Abstract
Corneal collagen cross-linking (CXL) was first described over a decade ago and is now considered to be one of the most important surgical innovations of modern ophthalmology. Prior to its introduction, no interventions were available to arrest, or slow down ectatic disease progression, with corneal transplantation required in the majority of cases. Unlike earlier treatments of corneal ectasias that attempted to only improve the consequences of the disease, CXL aims to address the corneal biomechanical weakening itself. The long-term safety and efficacy of CXL have been established in several studies that have documented significant improvements in all outcome measures (visual acuity, spherical equivalent, astigmatism, and keratometric findings). The emerging combination of CXL with other interventions (termed ‘CXL plus’) optimizes the visual and topographic outcomes. This, along with the expansion of the techniques’ indications for other clinical conditions, such as microbial keratitis, highlights the continuous improvement of the initial technique and confirms its wide acceptance. Overall, CXL has already demonstrated much promise and has several clinical indications, representing a clear example of recent advances in ocular therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Corneal collagen cross-linking (CXL) constitutes a minimally invasive surgical intervention employed for the management of ectatic corneal disorders, such as keratoconus, pellucid marginal corneal degeneration and post-laser in situ keratomileusis (LASIK) corneal ectasia [1–4]. It has been previously demonstrated that in keratoconus, the number of diagonal links of collagen fibrils is significantly reduced [5]. These fibrils provide the cornea with mechanical stability. When they are lacking, the cornea gradually becomes destabilized due in part to thinning of the central and para-central areas, which in turn causes irregular astigmatism, myopia and reduction in visual acuity.
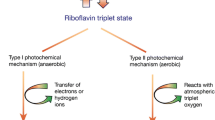
The CXL principle is based on the formation of chemical bonds (cross-links) among stromal collagen fibrils, thereby strengthening and stabilizing the diseased cornea. The use of riboflavin, also known as vitamin B2, in conjunction with ultraviolet-A (UV-A) irradiation facilitates the formation of cross-links between collagen fibrils in the corneal stroma, providing a stiffening effect capable of halting progression of the ectasia [1, 2].
Prior to the introduction of CXL, the possible treatment options for ectatic corneal disorders included spectacle correction, contact lenses, intrastromal corneal ring segment implantation [6] and, in advanced cases, lamellar or penetrating keratoplasty [7]. All these options had a single goal of symptomatic treatment and did not aim to stabilize the ectatic disorder per se. In contrast, CXL arrests the progression of the primary disorder, thereby addressing the pathophysiology of the disease rather than just its symptoms.
Original Surgical Technique (Dresden Protocol)
The term ‘cross-linking’ in the biological sciences is used to express the formation of chemical bridges following chemical reactions between proteins or other molecules. Usually, cross-links can be formed by chemical reactions that are initiated by heat, pressure, or radiation. The result of such reactions is the change in the biological molecules’ physical properties. Natural enzymatic cross-linking is part of the post-translational modification of collagen. During the aging process of the human body, both enzymatic and non-enzymatic cross-linking occur in various parts, such as the skin or the arteries. A key observation that resulted in the introduction of CXL for the management of keratoconus is the fact that diabetics often do not show progression of corneal ectatic disorders due to naturally occurring non-enzymatic cross-linking [1].
The standard CXL protocol was first described by Wollensak and colleagues [1] and is often referred to as the ‘Dresden protocol’. This treatment protocol constitutes the benchmark of the CXL procedure and has set the foundation for the evaluation of safety and efficacy of the technique. CXL is always conducted under sterile conditions in the operating room. After application of topical anesthesia, the central 8–9 mm of the epithelium is removed. It is now possible to perform mechanical removal of the epithelium with a blade (or more recently employing a rotating brush), removal with the use of alcohol (laser-assisted sub-epithelial keratectomy, LASEK), or removal with a laser (transepithelial CXL).
Riboflavin 0.1% solution is applied every 2–5 min for approximately 30 min to facilitate penetration of the corneal stroma, until the stroma is completely penetrated, as indicated by yellow flare in the anterior chamber. Different commercially available UV-A light sources can be used. The role of riboflavin in CXL is twofold. Not only does it work as a photo sensitizer for the induction of cross-links, but by acting as a selective filter, it also protects the underlying tissues from the harmful influence of UV-A. It has been shown by Wollensak and coworkers [8] that the cytotoxic irradiance level stands at 0.5 mW/cm2 for keratocytes after UV-A irradiation combined with the photosensitizer riboflavin, which is 10-times lower than the cytotoxic irradiance of 5 mW/cm2 after UV-A-irradiation alone.
Before treatment, the intended 3 mW/cm2 surface irradiance (5.4 J/cm2 surface dose) can be confirmed using a UV light meter. In a previous investigation, Wollensak [2] proposed a pre-operative corneal thickness of 400 μm as a minimum safety limit to avoid posterior corneal tissue damage during CXL. In rabbits, corneal endothelial toxicity was reached by irradiance of 0.36 mW/cm2, while this level of radiation exposure in human corneas reached a depth of less than 400 μm. Spoerl and coworkers [9] also reported that a safety threshold of 400 μm corneal pachymetry in the presence of riboflavin was necessary to limit UV-A irradiance to less than 1 J/cm2 at the level of the corneal endothelium, anterior chamber, lens and retina. Unquestionably, the presence of riboflavin enhances the safety profile. The cornea is exposed to the above stated level of UV-A energy for a total of 30 min. During treatment, riboflavin solution is applied every 2–5 min to ensure saturation of the tissue. After treatment, a bandage contact lens is applied until the epithelium is completely healed and is combined with the application of topical corticosteroids, antibiotic, and non-steroidal anti-inflammatory agents.
