Abstract
Introduction
The purpose of this study was to evaluate baseline and postoperative factors affecting outcomes after vitrectomy for diabetic macular edema (DME) using optical coherence tomography (OCT).
Methods
Vitrectomy combined with inner limiting membrane (ILM) peeling and additional laser photocoagulation therapy was performed on 36 eyes of 30 DME patients. Evaluations included the logarithm of the minimal angle of resolution (logMAR), best-corrected visual acuity (BCVA) and OCT parameters at baseline and 1, 3, 6, and 12 months postoperatively. Correlations between OCT parameters and BCVA were assessed at each follow-up visit. Correlations among postoperative BCVA and preoperative BCVA, foveal macular thickness (FMT), outer foveal thickness (OFT), and photoreceptor outer segment (PROS) length were evaluated using multiple regression analysis.
Results
BCVA significantly improved from 0.50 ± 0.25 to 0.34 ± 0.26 at 12 months postoperatively (P < 0.001). Mean FMT improved significantly from 526.4 ± 120.4 to 384.6 ± 120.5 at 1 month, 325.2 ± 100.3 at 3 months, 304.1 ± 102.5 at 6 months and 274.2 ± 86.6 μm at 12 months postoperatively (P < 0.001, respectively). OFT 1 month after surgery was significantly decreased 46.5 ± 14.7–40.2 ± 14.4 μm (P = 0.017), although at 3, 6, and 12 months it did not differ from the baseline value. PROS length 1 month after surgery significantly decreased from 31.7 ± 6.9–28.8 ± 6.8 μm (P = 0.015) and that at 3 months and 6 months recovered to the baseline value. PROS length 12 months after surgery was significantly increased to 34.3 ± 7.2 μm from baseline (P = 0.023). Mean FMT was not correlated with BCVA at any time point. Mean OFT and PROS length at 3, 6, and 12 months were correlated with BCVA. In multiple regression analysis, PROS length had the greatest effect on VA 12 months postoperatively (P = 0.0262, standard regression coefficient = −0.366).
Conclusion
Current surgery helps DME patients to maintain VA and foveal structures. The results suggest that PROS length predicts visual outcome in DME patients following vitrectomy with ILM peeling and additional laser photocoagulation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diabetic macular edema (DME) is a leading cause of severe visual loss in diabetic retinopathy patients [1]. In the past, the most widely used nonsurgical treatments for DME included intravitreal injection of triamcinolone acetonide (IVTA) [2], and focal/grid photocoagulation in which laser shots are applied directly to leaking microaneurysmas (focal treatment) or are delivered in a grid pattern on the thickened retina [3]. Recently, intravitreal injection of ranibizumab [4] or aflibercept [5] have been widely used worldwide. Resolution of the macular edema often leads to improved visual acuity (VA). However, despite complete resolution of macular edema, some patients have a poor visual outcome, and the results of recent case series show that macular edema persists in some patients despite multiple intravitreal anti-vascular endothelial growth factor (VEGF) injections over a long treatment period [6]. This discrepancy is caused by irreversible structural changes due to long-standing macular edema [6].
Vitrectomy has been advocated for the treatment of DME refractory to nonsurgical treatment [7, 8]. The postulated mechanisms of action of vitrectomy include the removal of possible sources of traction [9], improving transvitreal oxygenation of the retina [10], removal of the posterior hyaloids that harbor vasopermeable factors [11], and removal of condensed chemical mediators that promote vascular permeability [12]. Although several interventions have been applied clinically, it remains to be determined how individual interventions provide beneficial effects for visual function, which can lead to customized medicine [9].
Recent technological advances in optical coherence tomography (OCT) have enabled us to identify the external limiting membrane and photoreceptor layers. We previously reported that photoreceptor outer segment (PROS) length is a strong indicator of visual function and a prognostic factor for postoperative VA in patients with idiopathic epiretinal membrane (ERM) who underwent vitrectomy [13].
The purposes of this study are to evaluate the efficacy of vitrectomy and the correlations among various foveal microstructures using spectral domain (SD)-OCT images and VA before and after surgery in patients with DME.
