Abstract
Spinocerebellar ataxia type 7 is a neurodegenerative inherited disease caused by a CAG expansion in the coding region of the ATXN7 gene, which results in the synthesis of polyglutamine-containing ataxin-7. Expression of mutant ataxin-7 disturbs different cell processes, including transcriptional regulation, protein conformation and clearance, autophagy, and glutamate transport; however, mechanisms underlying neurodegeneration in SCA7 are still unknown. Implication of oxidative stress in the pathogenesis of various neurodegenerative diseases, including polyglutamine disorders, has recently emerged. We perform a cross-sectional study to determine for the first time pheripheral levels of different oxidative stress markers in 29 SCA7 patients and 28 age- and sex-matched healthy subjects. Patients with SCA7 exhibit oxidative damage to lipids (high levels of lipid hydroperoxides and malondialdehyde) and proteins (elevated levels of advanced oxidation protein products and protein carbonyls). Furthermore, SCA7 patients showed enhanced activity of various anti-oxidant enzymes (glutathione reductase, glutathione peroxidase, and paraoxonase) as well as increased total anti-oxidant capacity, which suggest that activation of the antioxidant defense system might occur to counteract oxidant damage. Strikingly, we found positive correlation between some altered oxidative stress markers and disease severity, as determined by different clinical scales, with early-onset patients showing a more severe disturbance of the redox system than adult-onset patients. In summay, our results suggest that oxidative stress might contribute to SCA7 pathogenesis. Furthermore, oxidative stress biomarkers that were found relevant to SCA7 in this study could be useful to follow disease progression and monitor therapeutic intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinocerebellar ataxia type 7 (SCA7) is an autosomal dominant inherited disorder caused by the expansion of unstable CAG repeats in exon 3 of the ATXN7 gene on chromosome 3p12–21.1 [1]. SCA7 is an autosomal dominant disorder characterized by neural loss, mainly in the cerebellum and regions of the brainstem and, particularly, the inferior olivary complex [2,3,4,5]. Frequent clinical neurological features include gait ataxia, hyperreflexia, dysarthria, dysmetria, dysphagia, spasticity, and dysdiadochokinesia [2,3,4,5]. Visual alterations include low saccadic movement, ophthalmplegia, abnormal color discrimination, and eventually total blindness. Gastrointestinal and urinary abnormalities as well as sensory-motor peripheral neuropathy are also present [5, 6]. The CAG repeat tract is polymorphic; it ranges from 4 to 18 in normal individuals and from 36 to up to 460 in patients with SCA7. Longer expansions are associated with earlier onset and a more severe clinical presentation in subsequent generations of an affected genealogy [7, 8].
The molecular basis underlying SCA7 pathogenesis still needs to be fully elucidated; however, since ataxin-7, the ATXN7 gene product, is part of the highly conserved transcriptional coactivator STAGA complex (SPT-ADA-GCN5-acetyltransferase), it has been proposed that mutant ataxin-7, containing an enlarged polyglutamine (polyQ) tract at its N-terminal domain, disturbs transcriptional regulation of genes involved in both neuronal and glial function by altering both deubiquitinase and histone acetyltransferase activities of STAGA [9,10,11]. Notwithstanding, it has been noted in literature that intranuclear inclusions of ataxin-7 are believed to cause neuronal toxicity and death [12]. In spite of recent advances in understanding SCA7 pathogenesis, the functional link between ataxin-7 aggregation and neuronal dysfunction is still unresolved. Interestingly, emerging evidence suggests that oxidative stress (oxidative damage to proteins, lipids, and DNA due to an imbalance of the oxidant/antioxidant system) might play a pivotal role in the selective neuronal death occurring in different neurodegenerative disorders, including Huntington’s disease, dentatorubral-pallidolusyan atrophy (DRPLA), SCA1, SCA2, and SCA3 [13,14,15,16,17,18]. Particularly, increased oxidative environment and cell toxicity were found to be associated with mutant ataxin-7 aggregation in PC12 cells [19], which implies a role for oxidative stress in the pathogenesis of SCA7.