Efficacy, Safety and Clinical Outcomes
Several clinical studies of CXL have now been conducted in Europe and the USA, all of which provide information on the efficacy and safety of the procedure in the short, medium and long term. Even though CXL has become common practice in Europe, in the USA, the US Food and Drug Administration (FDA) has not yet approved this treatment modality. Nevertheless, there are two ongoing clinical trials, the results of which may lead to FDA approval. This will positively impact availability and the cost of treatment options, and also liability issues in the USA.
The first study in human eyes was conducted in 2003 by Wollensak and coworkers and included 23 cases [1]. This study included follow-up data for up to 4 years and demonstrated topographic stability and improvement of the mean keratometric (K) values in approximately 70% of treated patients. Furthermore, 65% of treated patients also showed a small improvement in visual acuity [1].
Caporossi and coworkers [10] presented preliminary results on CXL, including ten cases with 6 months follow-up. Refractive results demonstrated a reduction of about 2.5 diopters (D) in the mean spherical equivalent, topographically confirmed by the reduction in mean K values. A second study by the same research group detected stability of the corneal ectatic disorder in 44 cases after a minimum of 48 months of follow-up [11]. Corneal symmetry improvement was seen in 85% of patients.
In a subsequent comparative study, Coscunseven and coworkers [12] confirmed the initial findings reported by Wollensak and coworkers [1]. Following CXL, this group detected a mean decrease in spherical equivalent refraction of 1.03 ± 2.22 D and an increase in uncorrected distance visual acuity (UDVA) and corrected distance visual acuity (CDVA) of 0.06 ± 0.05 and 0.10 ± 0.14 D, respectively, for the group treated. In contrast, they documented progression for all tested parameters in the eyes that were not treated.
Agrawal [13] presented his results in a series of Indian eyes showing that 1 year after CXL treatment, 54% of eyes gained at least one line of CDVA. The K value of the apex decreased by a mean of 2.73 D in 66% of eyes and remained stable (within ±0.50 D) in 22% of eyes. Similar 1-year follow-up results confirming the efficacy of the procedure were reported by Asri and coworkers [14] in a series of 142 eyes with a greater than 2-D difference in K readings in 21.3% of cases and stability in another 68.8%. Similar statistically significant improvements in all tested parameters after 12 months have been reported for two other studies [15, 16]. Vinciguerra and coworkers [17] detected similar gains in keratometric and refractive findings but also showed that corneal and total wavefront aberrations were reduced 1 year after CXL treatment in their series of 28 eyes. Koller and coworkers [18, 19] made an interesting observation during the 12-month follow-up period of their patients treated for keratoconus. They observed corneal flattening [18] with regularization of the corneal shape [19], as captured by means of Scheimpflug imaging. Thus, the authors concluded that the CXL ‘effect’ causes a progressive topographic improvement throughout the follow-up period.
In another study, Goldich and coworkers [20] observed a significant improvement in CDVA (0.21 ± 0.1 to 0.14 ± 0.1; p = 0.002) and stability in UDVA (0.62 ± 0.5 to 0.81 ± 0.49; p = 0.475). There was a significant decrease in the steepest-meridian keratometry (53.9 ± 5.9 to 51.5 ± 5.4 D, p = 0.001) recorded 24 months after CXL in eyes with keratoconus. Similar long-term, successful results (with 3-year follow-up) have been published by Raiskup-Wolf and coworkers [21], who conducted a study in 241 keratoconus cases. Only two patients needed a second CXL treatment because of apparent progression of the ectasia.
Kymionis and coworkers [22] established a significant increase in intraocular pressure (IOP) measurements by Goldman applanation tonometry (GAT) at 6 months (from 9.95 ± 3.01 to 11.40 ± 2.89 mmHg) and then at 12 months (from 9.95 ± 3.01 to 11.35 ± 3.38 mmHg) following CXL (both p < 0.001). The authors attributed this pressure rise to the increased corneal rigidity and stiffness, which came about due to the formation of cross-links in the corneal stroma of the treated cases. Gkika and coworkers [23] evaluated IOP with three different tonometers—GAT, Pascal dynamic contour tonometer (PDCT) and ocular response analyzer (ORA) tonometer—before and after CXL, and concluded that PDCT had greater accuracy in keratoconus patients before and after CXL. The same research group tried to assess corneal hysteresis and corneal resistance factor in keratoconic eyes before and after CXL, proving that CXL exerts a non-significant impact on ORA measurements [24].
Several studies in the literature have investigated the use of CXL in post-LASIK corneal ectasia with up to 25-month follow-up [3, 25, 26]. These studies have demonstrated no progression of ectasia in conjunction with visual and topographic improvement.