Methods
We retrospectively studied 36 eyes of 30 patients with DME treated with 25-gauge transconjunctival sutureless vitrectomy (TSV) and inner limiting membrane (ILM) peeling at St. Marianna University School of Medicine Hospital between April 2013 and June 2015. Patients with type 2 diabetes mellitus who had a reduction in best-corrected visual acuity (BCVA) due to DME were eligible for this clinical trial. The criterion for thickening of the central macula was defined as a foveal macular thickness (FMT) of 250 μm based on SD-OCT (Cirrus HD-OCT; Carl Zeiss Meditec, Dublin, CA, USA). All eyes had received some therapies prior to surgery. This study was approved by the Institutional Review Committee of St. Marianna University School of Medicine, and informed written consent was obtained from all patients before study enrollment. We registered this study in the University hospital Medical Information Network (UMIN ID: 000,026,906). All patients were followed up for at least 12 months postoperatively, until June 2016. The procedures used conformed to the tenets of the Declaration of Helsinki.
Patients were excluded if they had a history of prior scleral buckling and pars plana vitrectomy, and high myopia with a refractive error of greater than −8.00 diopters, severe cataract of higher than grade 3, glaucoma, or type 1 diabetes mellitus, and if grid or focal macular laser and/or panretinal photocoagulation (PRP), subtenon TA (STTA), intravitreal TA (IVTA) and intravitreal anti-VEGF injection had been performed within 3 months before study enrollment. Eyes shown to have a disrupted inner segment/outer segment (IS/OS) junction line (now usually called ellipsoid zone) and subretinal fluid in OCT were also excluded, since it would have been difficult to evaluate PROS length correctly.
All eyes of 30 patients who underwent 25-gauge TSV using the Alcon Constellation Vision System (Alcon Laboratories, Fort Worth, TX, USA) including a 3-port trocar cannula system (Total Plus Pak). ILM peeling was performed in all eyes with triamcinolone acetonide. Even if prior PRP had been performed, additional PRP was performed in the far peripheral area whether or not nonperfused areas (NPAs) defined by fluorescein angiography (FA) were present. Simultaneous phacoemulsification with intraocular lens implantation was carried out on all phakic eyes before the scleral incision for vitrectomy (excluding eyes with severe cataract of higher than grade 3). All cataract surgeries were performed through a clear corneal 2.4-mm incision, and all procedures were performed by one surgeon (J.K.).
The BCVA and SD-OCT findings were measured before and after surgery at 1, 3, 6, and 12 months. The BCVA was converted to the logarithm of the minimal angle of resolution (logMAR) units for statistical analysis. We assessed the associations among preoperative parameters, including logMAR VA, foveal macular thickness [FMT; the distance between the vitreoretinal surface and the retinal pigment epithelium (RPE) at the foveal center], outer foveal thickness (OFT; the distance between the ELM and RPE), PROS length (the distance between the IS/OS junction and RPE), and postoperative VA 12 months after surgery (Fig. 1). The FMT measurements were derived from the software (Cirrus 3.0; Carl Zeiss Meditec) provided by the manufacturer. Detailed image analysis was performed in the outer layer. The central PROS length and OFT measurements at the foveal center were performed using Image J software v.1.46 (National Institutes of Health, Bethesda, MD, USA; available at http://rsbweb.nih.gov) available from July 2011 and were true to scale. Within this selected area, longitudinal reflectivity profiles, arranged in a cross-sectional parallel manner, were calculated (Fig. 2). The OCT scans through the outer layer showed 3 clearly distinguishable, highly reflective bands [peak (P) 1–3; Fig. 2c]. These highly reflective bands represented the RPE (P1), IS/OS junction (P2), and ELM (P3). The PROS length (the distance between P1 and P2) and OFT (the distance between P1 and P3) measurements were performed on longitudinal reflectivity profiles. The repeatability of this manual analysis was investigated using a subset of both five images of healthy eyes and five images of study eyes. The 3-sigma repeatability of the image analysis was found to be not less than 2.8 μm, as previously reported [21]. The factors influencing postoperative BCVA were evaluated using multiple regression analysis. We assessed the correlation between foveal microstructures and final VA and investigated the prognostic factors related to PROS length that may be predictive of final VA in DME after surgery.