In this study, we show for the first time the analysis of circulating markers of oxidative stress in patients with SCA7. We found significant alteration in the levels of oxidative stress markers in patients with SCA7, and the pro-oxidative status was found in correlation with both age at onset and severity of the disease, which suggests that oxidative stress might be a contributor to the pathophysiology of SCA7.
Materials and Methods
Study Design and Subjects
This research was an exploratory, case-control, and cross-sectional study. The sample included Mexican patients with SCA7 and age- and gender-matched healthy controls that included blood relatives and unrelated subjects, who were confirmed as non-carriers of the SCA7 mutation by molecular testing (see below). Subjects were consecutively recruited from the Community of Tlaltetela (Veracruz State, Mexico) between July 2016 and April 2017. An informed consent form was signed by all subjects prior to examination and the research protocol was approved by the National Rehabilitation Institute (INR, Mexico City) Research and Ethical Committee. .Patients with secondary ataxias due to alcoholism, neoplasias, autoimmune or inflammatory diseases, vascular pathology, malformations, neuropharmacological treatment, and other non-genetic causes were excluded. Subjects with any of the following conditions were also excluded: severe sepsis, hepatic dysfunction, pregnancy, cancer, drug abuse, current therapy with immunosuppressive or anticonvulsant medicines or antioxidants, as well as nutritional deficiency or serum creatinine elevation.
Molecular and Clinical Assessment
Clinical and genetic blinded evaluation of patients and controls was carried following Mayo Clinic procedures [20]. Genotyping for CAG repeats in ATXN7 gene was carried out by capillary electrophoresis [21, 22]. Ataxia-associated symptoms were determined using the Scale for the Assessment and Rating of Ataxia (SARA) [23], while exploration for extracerebellar features was carried out by using the Inventory of Non-ataxia Symptoms scale (INAS) [24]. Klockgether disease severity score was defined according to disbility status: (1) fully ambulatory with moderate disability; (2) dependence of ambulatory assist devices; and (3) dependence of wheelchair or bedridden [25]. Information about the age at which the first symptom appears was provided by patients and/or their relatives.
Outcome Biochemical Analyses
Fasting blood samples were collected from all subjects at the same time. Blood was collected by venipuncture and placed in heparin or EDTA tubes (BD Vacutainer, USA). Paraoxonase (PON-I) activity and the cupric reducing antioxidant capacity (CUPRAC) determinations were performed in heparinized plasma, while EDTA plasma was used to measure the remaining oxidative stress markers. Samples were centrifuged at 2500 rpm for 10 min to obtain plasma and the white buffy layer (leukocytes) was removed. Erythrocytes were lysed in 4 volumes of ice-cold water and centrifuged at 10,000×g for 15 min at 4 °C to recover the supernatant. Erythrocyte lysates and plasma samples were stored at − 70 °C until analysis. Lipid oxidation was determined in plasma by quantification of LHP and MDA [26]. The calibration curve was obtained using 1 mM t-butylhydroperoxide as standard. The concentration of LHP was calculated using the molar absorptivity of I3 measured at 360 nm (є = 2.46 ± 0.25 × 104 M−1 cm−1), and the results expressed as nmol LHP/mg dry weight. MDA levels were evaluated using 15 mM 1-methyl-2-phenylindole (Sigma-Aldrich, MO, USA) for detection at 586 nm. The results were expressed as nmol MDA/mg dry weight [27]. The dry weight was determined according to Bernal et al. [28].
Protein damage was evaluated in plasma by quantification of three biomarkers. Dityrosines (DT) levels were determined using a fluorometric assay with 320-nm excitation and 405-nm emission and the results are expressed as nmol DT/mg protein (Malencik). Determination of advanced oxidation protein product (AOPP) levels was carried out according to Hanasand et al. [29], with the absorbance read at 340 nm. AOPP concentrations were expressed as nmol AOPP/mg protein. Protein carbonylation was determined by treatment with 2,4-dinitrophenylhydrazine (DNPH) which reacts with protein carbonylated (PC) derivatives to form stable hydrazones, which exhibit absorption peak at 370 nm [30]. A molar extinction coefficient of 21 × 103 M−1 cm−1 was used to quantify PC content. Values are expressed as nmol PC/mg protein. Protein levels were determined according to the method of Lowry [31].