There are now a number of published investigations that have evaluated the safety of the CXL technique [20, 27, 28]. The first study [20] evaluated the corneal endothelium by specular microscopy and the retina by comprehensive fundus examination and optical coherence tomography analysis. The investigators concluded that no morphologic abnormalities were detected after CXL, and that the endothelial cell density and foveal thickness remained unchanged [20]. A subsequent study [27] reported no changes in crystalline lens density and foveal thickness 12 months after CXL, while the third study [28] also confirmed the absence of retinal morphologic changes after CXL.
A recent study investigated CXL specifically in pediatric patients and reported encouraging preliminary results [29], but these observations must be confirmed in large controlled trials, and the technique must be applied with caution in children. Chatzis and Hafezi [30] documented visual, refractive and topographic stabilization and improvements after pediatric CXL similar to those reported for adult treatment outcomes over 2 years. Nevertheless, they did observe some keratometric progression at 3 years of follow-up. The findings suggest that pediatric CXL may not provide long-term stability comparable to adult treatment and these younger patients may require re-treatment, especially in a subset of those patients with persistent eye rubbing.
Corneal Collagen Cross-Linking in Thin Corneas (Under 400 μm)
It should be noted that there are many cases with keratoconus and post-LASIK ectasia who exhibit corneal stromal thickness less than 400 μm, and who achieve a satisfactory visual acuity by means of spectacles or contact lenses. In these cases, the current CXL treatment protocol prohibits the surgical procedure due to inadequate corneal thickness. Nevertheless, two groups have now proposed an alternative treatment protocol targeting thin corneas [31, 32]. They employ hypo-osmolar riboflavin solution with overall satisfactory results. Still, it is essential to bear in mind that since these ectatic corneas with less than 400 μm corneal thickness are outside the range of the Dresden protocol, the risk of the procedure is greater and a higher rate of complications may occur. For example, a significant postoperative decrease in endothelial cell density has been documented by Kymionis and coworkers in a few of these cases [33].
Complications
To date, few complications have been reported during and after CXL. Therefore, CXL is now generally considered a safe and effective surgical procedure. In some cases, stromal edema is detected immediately after CXL surgery, but this is transient and, fortunately, without clinical significance.
A case report of herpetic keratitis with iritis after CXL [34] has led to the belief that cross-linking can induce herpetic keratitis with inflammation in rare cases, even in patients with no history of herpetic disease. Another case of diffuse lamellar keratitis developing after CXL in a patient with post-LASIK ectasia has also been reported [35]. This case was successfully managed with intensive topical corticosteroids. Labiris and coworkers [36] published a case of acute inflammatory response after CXL resulting in corneal melting and descemetocele, which led to perforation.
Finally, a few cases of infectious keratitis post CXL have been reported in the literature [37–39]. These resulted in corneal ulceration and scarring. Another single case of ectasia progression despite CXL treatment in a pregnant woman has been published [40]. In this last case, one possible hypothesis could be that the high estrogen levels associated with pregnancy may have adversely affected the rigidity of the cornea resulting in failure of the CXL procedure [41].
New CXL Indications
A new promising line of indications for CXL in other types of corneal pathology is currently under investigation.
Infectious Keratitis
The treatment of microbial keratitis with the use of CXL has recently raised interest among the scientific community. To date, CXL has been shown clinically to be beneficial in cases of resilient pathogens, such as drug-resistant Streptococcus pneumoniae and Gram-negative Escherichia coli [42–44]. Martins and coworkers [43] have proven the in vitro antimicrobial efficacy of riboflavin/UV-A (365 nm) combination for bacterial and fungal isolates. In all published studies, there was a rapid decrease in pain and the corneal re-epithelialization process was accelerated following CXL. In a pertinent published case series of five patients with infectious keratitis and corneal melting, Iseli and coworkers [45] employed CXL surgery after topical and systemic antibiotic treatments had failed. Encouragingly in all cases, corneal melting ceased and emergency corneal transplantation became unnecessary [45].
It goes without saying that CXL should not be seen as the procedure of choice for infectious keratitis and should only be applied with caution as it may have toxic effects on these susceptible diseased corneas. Moreover, not all pathogens will respond positively to CXL treatment and this especially applies in the case of herpes simplex virus because the use of UV light may act as a stimulus for virus replication, exacerbating the infection and potentially leading to corneal perforation [46, 47].
Ulcerative Keratitis
CXL seems to have an anti-edematous effect on the cornea and, therefore, it has been successfully applied in cases of bullous keratopathy [48]. Two reports by Kozobolis and coworkers [49] and Ehlers and coworkers [50] have presented the results after CXL in patients with combined ulcerative keratitis and bullous keratopathy that was unresponsive to conventional treatment regimens. In both reports, the patients’ ulcer, visual acuity and corneal edema were significantly improved.
Recently, CXL has also been investigated for modifying donor tissue prior to keratoplasty [51] and as an adjunct to orthokeratology [52]. Finally, the use of CXL for prophylaxis in patients whose corneas are deemed to be at a high risk for developing corneal ectasia after LASIK surgery for myopia [53] has been proposed.