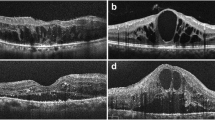
Data extraction from grayscale images. a Fluorescein angiography image of a patient with DME. b Complete 24-bit OCT image. c Enlarged extract from the outer layer in (b). Dashed line line scan from which the single-reflectivity profile (c) was calculated. d Three distinct peaks within the longitudinal reflectivity profile were easily detected. Reflectivity is in 0–255 levels of gray (x axis); scan depth is in micrometers (y axis). DME diabetic macular edema, OCT optical coherence tomography
a Changes in the mean logMAR visual acuity (VA) from baseline to 12 months after surgery. VA at 3 and 6 months after surgery was significantly improved compared with preoperatively. b Changes in the mean FMT from baseline to 12 months after surgery. c, d Changes in the mean PROS length and OFT from baseline to 12 months after surgery. The two parameters showed a similar clinical course. e Distribution of changes in FMT from baseline. Box-whisker plot demonstrating mean (dashed horizontal line), median (solid horizontal line), and 25th–75th percentiles (extremes of the box). logMAR logarithm of the minimal angle of resolution, FMT foveal macular thickness, PROS photoreceptor outer segment, OFT outer foveal thickness
Statistical Analysis
All statistical analyses were performed using StatView software (Abacus Concepts, Berkeley, CA, USA, 1992). Data correlations between final BCVA and SD-OCT characteristics or clinical features were investigated using the Spearman rank-correlation test. P values of less than 0.05 were considered to represent statistically significant differences. The Wilcoxon rank-sum test was used to compare pre- and postoperative BCVA and OCT parameters. P values of less than 0.05 were also considered to represent statistically significant differences in this analysis.
Results
The baseline characteristics and clinical data of the patients are shown in Table 1. Thirty eyes (83.3%) had received prior treatment for DME. Vitrectomy and ILM peeling were performed in all eyes without any intraoperative complications (iatrogenic tearing, severe retinal damage during ILM peeling). During follow-up to 12 months, no eyes required additional treatment such as intravitreal anti-VEGF, STTA, IVTA or laser photocoagulation, and no severe postoperative complications such as iris neovascularization or endophthalmitis occurred. A postoperative increase in IOP was noted in one eye, which was successfully treated with glaucoma medication.
The mean logMAR BCVA showed significant improvement from 0.50 ± 0.25 at baseline to 0.42 ± 0.25 at 3 months, 0.39 ± 0.25 at 6 months, and 0.34 ± 0.26 at 12 months postoperatively (P = 0.038, P = 0.012 and P < 0.001, respectively) (Fig. 2a). Three eyes had worsening of VA, defined as a change of >0.2 logMAR units 12 months postoperatively.
The mean foveal macular thickness (FMT) improved significantly from 526.4 ± 120.4 μm (interquartile range, 429–618 μm) at baseline to 384.6 ± 120.5 μm at 1 month, 325.2 ± 100.3 μm at 3 months, 304.1 ± 102.5 μm at 6 months, and 274.2 ± 86.6 μm at 12 months postoperatively (P < 0.001, respectively) (Fig. 2b). Postoperative changes in FMT are shown in Fig. 2e. A reduction of FMT to <250 μm occurred in 3 eyes (8.3%) at 1 month, 5 eyes (13.9%) at 3 months, 15 eyes (41.2%) at 6 months, and 15 eyes (41.2%) at 12 months postoperatively.
Time Course of Changes in PROS Length and OFT
OFT decreased significantly from 46.5 ± 14.7 μm at baseline to 40.2 ± 14.4 μm 1 month after surgery (P = 0.017), although OFT 3, 6, and 12 months after surgery was not significantly different from the baseline value (43.3 ± 15.2, 45.5 ± 13.0, and 48.6 ± 13.6 μm, P > 0.05, respectively) (Fig. 2c). PROS length 1 month after surgery decreased significantly from 31.7 ± 6.9 μm at baseline to 28.8 ± 6.8 μm (P = 0.015) and that at 3 and 6 months recovered to 32.9 ± 9.6 μm (P = 0.45 vs. baseline) and 32.8 ± 7.1 μm (P = 0.12 vs. baseline), respectively (Fig. 2d). However, PROS length 12 months after surgery increased significantly to 34.3 ± 7.2 μm compared with baseline (P = 0.023).
Correlation Between SD-OCT Parameters and BCVA at Each Time Point
Figure 3 shows correlations between FMT and BCVA. FMT was not significantly correlated with BCVA at any time point. OFT at 3, 6, and 12 months was significantly correlated with BCVA at each time point (3 months: r = 0.391, P = 0.012, 6 months: r = 0.253, P = 0.035, 12 months: r = 0.253, P = 0.022) (Fig. 4). PROS length at 3, 6, and 12 months was significantly correlated with BCVA at each time point (3 months: r = 0.402, P = 0.002; 6 months: r = 0437, P = 0.012; 12 months: r = 0.49, P < 0.001) (Fig. 5).
