Abstract
Introduction
Propolis is the generic name for the resinous substance collected by honeybees, which is known to have antioxidant, anti-inflammatory, apoptosis-inducible effects. Anastomotic dehiscence after colorectal surgery is an important cause of morbidity and mortality. We aimed to assess the effect of propolis on healing in an experimental colon anastomosis in rats.
Methods
Forty adult male Wistar albino rats were randomly assigned into 5 treatment groups with 8 rats in each: Group I, anastomosis+no treatment; Group II, anastomosis+oral propolis (600 mg/kg/d); Group III, anastomosis+oral ethyl alcohol (1 cc/d); Group IV, anastomosis+rectal propolis (600 mg/kg/d); Group V, anastomosis+rectal ethyl alcohol (1 cc/d). The bursting pressures, hydroxiproline levels and histopathological changes in each group were measured.
Results
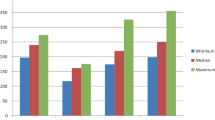
When bursting pressures were compared between groups, we observed that they were increased in the groups treated with propolis in contrast to all other groups. Hydroxiproline levels in the propolis groups were also significantly increased in contrast to the other groups. There was also a statistically significant difference in histopathological changes between the treatment types. When propolis administration methods were compared, we did not observe a statistically significant difference.
Conclusion
Propolis has a significantly favourable effect on healing in experimental colon anastomosis, independent from the method of administration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nagaoka T, Banskota AH, Tezuka Y, Saiki I, Kadota S. Selective antiproliferative activity of caffeic acid phenethyl ester analogues on highly liver-metastatic murine colon 26-L5 carcinoma cell line. Bioorg Med Chem. 2002;10:3351–3359.
Vieira O, Laranjinha J, Madeira V, Almeida L. Cholesteryl ester hydroperoxide formation in myoglobin-catalyzed low density lipoprotein oxidation: concerted antioxidant activity of caffeic and p-coumaric acids with ascorbate. Biochem Pharmacol. 1998;55:333–340.
Koshihara Y, Neichi T, Murota S, Lao A, Fujimoto Y, Tatsuno T. Caffeic acid is a selective inhibitor for leukotriene biosynthesis. Biochim Biophys Acta. 1984;792:92–97.
Michaluart P, Masferrer JL, Carothers AM, et al. Inhibitory effects of caffeic acid phenethyl ester on the activity and expression of cyclooxygenase-2 in human oral epithelial cells and in a rat model of inflammation. Cancer Res. 1999;59:2347–2352.
Chan WS, Wen PC, Chiang HC. Structure-activity relationship of caffeic acid analogues on xanthine oxidase inhibition. Anticancer Res. 1995;15:703–707.
Ploemen JH, van Ommen B, de Haan A, Schefferlie JG, van Bladeren PJ. In vitro and in vivo reversible and irreversible inhibition of rat glutathione S-transferase isoenzymes by caffeic acid and its 2-Sglutathionyl conjugate. Food Chem Toxicol. 1993;31:475–482.
Tanaka T, Kojima T, Kawamori T, et al. Inhibition of 4-nitroquinoline-1-oxideinduced rat tongue carcinogenesis by the naturally occurring plant phenolics caffeic, ellagic, chlorogenic and ferulic acids. Carcinogenesis. 1993;14:1321–1325.
Frenkel K, Wei H, Bhimani R, et al. Inhibition of tumor promoter-mediated processes in mouse skin and bovine lens by caffeic acid phenethyl ester. Cancer Res. 1993;53:1255–1261.
Fernandez MA, Saenz MT, Garcia MD. Anti-inflammatory activity in rats and mice of phenolic acids isolated from Scrophularia frutescens. J Pharm Pharmacol. 1998;50:1183–1186.
Liao HF, Chen YY, Liu JJ, et al. Inhibitory effect of caffeic acid phenethyl ester on angiogenesis, tumor invasion, and metastasis. J Agric Food Chem. 2003;51:7907–7912.