CXL Plus
The term ‘CXL plus’ was introduced in 2011 and refers to several combined procedures aimed at enhancing the success of CXL [54]. It is well documented that when performed on its own, the CXL procedure is not intended to improve vision. However, at our disposal, there are now additional interventions to the original CXL protocol that can improve visual acuity and thus optimize the final surgical outcome. To date, controlled clinical evidence exists for the use of several complementary steps to the CXL procedure:
-
Transepithelial phototherapeutic keratectomy (t-PTK) [55 – 57]. Kymionis and coworkers [56] proved that epithelial removal using t-PTK (Cretan protocol) during CXL resulted in better visual and refractive outcomes in comparison with mechanical epithelial debridement.
-
Topography-guided and other forms of photorefractive keratectomy (PRK) [58 – 71]. The use of topography-guided PRK–CXL in post-LASIK ectasias, such as the Athens protocol described by Kanellopoulos and coworkers [68], has been successfully applied.
-
Corneal implants, also known as intracorneal ring segments [72 – 83]
-
Phakic intraocular lens implantation [84 – 89]. Labiris and coworkers [90, 91] investigated the effect of keratoconus, CXL and CXL combined with topography-guided photorefractive keratectomy (t-PRK) on self-reported quality of life (QOL) by means of the 25-item National Eye Institute Visual Function Questionnaire (NEI-VFQ 25) and concluded that keratoconus has a significant impact on patients’ QOL, even in its early stages, with functional best-spectacle-corrected visual acuity. Moreover, CXL, and especially CXL combined with t-PRK, appeared to exert a beneficial impact on self-reported QOL.
CXL Extra
In an attempt to accelerate the time required for CXL treatment utilizing the Dresden standard protocol (usually 1 h of surgical time), investigators have explored two different research avenues: riboflavin application by iontophoresis aiming at rapid stromal saturation, and the use of high fluence irradiation of UV-A light [92, 93].
The Dresden protocol relies on the application of UV-A light (365 nm) at the intensity of 3 mW/cm2 for 30 min, delivering a total of 5.4 J/cm2 energy onto the cornea [1]. In accordance with the Bunsen–Roscoe photochemical law of reciprocity, if the intensity and time change while the total energy is maintained, the effects of any photochemical reaction (in the current context, the CXL procedure) are similar. This implies that the total energy delivered and amount of cross-linkage induced in a standard CXL session should be similar to irradiation at 9 mW/cm2 for 10 min, 15 mW/cm2 for 6 min, and 30 mW/cm2 for 3 min, with all ultimately delivering the same energy (5.4 J/cm2) [93]. These new treatment protocols are referred to as accelerated CXL or ‘CXL extra’.
The main concerns for accelerated CXL are its repercussions on the safety of the procedure, given that despite a similar total energy being applied in CXL extra, the intensity of irradiation is higher and may have a harmful effect on the corneal endothelium. Initial accounts of CXL extra, however, report results comparable to those obtained with the standard Dresden protocol [94–96]. Epithelial healing occurs uneventfully and there are no detectable alterations in endothelial cell density as documented by confocal microscopy. In contrast, Cingü and coworkers [97] reported transient corneal endothelial changes following accelerated CXL (18 mW/cm2 for 5 min) for the treatment of progressive keratoconus in a case–control study. In this investigation, which employed corneal specular microscopy, a decrease in endothelial cell density was observed postoperatively at 1 month, which returned to pre-operative values after 6 months of follow-up.
Overall, accelerated CXL protocols seem to be a promising alternative in minimizing the duration of the treatment and lessening patient discomfort. Future large, controlled studies are needed to confirm the immediate and long-term safety of the procedure.
Conclusion
CXL has marked a new, less invasive era in the management of corneal ectatic disorders. Since the first pilot studies over a decade ago, many modifications and several improvements to the original protocol have been successfully carried out. These steps maximize the cross-linking effect and by doing so, halt progression and postpone or even avoid the need for corneal transplantation, as well as improving functional vision in patients with ectasias. The CXL plus and CXL extra protocols may represent the future of this procedure, but more research is needed before these steps are widely adopted. The clinical utility of CXL has already been well demonstrated even though there is, to date, no adequate knowledge regarding long-term, unforeseen consequences. Future research will further elucidate and consolidate the place of CXL among the most innovative surgical therapies in ophthalmology.
References
Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135:620–7.
Wollensak G. Crosslinking treatment of progressive keratoconus: new hope. Curr Opin Ophthalmol. 2006;17:356–60.
Hafezi F, Kanellopoulos J, Wiltfang R, Seiler T. Corneal collagen crosslinking with riboflavin and ultraviolet A to treat induced keratectasia after laser in situ keratomileusis. J Cataract Refract Surg. 2007;33:2035–40.
Spadea L. Corneal collagen cross-linking with riboflavin and UVA irradiation in pellucid marginal degeneration. J Refract Surg. 2010;26:375–7.
Sherwin T, Brookes NH. Morphological changes in keratoconus: pathology or pathogenesis. Clin Experiment Ophthalmol. 2004;32:211–7.
Siganos CS, Kymionis GD, Kartakis N, Theodorakis MA, Astyrakakis N, Pallikaris IG. Management of keratoconus with Intacs. Am J Ophthalmol. 2003;135:64–70.
Frost NA, Wu J, Lai TF, Coster DJ. A review of randomized controlled trials of penetrating keratoplasty techniques. Ophthalmology. 2006;113:942–9.