Factors Affecting Postoperative BCVA in Multiple Regression Analysis
In multiple regression analysis, the model with the highest adjusted R 2 was determined by stepwise selection (adjusted R 2 = 0.331). The model R 2 for this analysis was 0.386. In this model, PROS length had the most significant affect on VA 6 months after surgery (P = 0.0262, standard regression coefficient = −0.366) (Table 2).
Discussion
In this study, FMT was markedly reduced and VA was significantly improved after surgery. A marked reduction in FMT was observed in many eyes throughout the 12-month follow-up period. Additionally, BCVA improved significantly during the 12-month follow-up. A possible explanation for these is that the ILM was removed in all patients in this study. ILM removal was suggested to have a role in the resolution of nontractional DME by eliminating the tangential traction force [14]. The efficacy of ILM peeling has remained controversial in spite of the many studies conducted. Kumagai et al. reported that visual outcomes after ILM peeling were better than those associated with preservation of the ILM in univariate analysis [15]. The results of Dillinger and Mester indicated that discrepancies between anatomical and functional results after ILM removal in chronic DME were likely caused by structural changes in the macula due to long-standing edema [16]. However, we included patients who had preserved IS/OS lines in this study, meaning that their photoreceptor damage may have been less than in patients in the previous studies and that such damage may be reversible. Moreover, the effects of cataract surgery may exaggerate the effects of vitrectomy; however, we believe that the exaggeration was minimal because we excluded eyes with cataract higher than grade 3.
One reason for the marked reduction in FMT in this study may be the additional laser photocoagulation performed in all eyes. VEGF is generally released from hypoxic or ischemic retinas. Wessel et al. reported a significant association between the degree of DME and width of NPAs [17]. This suggests that retinal ischemia is associated with the pathogenesis of DME. Recently, targeted retinal photocoagulation (TRP) has been designed to treat areas of retinal capillary NPAs and intermediate retinal ischemic zones in proliferative diabetic retinopathy [18]. Takamura et al. showed the effects of peripheral TRP of NPAs on the recurrence of DME after intravitreal injection of bevacizumab (IVB) [19]. They found that TRP not only prevented the increase in CMT but also improved BCVA after IVB. In the current study, the mean FMT improved significantly from 526.4 ± 120.4 at baseline to 274.2 ± 86.6 μm at 12 months postoperatively.
Moreover, no eyes required additional treatment. We believe that the effect of additional laser photocoagulation contributes to the reduction of VEGF production from ischemic areas, which may lead to the recurrence of macular edema. However, longer follow-up periods are needed to assess the surgical outcomes in eyes with DME.
We demonstrated that PROS length and OFT were correlated with VA at 3, 6, and 12 months postoperatively. Moreover, preoperative PROS length was most significantly correlated with VA 12 months postoperatively and was a potent predictor of visual prognosis in multiple regression analysis. Several groups reported on prognostic factors after vitrectomy for DME, one of which was retinal thickness at the fovea. Greater VA improvement occurred in eyes with worse baseline VA [20]. Murakami et al. reported that total thickness in the nasal quadrant is the most relevant for predicting postoperative logMAR VA [21]. Disorganization of the retinal inner layers is a correlated predictive biomarker for VA which is independent of central retinal thickness in eyes with baseline center-involved DME [22]. Many researchers showed better postoperative VA in patients with good baseline vision [15, 20, 21], and our results also showed that baseline VA was an independent factor predicting postoperative visual outcome. However, our multiple regression analysis showed that preoperative PROS length has a higher correlation with visual outcome than preoperative VA. This suggests that PROS length is a better indicator of final visual outcome after DME surgery. Forooghian et al. reported that PROS length has a qualitative correlation with VA in DME patients; eyes with poor VA had shorter PROS length [23]. We also showed a correlation between VA and PROS length, and OFT also correlated with VA at 3, 6, and 12 months postoperatively, although the two parameters were not correlated at baseline and 1 month postoperatively. One possible reason for this is that most eyes had greater intraretinal edema at earlier visits. Only 3 eyes (8.3%) showed a reduction of FMT to <250 μm at 1 month postoperatively in contrast to 5 eyes (13.9%) at 3 months, 15 eyes (41.2%) at 6 months, and 15 eyes (41.2%) at 12 months postoperatively. This condition might deteriorate the intraretinal visual pathway. Jansson et al. reported that mean retinal thickness was significantly associated with the b-wave amplitude of photopic single-flash and 30-Hz flicker responses [24]. Thus, PROS length and OFT at baseline and 1 month did not correlate with VA due to the influence of macular edema. These results suggest that PROS length and OFT are parameters predicting visual outcome rather than reflecting postoperative VA after DME surgery. Although OFT and PROS length were estimated in similar regions, preoperative PROS length was more strongly correlated with final VA than was OFT. Estimation of PROS length may be more accurate than estimation of OFT in predicting visual function, because the visual substance is an opsin and all-trans retinal complex and is present in the outer segment [21, 25]. Postoperatively, changes in PROS length and OFT were similar over time. Decreases in PROS length and OFT were noted 1 month after surgery, and both parameters returned to preoperative values at 3 months. These results suggest that the decrease in PROS length and OFT at 1 month is caused by a perioperative traumatic mechanism such as postoperative inflammation or intraoperative injury, which reflects the extent of reversible damage to the photoreceptor layer [21].
