, Xiong C, Zhao S, et al. Evaluation of right ventricular volume and ejection fraction by gated 18F-FDG PET in patients with pulmonary hypertension: Comparison with cardiac MRI and CT. J Nucl Cardiol 2013;20:242-52.
Galiè N, Hoeper MM, Humbert M, Torbicki A, Vachiery JL, Barbera JA, et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: The Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur Heart J 2009;30:2493-537.
Garcia EV, Cooke CD, Van Train KF, Folks R, Peifer J, DePuey EG, et al. Technical aspects of myocardial SPECT imaging with technetium-99m sestamibi. Am J Cardiol 1990;66:23E-31E.
Chen J, Garcia EV, Folks RD, Cooke CD, Faber TL, Tauxe EL, et al. Onset of left ventricular mechanical contraction as determined by phase analysis of ECG-gated myocardial perfusion SPECT imaging: Development of a diagnostic tool for assessment of cardiac mechanical dyssynchrony. J Nucl Cardiol 2005;12:687-95.
Chen J, Faber TL, Cooke CD, Garcia EV. Temporal resolution of multiharmonic phase analysis of ECG-gated myocardial perfusion SPECT studies. J Nucl Cardiol 2008;15:383-91.
Trimble MA, Velazquez EJ, Adams GL, Honeycutt EF, Pagnanelli RA, Barnhart HX, et al. Repeatability and reproducibility of phase analysis of gated single-photon emission computed tomography myocardial perfusion imaging used to quantify cardiac dyssynchrony. Nucl Med Commun 2008;29:374-81.
Pazhenkottil AP, Buechel RR, Herzog BA, Nkoulou RN, Valenta I, Fehlmann U, et al. Ultrafast assessment of left ventricular dyssynchrony from nuclear myocardial perfusion imaging on a new high-speed gamma camera. Eur J Nucl Med Mol Imaging 2010;37:2086-92.
Aljaroudi W, Koneru J, Heo J, Iskandrian AE. Impact of ischemia on left ventricular dyssynchrony by phase analysis of gated single photon emission computed tomography myocardial perfusion imaging. J Nucl Cardiol 2011;18:36-42.
AlJaroudi W, Iqbal F, Heo J, Iskandrian AE. Relation between heart rate and left ventricular mechanical dyssynchrony in patients with end-stage renal disease. Am J Cardiol 2011;107:1235-40.
Cheung A, Zhou Y, Faber TL, Garcia EV, Zhu L, Chen J. The performance of phase analysis of gated SPECT myocardial perfusion imaging in the presence of perfusion defects: A simulation study. J Nucl Cardiol 2012;19:500-6.
Henneman MM, Chen J, Ypenburg C, Dibbets P, Bleeker GB, Boersma E, et al. Phase analysis of gated myocardial perfusion single-photon emission computed tomography compared with tissue Doppler imaging for the assessment of left ventricular dyssynchrony. J Am Coll Cardiol 2007;49:1708-14.
Marsan NA, Henneman MM, Chen J, Ypenburg C, Dibbets P, Ghio S, et al. Left ventricular dyssynchrony assessed by two three-dimensional imaging modalities: Phase analysis of gated myocardial perfusion SPECT and tri-plane tissue Doppler imaging. Eur J Nucl Med Mol Imaging 2008;35:166-73.
López-Candales A, Dohi K, Rajagopalan N, Suffoletto M, Murali S, Gorcsan J, et al. Right ventricular dyssynchrony in patients with pulmonary hypertension is associated with disease severity and functional class. Cardiovasc Ultrasound 2005;3:23.
Hardziyenka M, Surie S, de Groot JR, de Bruin-Bon HA, Knops RE, Remmelink M, et al. Right ventricular pacing improves haemodynamics in right ventricular failure from pressure overload: an open observational proof-of-principle study in patients with chronic thromboembolic pulmonary hypertension. Europace 2011;13:1753-9.
Smith BC, Dobson G, Dawson D, Charalampopoulos A, Grapsa J, Nihoyannopoulos P. Three-dimensional speckle tracking of the right ventricle: Toward optimal quantification of right ventricular dysfunction in pulmonary hypertension. J Am Coll Cardiol 2014;64:41-51.
Badagliacca R, Poscia R, Pezzuto B, Papa S, Gambardella C, Francone M, et al. Dario Vizza C. Right ventricular dyssynchrony in idiopathic pulmonary arterial hypertension: Determinants and impact on pump function. J Heart Lung Transplant 2015;34:381-9.