Wollensak G, Spörl E, Reber F, et al. Corneal endothelial cytotoxicity of riboflavin/UVA treatment in vitro. Ophthalmic Res. 2003;35:324–8.
Spoerl E, Mrochen M, Sliney D, Trokel S, Seiler T. Safety of UVA-riboflavin cross-linking of the cornea. Cornea. 2007;26:385–9.
Caporossi A, Baiocchi S, Mazzotta C, Traversi C, Caporossi T. Parasurgical therapy for keratoconus by riboflavin-ultraviolet type A rays induced cross-linking of corneal collagen: preliminary refractive results in an Italian study. J Cataract Refract Surg. 2006;32:837–45.
Caporossi A, Mazzotta C, Baiocchi S, Caporossi T. Long-term results of riboflavin ultraviolet a corneal collagen cross-linking for keratoconus in Italy: the Siena eye cross study. Am J Ophthalmol. 2010;149:585–93.
Coskunseven E, Jankov MR 2nd, Hafezi F. Contralateral eye study of corneal collagen cross-linking with riboflavin and UVA irradiation in patients with keratoconus. J Refract Surg. 2009;25:371–6.
Agrawal VB. Corneal collagen cross-linking with riboflavin and ultraviolet - a light for keratoconus: results in Indian eyes. Indian J Ophthalmol. 2009;57:111–4.
Asri D, Touboul D, Fournie P, et al. Corneal collagen crosslinking in progressive keratoconus: multicenter results from the French National Reference Center for Keratoconus. J Cataract Refract Surg. 2011;37:2137–43.
Arbelaez MC, Sekito MB, Vidal C, Choudhury SR. Collagen cross-linking with riboflavin and ultraviolet-A light in keratoconus: one-year results. Oman J Ophthalmol. 2009;1:33–8.
Saffarian L, Khakshoor H, Zarei-Ghanavati M, Esmaily H. Corneal cross-linking for keratoconus in Iranian patients: outcomes at 1 year following treatment. Middle East Afr J Ophthalmol. 2010;17:365–8.
Vinciguerra P, Albè E, Trazza S, et al. Refractive, topographic, tomographic, and aberrometric analysis of keratoconic eyes undergoing corneal cross-linking. Ophthalmology. 2009;116:369–78.
Koller T, Pajic B, Vinciguerra P, Seiler T. Flattening of the cornea after collagen cross-linking for keratoconus. J Cataract Refract Surg. 2011;37:1488–92.
Koller T, Iseli HP, Hafezi F, Vinciguerra P, Seiler T. Scheimpflug imaging of corneas after collagen cross-linking. Cornea. 2009;28:510–5.
Goldich Y, Marcovich AL, Barkana Y, et al. Clinical and corneal biomechanical changes after collagen cross-linking with riboflavin and UV irradiation in patients with progressive keratoconus: results after 2 years of follow-up. Cornea. 2012;31:609–14.
Raiskup-Wolf F, Hoyer A, Spoerl E, Pillunat LE. Collagen cross-linking with riboflavin and ultraviolet-A light in keratoconus: long-term results. J Cataract Refract Surg. 2008;34:796–801.
Kymionis GD, Grentzelos MA, Kounis GA, et al. Intraocular pressure measurements after corneal collagen cross-linking with riboflavin and ultraviolet A in eyes with keratoconus. J Cataract Refract Surg. 2010;36:1724–7.
Gkika MG, Labiris G, Kozobolis VP. Tonometry in keratoconic eyes before and after riboflavin/UVA corneal collagen crosslinking using three different tonometers. Eur J Ophthalmol. 2012;22:142–52.
Gkika M, Labiris G, Giarmoukakis A, Koutsogianni A, Kozobolis V. Evaluation of corneal hysteresis and corneal resistance factor after corneal cross-linking for keratoconus. Graefes Arch Clin Exp Ophthalmol. 2012;250:565–73.
Li G, Fan ZJ, Peng X. Corneal collagen cross-linking for corneal ectasia of post-LASIK: one-year results. Int J Ophthalmol. 2012;5:190–5.
Salgado JP, Khoramnia R, Lohmann CP, Winkler von Mohrenfels C. Corneal collagen cross-linking in post-LASIK keratectasia. Br J Ophthalmol. 2011;95:493–7.
Grewal DS, Brar GS, Jain R, Sood V, Singla M, Grewal SP. Corneal collagen cross-linking using riboflavin and ultraviolet-A light for keratoconus: one year analysis using Scheimpflug imaging. J Cataract Refract Surg. 2009;35:425–32.
Romano MR, Quaranta G, Bregu M, Albe E, Vinciguerra PS. No retinal morphology changes after use of riboflavin and long-wavelength ultraviolet light for treatment of keratoconus. Act Ophthalmol Scand. 2012;90:e79–80.
Kankariya VP, Kymionis GD, Diakonis VF, Yoo SH. Management of pediatric keratoconus - evolving role of corneal collagen cross-linking: an update. Indian J Ophthalmol. 2013;61:435–40.
Chatzis N, Hafezi F. Progression of keratoconus and efficacy of pediatric [corrected] corneal collagen cross-linking in children and adolescents. J Refract Surg. 2012;28:753–8.
Hafezi F, Mrochen M, Iseli HP, Seiler T. Collagen crosslinking with ultraviolet-A and hypoosmolar riboflavin solution in thin corneas. J Cataract Refract Surg. 2009;35:621–4.