Study Limitation
This study had certain limitations. The number of eyes was relatively small, the study was retrospective, and the follow-up period was short. The lack of a concurrent control group is also a study weakness. DME does not occur in isolation, and the patients’ diabetes, blood pressure and cholesterol control and general health all have significant impact on DME; however, we did not analyze these. Also, macular ischemia is a very relevant piece of information in refractory DME. However, we did not routinely perform FA after surgery. We further consider that microperimetry is necessary for correlation between the morphological change and functional change, but this was not performed in the current study. These conditions might affect the results of our study.
Conclusion
In summary, our study results suggest that vitrectomy combined with ILM peeling and additional laser photocoagulation therapy is useful for DME patients to maintain VA and foveal structures. The efficacy of vitrectomy compared with other therapies in the management of DME remains uncertain as the potential benefits and risks have not been clearly defined. Recently, anti-VEGF therapy for DME has begun to become established in many parts of the world. However, many patients are burdened by the repeated injections. Further controlled studies with longer follow-up durations are needed to evaluate the surgical outcomes in eyes with DME in the era of anti-VEGF therapy. However, we believe that early surgical intervention may be helpful for eyes with chronic DME persisting after other forms of treatment. We demonstrated that PROS length is a potent predictor of visual outcome in patients with DME. However, PROS length cannot explain all the variation in VA, and other factors such as macular ischemia and retinal cell function may also be important.
References
Klein R, Klein BE, Moss SE, Linton KL. Retinopathy in adults with newly discovered and previously diagnosed diabetes mellitus. Ophthalmology. 1992;99:58–62.
Martidis A, Duker JS, Greenberg PB, Rogers AH, Puliafito CA, Reichel E, et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology. 2002;109:920–7.
ETDRS-Group. Treatment techniques and clinical guidelines for photocoagulation of diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report Number 2. Ophthalmology. 1987;94:761–74.
Chen DW, Heier JS, Topping TM, Duker JS, Bankert JM. A pilot study of multiple intravitreal injections of ranibizumab in patients with center-involving clinically significant diabetic macular edema. Ophthalmology. 2006;113:1706–12.
Korobelnik JF, Do DV, Schmidt-Erfurth U, Boyer DS, Holz FG, Heier JS, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121:2247–54.
Massin P, Bandello F, Garweg JG, Hansen LL, Harding SP, Larsen M, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RE-SOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes Care. 2010;33:2399–405.
Lewis H, Abrams GW, Blumenkranz MS, Campo RV. Vitrectomy for diabetic macular traction and edema associated with posterior hyaloidal traction. Ophthalmology. 1992;99(6):753–9.
Tachi N, Ogino N. Vitrectomy for diffuse macular edema in cases of diabetic retinopathy. Am J Ophthalmol. 1996;122:258–60.
Kimura T, Kiryu J, Nishiwaki H, Oh H, Suzuma K, Watanabe D, et al. Efficacy of surgical removal of the internal limiting membrane in diabetic cystoid macular edema. Retina. 2005;25:454–61.
Stefansson E. The therapeutic effects of retinal laser treatment and vitrectomy: a theory based on oxygen and vascular physiology. Acta Ophthalmol Scand. 2001;79:435–40.