Hsu TH, Huang WS, Chen CC, Hung GU, Chen TC, Kao CH, et al. Left ventricular systolic and diastolic dyssynchrony assessed by phase analysis of gated SPECT myocardial perfusion imaging: A comparison with speckle tracking echocardiography. Ann Nucl Med 2013;27:764-71.
Lador F, Soccal PM, Sitbon O. Biomarkers for the prognosis of pulmonary arterial hypertension: Holy Grail or flying circus? J Heart Lung Transplant 2014;33:341-3.
Blyth KG, Groenning BA, Mark PB, Martin TN, Foster JE, Steedman T, et al. NT-proBNP can be used to detect right ventricular systolic dysfunction in pulmonary hypertension. Eur Respir J 2007;29:737-44.
Miyamoto S, Nagaya N, Satoh T, Kyotani S, Sakamaki F, Fujita M, et al. Clinical correlates and prognostic significance of six-minute walk test in patients with primary pulmonary hypertension: Comparison with cardiopulmonary exercise testing. Am J Respir Crit Care Med 2000;161:487-92.
Ichikawa K, Dohi K, Sugiura E, Sugimoto T, Takamura T, Ogihara Y, et al. Ventricular function and dyssynchrony quantified by speckle-tracking echocardiography in patients with acute and chronic right ventricular pressure overload. J Am Soc Echocardiogr 2013;26:483-92.
Mauritz GJ, Vonk-Noordegraaf A, Kind T, Surie S, Kloek JJ, Bresser P, et al. Pulmonary endarterectomy normalizes interventricular dyssynchrony and right ventricular systolic wall stress. J Cardiovasc Magn Reson 2012;14:5.
Kong D, Shu X, Dong L, Pan C, Cheng L, Yao H, et al. Right ventricular regional systolic function and dyssynchrony in patients with pulmonary hypertension evaluated by three-dimensional echocardiography. J Am Soc Echocardiogr 2013;26:649-56.
Haeck ML, Höke U, Marsan NA, Holman ER, Wolterbeek R, Bax JJ, et al. Impact of right ventricular dyssynchrony on left ventricular performance in patients with pulmonary hypertension. Int J Cardiovasc Imaging 2014;30:713-20.
Oikawa M, Kagaya Y, Otani H, Sakuma M, Demachi J, Suzuki J, et al. Increased [18F]fluorodeoxyglucose accumulation in right ventricular free wall in patients with pulmonary hypertension and the effect of epoprostenol. J Am Coll Cardiol 2005;45:1849-55.
Yang T, Wang L, Xiong CM, He JG, Zhang Y, Gu Q, et al. The ratio of 18F-FDG activity uptake between the right and left ventricle in patients with pulmonary hypertension correlates with the right ventricular function. Clin Nucl Med 2014;39:426-30.
Tatebe S, Fukumoto Y, Oikawa-Wakayama M, Sugimura K, Satoh K, Miura Y, et al. Enhanced [18F]fluorodeoxyglucose accumulation in the right ventricular free wall predicts long-term prognosis of patients with pulmonary hypertension: a preliminary observational study. Eur Heart J Cardiovasc Imaging 2014;15:666-72.
Acknowledgments
We would like to thank Dr Qizhe Cai from Department of echocardiography, Fuwai Hospital, National Center of Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College for her kind help on speckle tracking analysis. This research was supported by Grants from National Natural Science Foundation of China (Project Number: 81371586), National Key Technology R&D Program of China (Project Number: 2011BAI11B15). This research was also partly supported by a grant from the American Heart Association (15POST22690035, PI: Weihua Zhou, Ph.D.) and a New Faculty startup grant from the University of Southern Mississippi (Project Number: DE01791).
Disclosures
Drs Chen and Garcia receive royalties from the sales of Emory Cardiac Toolbox. The terms of this arrangement have been reviewed and approved by Emory University in accordance with its conflict-of-interest practice. Drs Wang, Zhou, Liang, Yang, and Fang declare that he/she has no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Lei Wang, Weihua Zhou, and Yu Liang have contributed equally.
See related editorial, doi:10.1007/s12350-015-0383-1.
Rights and permissions
About this article
Cite this article
Wang, L., Zhou, W., Liang, Y. et al. Right ventricular dyssynchrony in pulmonary hypertension: Phase analysis using FDG-PET imaging. J. Nucl. Cardiol. 24, 69–78 (2017). https://doi.org/10.1007/s12350-015-0341-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-015-0341-y