Raiskup F, Spoerl E. Corneal cross-linking with hypo-osmolar riboflavin solution in thin keratoconic corneas. Am J Ophthalmol. 2011;152:28–32.
Kymionis GD, Portaliou DM, Diakonis VF, et al. Corneal collagen cross-linking with riboflavin and ultraviolet-A irradiation in patients with thin corneas. Am J Ophthalmol. 2012;153:24–8.
Kymionis GD, Portaliou DM, Bouzoukis DI, et al. Herpetic keratitis with iritis after corneal crosslinking with riboflavin and ultraviolet A for keratoconus. J Cataract Refract Surg. 2007;33:1982–4.
Kymionis GD, Bouzoukis DI, Diakonis VF, Portaliou DM, Pallikaris AI, Yoo SH. Diffuse lamellar keratitis after corneal crosslinking in a patient with post-laser in situ keratomileusis corneal ectasia. J Cataract Refract Surg. 2007;33:2135–7.
Labiris G, Kaloghianni E, Koukoula S, Zissimopoulos A, Kozobolis VP. Corneal melting after collagen cross linking for keratoconus: a case report. J Med Case Rep. 2011;16(5):152.
Hafezi F. Significant visual increase following infectious keratitis after collagen cross-linking. J Refract Surg. 2012;28:587–8.
Pollhammer M, Cursiefen C. Bacterial keratitis early after corneal crosslinking with riboflavin and ultraviolet-A. J Cataract Refract Surg. 2009;35:588–9.
Zamora KV, Ales JJ. Polymicrobial keratitis after a collagen cross-linking procedure with postoperative use of a contact lens: a case report. Cornea. 2009;28:474–6.
Hafezi F, Iseli HP. Pregnancy-related exacerbation of iatrogenic keratectasia despite corneal collagen crosslinking. J Cataract Refract Surg. 2008;34:1219–21.
Spoerl E, Zubaty V, Raiskup-Wolf F, Pillunat LE. Oestrogen-induced changes in biomechanics in the cornea as a possible reason for keratectasia. Br J Ophthalmol. 2007;91:1547–50.
Moren H, Malmsjo M, Mortensen J, Ohrstrom A. Riboflavin and ultraviolet a collagen crosslinking of the cornea for the treatment of keratitis. Cornea. 2010;29:102–4.
Martins SA, Combs JC, Noguera G, et al. Antimicrobial efficacy of riboflavin/UVA combination (365 nm) in vitro for bacterial and fungal isolates: a potential new treatment for infectious keratitis. Invest Ophthalmol Vis Sci. 2008;49:3402–8.
Micelli Ferrari T, Leozappa M, Lorusso M, Epifani E. Micelli Ferrari L. Escherichia coli keratitis treated with ultraviolet A/riboflavin corneal cross-linking: a case report. Eur J Ophthalmol. 2009;19:295–7.
Iseli HP, Thiel AM, Hafezi F, et al. Ultraviolet A/riboflavin corneal cross-linking for infectious keratitis associated with corneal melts. Cornea. 2008;27:590–4.
Kymionis GD, Portaliou DM. Corneal collagen crosslinking and herpetic keratitis. J Cataract Refract Surg. 2013;39:1281.
Ferrari G, Iuliano L, Viganò M, Rama P. Impending corneal perforation after collagen cross-linking for herpetic keratitis. J Cataract Refract Surg. 2013;39:638–41.
Kozobolis V, Labiris G, Gkika M, Sideroudi H. Additional applications of corneal cross linking. Open Ophthalmol J. 2011;5:17–8.
Kozobolis V, Labiris G, Gkika M, et al. UVA collagen cross linking treatment of bullous keratopathy combined with corneal ulcer. Cornea. 2010;29:235–8.
Ehlers N, Hjortdal J, Nielsen K, Søndergaard A. Riboflavin-UVA treatment in the management of edema and nonhealing ulcers of the cornea. J Refract Surg. 2009;25:803–6.
Wang F. UVA/riboflavin-induced apoptosis in mouse cornea. Ophthalmologica. 2008;222:369–72.
Nguyen MK, Chuck RS. Corneal collagen cross-linking in the stabilization of PRK, LASIK, thermal keratoplasty, and orthokeratology. Curr Opin Ophthalmol. 2013;24:291–5.
Kanellopoulos AJ. Long-term safety and efficacy follow-up of prophylactic higher fluence collagen cross-linking in high myopic laser-assisted in situ keratomileusis. Clin Ophthalmol. 2012;6:1125–30.
Kymionis GD. Corneal collagen cross linking - PLUS. Open Ophthalmol J. 2011;5:10.
Haque S, Simpson T, Jones L. Corneal and epithelial thickness in keratoconus: a comparison of ultrasonic pachymetry, Orbscan II, and optical coherence tomography. J Refract Surg. 2006;22:486–93.
Kymionis GD, Grentzelos MA, Karavitaki AE, et al. Transepithelial phototherapeutic keratectomy using a 213-nm solid state laser system followed by corneal collagen cross-linking with riboflavin and UVA irradiation. J Ophthalmol. 2010;2010:146543.