Antioxidant defense was assesed throughout the activity of glutathione reductase (GR) and glutathione peroxidase (GPx) enzymes in erythrocyte lysates by measuring the rate of NADPH oxidation, according to the manufacturer’s conditions (GR activity by Cayman’s, Assay Kit, Item No. 703202 and GPx activity by Cayman’s, Glutathione Peroxidase Assay Kit, Item No. 703102, USA). Both reactions were measured spectrophotometrically by a decrease in the absorbance at 340 nm using the extinction coefficient 6220 M−1 cm−1 for NADPH. Data are expressed as nmol GR/min/mg protein for GR enzyme activity and nmol GPx/min/mg protein for GPx enzyme activity. PON-I activity was evaluated in plasma by the hydrolysis of diethyl p-nitrophenyl phosphate (paraoxon), and the absorbance was measured at 405 nm. Enzyme activity was calculated according to the molar extinction coefficient of p-nitrophenol (18,053 [mol/L]−1 cm−1 and expressed as nmol p-nitrophenol/min/mg protein [32]). CUPRAC was measured in plasma by using the copper (II) (10 mM) and neocuproine reagent (7.5 mM). The reaction mixture was incubated for 20 min at 50 °C and the absorbance was determined spectrophotometrically at 450 nm. The calibration curves were obtained using 2 mM trolox (6-hydroxy-2, 5, 7, 8-tetramethylchromane-2-carboxylic acid) (Sigma-Aldrich, MO, USA) as standard. Values were expressed as mM TR-equivalent/mg protein [33]. The equivalent is defined as the nanomolar concentration of a Trolox solution having the antioxidant capacity-equivalent to a 2.M solution of the substance.
Statistical Analysis
All analyzed variables showed normal distribution via a one-sample Kolmogorov–Smirnov test. Descriptive statistics were used to show central tendencies and dispersion of the variables under study. Baseline characteristics and the pro-oxidative status between patients with SCA7 and healthy subjects were compared by using the independent Student’s t test. Differences in oxidative stress markers among all the groups were assessed using analysis of variance (ANOVA), with Bonferroni correction for multiple comparisons. The Pearson coefficient was used to test for correlation between oxidative stress markers and SARA, INAS, Klockgether, age at onset of the disease, disease duration, or the CAG repeat size. The significance level was set at p < 0.05. The STATA 10.0 (Stata Corporation, College Station, TX, USA) statistical software package was used for calculations.
Results
Characteristics of Studied Subjects
The study sample includes 29 patients with SCA7 and 28 age- and gender-matched relative healthy subjects. Clinical characteristics of the discovery cohort as well as the demographic features of healthy subjects are described in Table 1. All patients showed SCA7-associated clinical features, including gait ataxia and cerebellar syndrome, hyperreflexia, dysmetria, dysdiadochokinesia, and visual impairment. Because patients with SCA7 display diverse clinical presentation that ranges from mild to very severe symptomatology, we categorized them into two main phenotypes, based on the age at onset of the first referred symptoms: early onset (EO, below 18 years) and adult onset (AO, above 18 years), as previously reported [5]. Interestingly, the type of first referred symptoms in EO patients was visual symptoms (decreased visual acuity or macular dysfunction) within the first two decades of their life, while AO referred cerebellar symptoms (alterations in motor coordination) as the first symptom until after the third decade of life (Table 1).