Funatsu H, Yamashita H, Ikeda T, Mimura T, Shimizu E, Hori S, et al. Relation of diabetic macular edema to cytokines and posterior vitreous detachment. Am J Ophthalmol. 2003;135:321–7.
Otani T, Kishi S. A controlled study of vitrectomy for diabetic macular edema. Am J Ophthalmol. 2002;134:214–9.
Shiono A, Kogo J, Klose G, Takeda H, Ueno H, Tokuda N, et al. Photoreceptor outer segment length: a prognostic factor for idiopathic epiretinal membrane surgery. Ophthalmology. 2013;120(4):788–94.
Gandorfer A, Messmer EM, Ulbig MW, Kampik A. Resolution of diabetic macular edema after surgical removal of the posterior hyaloid and the inner limiting membrane. Retina. 2000;20(2):126–33.
Kumagai K, Furukawa M, Ogino N, Larson E, Iwaki M, Tachi N. Long-term follow-up of vitrectomy for diffuse nontractional diabetic macular edema. Retina. 2009;29(4):464–72.
Dillinger P, Mester U. Vitrectomy with removal of the internal limiting membrane in chronic diabetic macular oedema. Graefes Arch Clin Exp Ophthalmol. 2004;242(8):630–7.
Wessel MM, Nair N, Aaker GD, Ehrlich JR, D’Amico DJ, Kiss S. Peripheral retinal ischaemia, as evaluated by ultra-widefield fluorescein angiography, is associated with diabetic macular oedema. Br J Ophthalmol. 2012;96(5):694–8.
Muqit MM, Marcellino GR, Henson DB, Young LB, Patton N, Charles SJ, et al. Optos-guided pattern scan laser (Pascal)-targeted retinal photocoagulation in proliferative diabetic retinopathy. Acta Ophthalmol. 2013;91(3):251–8.
Takamura Y, Tomomatsu T, Matsumura T, Arimura S, Gozawa M, Takihara Y, et al. The effect of photocoagulation in ischemic areas to prevent recurrence of diabetic macular edema after intravitreal bevacizumab injection. Invest Ophthalmol Vis Sci. 2014;55(8):4741–6.
Flaxel CJ, Edwards AR, Aiello LP, Arrigg PG, Beck RW, Bressler NM, et al. Factors associated with visual acuity outcomes after vitrectomy for diabetic macular edema: diabetic Retinopathy Clinical Research Network. Retina. 2010;30(9):1488–95.
Murakami T, Nishijima K, Akagi T, Uji A, Horii T, Ueda-Arakawa N, et al. Segmentational analysis of retinal thickness after vitrectomy in diabetic macular edema. Invest Ophthalmol Vis Sci. 2012;53(10):6668–74.
Sun JK, Lin MM, Lammer J, Prager S, Sarangi R, Silva PS, et al. Disorganization of the retinal inner layers as a predictor of visual acuity in eyes with center-involved diabetic macular edema. JAMA Ophthalmol. 2014;132(11):1309–16.
Forooghian F, Stetson PF, Meyer SA, Chew EY, Wong WT, Cukras C, et al. Relationship between photoreceptor outer segment length and visual acuity in diabetic macular edema. Retina. 2010;30(1):63–70.
Jansson RW, Raeder MB, Krohn J. Photopic full-field electroretinography and optical coherence tomography in type 1 diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol. 2015;253(7):989–97.
Araki M, Watanabe K, Yasuda K. Immunocytochemical localization of rhodopsin-like immunoreactivity in the outer segments of the rods and single cones of chick retina. Cell Struct Funct. 1984;9:1–12.
Acknowledgements
No funding or sponsorship was received for this study or publication of this article. All named authors meet the International Committee of Medical Journal Editors criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published.
Disclosures
Jiro Kogo, Akira Shiono, Hiroki Sasaki, Ryo Yomoda, Tatsuya Jujo, Yasushi Kitaoka and Hitoshi Takagi have nothing to disclose.
Compliance with Ethics Guidelines
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964, as revised in 2013. Informed consent was obtained from all patients for being included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/D8F8F06014972D99.
Rights and permissions
About this article
Cite this article
Kogo, J., Shiono, A., Sasaki, H. et al. Foveal Microstructure Analysis in Eyes with Diabetic Macular Edema Treated with Vitrectomy. Adv Ther 34, 2139–2149 (2017). https://doi.org/10.1007/s12325-017-0598-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-017-0598-4