Abstract
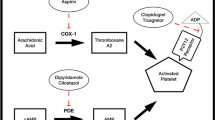
The risk of secondary events following noncardioembolic ischemic stroke or transient ischemic attack (TIA) is high and especially pronounced in the first days and weeks following the initial event; to reduce this risk, it is recommended that antiplatelet therapy be initiated immediately. Although the risk and impact of antiplatelet-associated side effects are generally far less substantial than those of secondary events, some (especially bleeding) can be severe and even life-threatening, and others may reduce adherence to antiplatelet regimens. Therefore, clinicians should implement strategies to reduce the risk of side effects and to manage those that occur. Three antiplatelet regimens have demonstrated substantial reductions in secondary event risk and are currently recommended by consensus panels: aspirin monotherapy at 50–325 mg/day; the combination of aspirin plus extended-release dipyridamole (ER-DP); and clopidogrel monotherapy. Bleeding is potentially the most significant antiplatelet-associated side effect. As bleeding risk with aspirin monotherapy is dose dependent, while preventive efficacy appears similar at all doses above 50 mg/day, aspirin doses should be kept as low as possible. Clopidogrel bleeding risk is similar to aspirin, although a reduced incidence of gastrointestinal bleeding events suggests lower gastrotoxicity. Clopidogrel should not be combined with aspirin after stroke or TIA, as the combination increases bleeding risk without improving antiplatelet efficacy. Patients should be assessed for bleeding risk (especially gastrointestinal bleeding) before initiating antiplatelet therapy; those at elevated risk should be made aware of the signs and symptoms of bleeding events to facilitate prompt treatment. The addition of ER-DP to aspirin does not increase bleeding risk, although ER-DP is associated with risk of headache, which may be severe. The prevalence of headache drops rapidly following initiation of ER-DP, suggesting most patients are able to “push through” this side effect; for those who find headache intolerable, short-term use of a reduced-dose regimen may be helpful.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lloyd-Jones D, Adams RJ, Brown TM, et al. Heart disease and stroke statistics — 2010 update: a report from the American Heart Association. Circulation. 2010;121:e46–e215.
Furie KL, Kasner SE, Adams RJ, et al. Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack. A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:227–276.
Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke. 2009;40:2276–2293.
Rothwell PM, Warlow CP. Timing of TIAs preceding stroke: time window for prevention is very short. Neurology. 2005;64:817–820.
Johnston SC, Nguyen-Huynh MN, Schwarz ME, et al. National Stroke Association guidelines for the management of transient ischemic attacks. Ann Neurol. 2006;60:301–313.
Coull AJ, Rothwell PM. Underestimation of the early risk of recurrent stroke: evidence of the need for a standard definition. Stroke. 2004;35:1925–1929.
Johnston SC, Gress DR, Browner WS, Sidney S. Short-term prognosis after emergency department diagnosis of TIA. JAMA. 2000;284:2901–2906.
Lovett JK, Dennis MS, Sandercock PA, Bamford J, Warlow CP, Rothwell PM. Very early risk of stroke after a first transient ischemic attack. Stroke. 2003;34:e138–140.
Fonarow GC, Smith EE, Reeves MJ, et al. Hospital-level variation in mortality and rehospitalization for Medicare beneficiaries with acute ischemic stroke. Stroke. 2011;42:159–166.
Albers GW, Amarenco P, Easton JD, Sacco RL, Teal P. Antithrombotic and thrombolytic therapy for ischemic stroke: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(Suppl. 6):630S–669S.
Diener HC, Cunha L, Forbes C, Sivenius J, Smets P, Lowenthal A. European Stroke Prevention Study 2. Dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J Neurol Sci. 1996;143:1–13.
[No authors listed.] A comparison of two doses of aspirin (30 mg vs. 283 mg a day) in patients after a transient ischemic attack or minor ischemic stroke. The Dutch TIA Trial Study Group. N Engl J Med. 1991;325:1261–1266.
[No authors listed.] Swedish Aspirin Low-Dose Trial (SALT) of 75 mg aspirin as secondary prophylaxis after cerebrovascular ischaemic events. The SALT Collaborative Group. Lancet. 1991;338:1345–1349.
Farrell B, Godwin J, Richards S, Warlow C. The United Kingdom transient ischaemic attack (UK-TIA) aspirin trial: final results. J Neurol Neurosurg Psychiatry. 1991;54:1044–1054.
[No authors listed.] A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee. Lancet. 1996;348:1329–1339.
Diener HC, Bogousslavsky J, Brass LM, et al. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial. Lancet. 2004;364:331–337.
Bhatt DL, Fox KA, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med. 2006;354:1706–1717.
The ESPIRIT study group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPIRIT): a randomised controlled trial. Lancet. 2006;367:1665–1673.
Aggrenox (aspirin/extended-release dipyridamole) capsules. Prescribing information. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc.; 2009.
Plavix (clopidogrel bisulfate). Prescribing information. Bridgewater, NJ: Bristol-Myers Squibb/Sanofi Pharmaceuticals Partnership; 2010.
Derendorf H, VanderMaelen CP, Brickl RS, MacGregor TR, Eisert W. Dipyridamole bioavailability in subjects with reduced gastric acidity. J Clin Pharmacol. 2005;45:845–850.
Sacco RL, Diener HC, Yusuf S, et al. Aspirin and extended-release dipyridamole versus clopidogrel for recurrent stroke. N Engl J Med. 2008;359:1238–1251.
Burger W, Chemnitius JM, Kneissl GD, Rucker G. Low-dose aspirin for secondary cardiovascular prevention — cardiovascular risks after its perioperative withdrawal versus bleeding risks with its continuation — review and meta-analysis. J Intern Med. 2005;257:399–414.
Rahman M, Beattie J. Peri-operative medication in patients with cardiovascular disease. Pharm J. 2004;272:352–354.
Douketis JD, Berger PB, Dunn AS, et al. The perioperative management of antithrombotic therapy: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2008;133(Suppl. 6):299S–339S.
Patrano, Carlo, Baigent C, Hirsh, Roth G. Antiplatelet drugs: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2008;133(Suppl. 6):199S–233S.
Theis JG, Deichsel G, Marshall S. Rapid development of tolerance to dipyridamole-associated headaches. Br J Clin Pharmacol. 1999;48:750–755.
Chang YJ, Ryu SJ, Lee TH. Dose titration to reduce dipyridamole-related headache. Cerebrovasc Dis. 2006;22:258–262.
Bennett CJ, Connors JM, Carwile JM, et al. Thrombotic thrombocytopenic purpura associated with clopidogrel. N Engl J Med. 2000;342:1773–1777.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guthrie, R. Review and management of side effects associated with antiplatelet therapy for prevention of recurrent cerebrovascular events. Adv Therapy 28, 473–482 (2011). https://doi.org/10.1007/s12325-011-0026-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-011-0026-0