Yeung SN, Low SA, Ku JY, et al. Transepithelial phototherapeutic keratectomy combined with implantation of a single inferior intrastromal corneal ring segment and collagen crosslinking in keratoconus. J Cataract Refract Surg. 2013;39:1152–6.
Kanellopoulos AJ, Binder PS. Collagen cross-linking (CCL) with sequential topography-guided PRK: a temporizing alternative for keratoconus to penetrating keratoplasty. Cornea. 2007;26:891–5.
Kymionis GD, Karavitaki AE, Kounis GA, Portaliou DM, Yoo SH, Pallikaris IG. Management of pellucid marginal corneal degeneration with simultaneous customized photorefractive keratectomy and collagen crosslinking. J Cataract Refract Surg. 2009;35:1298–301.
Kymionis GD, Kontadakis GA, Kounis GA, et al. Simultaneous topography-guided PRK followed by corneal collagen cross-linking for keratoconus. J Refract Surg. 2009;25:S807–11.
Kanellopoulos AJ. Comparison of sequential vs same-day simultaneous collagen cross-linking and topography-guided PRK for treatment of keratoconus. J Refract Surg. 2009;25:S812–8.
Kymionis GD, Portaliou DM, Kounis GA, Limnopoulou AN, Kontadakis GA, Grentzelos MA. Simultaneous topography-guided photorefractive keratectomy followed by corneal collagen cross-linking for keratoconus. Am J Ophthalmol. 2011;152:748–55.
Kymionis GD, Portaliou DM, Diakonis VF, et al. Posterior linear stromal haze formation after simultaneous photorefractive keratectomy followed by corneal collagen cross-linking. Invest Ophthalmol Vis Sci. 2010;51:5030–3.
Stojanovic A, Zhang J, Chen X, Nitter TA, Chen S, Wang Q. Topography-guided transepithelial surface ablation followed by corneal collagen cross-linking performed in a single combined procedure for the treatment of keratoconus and pellucid marginal degeneration. J Refract Surg. 2010;26:145–52.
Alessio G, L’abbate M, Sborgia C, La Tegola MG. Photorefractive keratectomy followed by cross-linking versus cross-linking alone for management of progressive keratoconus: two-year follow-up. Am J Ophthalmol. 2013;155:54–65.
Tuwairqi WS, Sinjab MM. Safety and efficacy of simultaneous corneal collagen cross-linking with topography-guided PRK in managing low-grade keratoconus: 1-year follow-up. J Refract Surg. 2012;28:341–5.
Kymionis GD, Portaliou DM, Diakonis VF, et al. Management of post laser in situ keratomileusis ectasia with simultaneous topography guided photorefractive keratectomy and collagen cross-linking. Open Ophthalmol J. 2011;11(5):11–3.
Kanellopoulos AJ, Binder PS. Management of corneal ectasia after LASIK with combined, same-day, topography-guided partial transepithelial PRK and collagen cross-linking: the Athens protocol. J Refract Surg. 2011;27:323–31.
Kymionis GD, Grentzelos MA, Portaliou DM, et al. Photorefractive keratectomy followed by same-day corneal collagen crosslinking after intrastromal corneal ring segment implantation for pellucid marginal degeneration. J Cataract Refract Surg. 2010;36:1783–5.
Iovieno A, Légaré ME, Rootman DB, Yeung SN, Kim P, Rootman DS. Intracorneal ring segments implantation followed by same-day photorefractive keratectomy and corneal collagen cross-linking in keratoconus. J Refract Surg. 2011;27:915–8.
Kanellopoulos AJ, Skouteris VS. Secondary ectasia due to forceps injury at childbirth: management with combined topography-guided partial PRK and collagen cross-linking (Athens Protocol) and subsequent phakic IOL implantation. J Refract Surg. 2011;27:635–6.
Burris TE, Ayer CT, Evensen DA, Davenport JM. Effects of intrastromal corneal ring size and thickness on corneal flattening in human eyes. Refract Corneal Surg. 1991;7:46–50.
Alió JL, Shabayek MH, Artola A. Intracorneal ring segments for keratoconus correction: long-term follow-up. J Cataract Refract Surg. 2006;32:978–85.
Chan CC, Sharma M, Wachler BS. Effect of inferior-segment Intacs with and without C3-R on keratoconus. J Cataract Refract Surg. 2007;33:75–80.
Coskunseven E, Jankov M, Hafezi F, Atun S, Arslan E, Kymionis G. Effect of treatment sequence in combined intrastromal corneal rings and corneal collagen cross-linking for keratoconus. J Cataract Refract Surg. 2009;35:2084–91.
Renesto Ada C, Melo LA Jr, Sartori Mde F, Campos M. Sequential topical riboflavin with or without ultraviolet a radiation with delayed intracorneal ring segment insertion for keratoconus. Am J Ophthalmol. 2012;153:982–93.
El Awady H, Shawky M, Ghanem AA. Evaluation of collagen crosslinking in keratoconus eyes with Kera intracorneal ring implantation. Eur J Ophthalmol. 2012;22(Suppl 7):S62–8.
El-Raggal TM. Sequential versus concurrent KERARINGS insertion and corneal collagen cross-linking for keratoconus. Br J Ophthalmol. 2011;95:37–41.