Burdock GA. Review of the biological properties and toxicity of bee propolis (propolis). Food Chem Toxicol. 1998;36:347–363.
Waninger J, Kauffmann GW, Shah IA, Farthmann EH. Influence of the distance between interrupted sutures and the tension of sutures on the healing of experimental colonic anastomoses. Am J Surg. 1992;163:319–323.
Nursal TZ, Anarat R, Bircan S, Yildirim S, Tarim A, Haberal M. The effect of tissue adhesive, octyl-cyanoacrylate, on the healing of experimental high-risk and normal colonic anastomoses. Am J Surg. 2004;187:28–32.
Sakallioglu AE, Yagmurlu A, Dindar H, Hasirci N, Renda N, Deveci MS. Sustained local application of low-dose epidermal growth factor on steroid-inhibited colonic wound healing. J Pediatr Surg. 2004;39:591–595.
Hendriks JM, Hubens G, Wuyts FL, Vermeulen P, Hubens A, Eyskens E. Experimental study of intraperitoneal suramin on the healing of colonic anastomosis. Br J Surg. 1999;86:1171–1175.
Turkcapar AG, Demirer S, Sengul N, et al. The adverse effects of octreotide on the healing of colonic anastomosis. Surg Today. 1998;28:279–284.
Jin UH, Chung TW, Kang SK, et al. Caffeic acid phenyl ester in propolis is a strong inhibitor of matrix metalloproteinase-9 and invasion inhibitor: isolation and identification. Clin Chim Acta. 2005;362:57–64.
Krell R. Value-Added Products From Beekeeping. FAO Agricultural Services Bulletin No. 124. Food and Agriculture Organization of the United Nations: Rome; 1996.
Sahinler N, Kaftanoglu O. Natural product propolis: chemical composition. Nat Prod Res. 2005;19:183–188.
Bergman I, Loxly R. Two impaired and simplified methods for the spectro-photometric determination of hydroxyproline. Ann Chem. 1963;35:1961–1965.
Castaldo S, Capasso F. Propolis, an old remedy used in modern medicine. Fitoterapia. 2002;73(suppl 1):S1–S6.
Brasken P, Lehto M, Renvall S. Changes in the connective tissue composition of the submucosal layer of colonic anastomosis. Acta Chir Scand. 1989;155:413–419.
Kologlu M, Yorganci K, Renda N, Sayek I. Effect of local and remote ischemiareperfusion injury on healing of colonic anastomoses. Surgery. 2000;128:99–104.
Thornton FJ, Barbul A. Healing in the gastrointestinal tract. Surg Clin North Am. 1997;77:549–573.
Kologlu M, Sayek I, Kologlu LB, Onat D. Effect of persistently elevated intraabdominal pressure on healing of colonic anastomoses. Am J Surg. 1999;178:293–297.
Graham MF, Blowquist P, Zederfeldt B. The alimentary canal. In: Cohen IK, Deigelman RF, Lindblad WJ, eds. Wound Healing: Biochemical and Clinical Aspects. Philadelphia: WB Saunders; 1992:433–449.
Demirogullari B, Sonmez K, Turkyilmaz Z, et al. Comparison of consequent small bowel anastomoses after transient ischemia: an experimental study in rats. J Pediatr Surg. 1998;33:91–93.
Agren MS, Andersen TL, Mirastschijski U, et al. Action of matrix metalloproteinases at restricted sites in colon anastomosis repair: an immunohistochemical and biochemical study. Surgery. 2006;140:72–82.
Stumpf M, Klinge U, Wilms A, et al. Changes of the extracellular matrix as a risk factor for anastomotic leakage after large bowel surgery. Surgery. 2005;137:229–234
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Temiz, M., Aslan, A., Canbolant, E. et al. Effect of propolis on healing in experimental colon anastomosis in rats. Adv Therapy 25, 159–167 (2008). https://doi.org/10.1007/s12325-008-0013-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-008-0013-2