Saelens IE, Bartels MC, Bleyen I, Van Rij G. Refractive, topographic, and visual outcomes of same-day corneal cross-linking with Ferrara intracorneal ring segments in patients with progressive keratoconus. Cornea. 2011;30:1406–8.
Kılıç A, Kamburoglu G, Akıncı A. Riboflavin injection into the corneal channel for combined collagen crosslinking and intrastromal corneal ring segment implantation. J Cataract Refract Surg. 2012;38:878–83.
El-Raggal TM. Effect of corneal collagen crosslinking on femtosecond laser channel creation for intrastromal corneal ring segment implantation in keratoconus. J Cataract Refract Surg. 2011;37:701–5.
Kamburoglu G, Ertan A. Intacs implantation with sequential collagen cross-linking treatment in postoperative LASIK ectasia. J Refract Surg. 2008;24:S726–9.
Huang D, Schallhorn SC, Sugar A, et al. Phakic intraocular lens implantation for the correction of myopia: a report by the American Academy of Ophthalmology. Ophthalmology. 2009;116:2244–58.
Kymionis GD, Grentzelos MA, Karavitaki AE, Zotta P, Yoo SH, Pallikaris IG. Combined corneal collagen cross-linking and posterior chamber toric implantable collamer lens implantation for keratoconus. Ophthalmic Surg Lasers Imaging. 2011;17:42.
Kurian M, Nagappa S, Bhagali R, Shetty R, Shetty BK. Visual quality after posterior chamber phakic intraocular lens implantation in keratoconus. J Cataract Refract Surg. 2012;38:1050–7.
Fadlallah A, Dirani A, El Rami H, Cherfane G, Jarade E. Safety and visual outcome of Visian toric ICL implantation after corneal collagen cross-linking in keratoconus. J Refract Surg. 2013;29:84–9.
Izquierdo L Jr, Henriquez MA, McCarthy M. Artiflex phakic intraocular lens implantation after corneal collagen cross-linking in keratoconic eyes. J Refract Surg. 2011;27:482–7.
Güell JL, Morral M, Malecaze F, Gris O, Elies D, Manero F. Collagen crosslinking and toric iris-claw phakic intraocular lens for myopic astigmatism in progressive mild to moderate keratoconus. J Cataract Refract Surg. 2012;38:475–84.
Coşkunseven E, Sharma DP, Jankov MR 2nd, Kymionis GD, Richoz O, Hafezi F. Collagen copolymer toric phakic intraocular lens for residual myopic astigmatism after intrastromal corneal ring segment implantation and corneal collagen crosslinking in a 3-stage procedure for keratoconus. J Cataract Refract Surg. 2013;39:722–9.
Labiris G, Giarmoukakis A, Sideroudi H, Gkika M, Fanariotis M, Kozobolis V. Impact of keratoconus, cross-linking and cross-linking combined with photorefractive keratectomy on self-reported quality of life. Cornea. 2012;31:734–9.
Labiris G, Giarmoukakis A, Sideroudi H, Kozobolis V. Impact of keratoconus, cross-linking and cross-linking combined with topography-guided photorefractive keratectomy on self-reported quality of life: a 3-year update. Cornea. 2013;32:e186–8.
Vinciguerra P, Rechichi M, Rosetta P, et al. High fluence iontophoretic corneal collagen cross-linking: in vivo OCT imaging of riboflavin penetration. J Refract Surg. 2013;29:376–7.
Wernli J, Schumacher S, Spoerl E, Mrochen M. The efficacy of corneal cross-linking shows a sudden decrease with very high intensity UV light and short treatment time. Invest Ophthalmol Vis Sci. 2013;54:1176–80.
Touboul D, Efron N, Smadja D, et al. Corneal confocal microscopy following conventional, transepithelial, and accelerated corneal collagen cross-linking procedures for keratoconus. J Refract Surg. 2012;28:769–76.
Mrochen M. Current status of accelerated corneal cross-linking. Indian J Ophthalmol. 2013;61:428–9.
Kanellopoulos AJ. Long term results of a prospective randomized bilateral eye comparison trial of higher fluence, shorter duration ultraviolet A radiation, and riboflavin collagen cross linking for progressive keratoconus. Clin Ophthalmol. 2012;6:97–101.
Cingü AK, Sogutlu-Sari E, Cınar Y, et al. Transient corneal endothelial changes following accelerated collagen cross-linking for the treatment of progressive keratoconus. Cutan Ocul Toxicol. 2013 Jul 17. [Epub ahead of print].
Acknowledgments
No funding or sponsorship was received for this study or publication of this article. Dr. Anastasios Konstas is the guarantor for this article, and takes responsibility for the integrity of the work as a whole.
Conflict of interest
George Kymionis, Dimitrios Mikropoulos, Dimitra Portaliou, Irini Voudouragkaki, Vassilios Kozobolis, and Anastasios Konstas declare that they have no conflict of interest in any of the materials or methods described herein.
Compliance with ethics guidelines
The analysis in this article is based on previously conducted studies, and does not involve any new studies of human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kymionis, G.D., Mikropoulos, D.G., Portaliou, D.M. et al. An Overview of Corneal Collagen Cross-Linking (CXL). Adv Ther 30, 858–869 (2013). https://doi.org/10.1007/s12325-013-0065-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-013-0065-9