Oxidative Status in SCA7 Patients
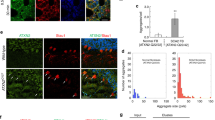
Circulating biomarkers of oxidative stress were assesed in patients with SCA7 and healthy controls, including molecules that can be modified by reactive oxygen species (ROS) in vivo (lipids and proteins) and enzymes of the antioxidant defense system of the cell (Table 2). Interestingly, SCA7 patients showed significant elevated levels of lipid hydroperoxide (LHP) and malondialdehyde (MDA), with increases of 18% (CI 95% 0.056–0.061) and 18.5% (CI 95% 0.029–0.036), respectively. Likewise, plasma levels of both advanced oxidation protein products (AOPP) and protein carbonyl derivatives increased in patients with SCA7 by 2.65% (CI 95% 249.1–419.1) and 94.91% (CI 95% 7.4–7.8), respectively, compared with healthy subjects (Table 2). In contrast, dityrosines (protein product of oxidative stress) decreased by 28.50% (CI 95% 45.57–51.55) in patients. Consistent with the idea of an altered oxidative state in SCA7, upregulated activity of different anti-oxidant enzimes was found in patients, including glutathione reductase, glutathione peroxidase, and paraoxonase (PON-1), with increases of 17.09% (CI 95% 18.73–22.80), 55% (CI 95% 0.017–0.020), and 109.6% (CI 95% 0.256–0.373), respectively. Consistently, the cupric reducing antioxidant capacity (CUPRAC) showed a 20.47% (CI 95% 2244–2758) increase in the SCA7 group, compared to healhty controls (Table 2). Next, we analyzed whether the degree of oxidative damage is associated with the phenotype of disease, based on disease onset. LHP plasma concentration was found augmented in both AO and EO patients (p < 0.0001), while MDA levels increased exclusively in EO patients (p < 0.0001) (Fig. 1a, b). With respect to protein damage, decreased dityrosine levels were observed in both AO (p < 0.05) and EO (p < 0.01) patients, while decreased AOPPs serum levels were present only in EO patients (p < 0.05) (Fig. 1c, d). Furthermore, elevated concentration of protein carbonyls was shown by both AO and EO subjects (p < 0.0001) (Fig. 1e). The antioxidant defense activity was significantly increased in both AO and EO patients (Fig. 1f–i). Particulartly, a significant increase in GR activity was detected in EO patients (p < 0.01), while higher activity of GPx (in AO: p < 0.01; and EO: p < 0.01) and PON-1 (in AO: p < 0.05; and in EO: p < 0.0001) was shown in both SCA7 patients, as well as an increased CUPRAC index that was found exclusively in EO patients (p < 0.01).
Oxidative stress markers in early onset [EO] and adult onset [AO] phenotypes. Markers of damage to lipids: a lipohydroperoxides [LPH] and b malondialdehyde [MDA]. Markers of damage to proteins: c dityrosines [DT], d advanced oxidation protein products [AOPP’s], and e protein carbonylated [PC]. The antioxidant defense enzymes evaluated were as follows: f glutathione reductase [GR], g glutathione peroxidase [GPx], h paraoxonase-1 [PON-1], and i cupric ion reducing capacity [CUPRAC]. Data is presented as scattered plots
Correlation Between Oxidative Stress Biomarkers and Disease
Finally, we evaluated the correlation between oxidative stress and disease severity, using clinical scales INAS, SARA, and Klockgether. In addition, we correlated oxidative stress markers with the length of the CAG repeat, the age at onset, and duration of the disease (Table 3). Remarkable, circulating levels of both MDA and protein carbonyls positively correlated with all clinical scales and with the CAG repeat length, while negative correlation of these two parameter was observed with age at onset of the disease. With respect to the antioxidant defense, GR was found to be significatively associated with all clinical scales, while CUPRAC exclusively correlated with the Klockgether score. Finally, AOPPs showed negative and postive correlations with the CAG repeat length and age at onset of the disease, respectively.
Discussion
In this study, we analyzed to our knowledge for the first time the peripheral oxidative state of patients with SCA7 by measuring biomarkers of the oxido-reduction system. Oxidative stress is defined as an impairment in the balance between pro-oxidants and antioxidants and can arise from increased ROS or decreased antioxidant defense. Interestingly, increased plasma levels of lipid peroxides (LHP and MDA) and protein carbonyls were found in SCA7 patients. The physiological reaction to oxidative stress comprises induction of antioxidant enzymes; consistent with this, high activity of anti-oxidant enzymes (GR, GPx, and PON-1) and increased CUPRAC were also observed in patients. These results suggest that extensive protein and lipid oxidation are taking place in SCA7, which in turn activates the antioxidant defense system, as an attempt to counteract oxidative insult.
Increased levels of oxidative stress markers and/or alterations in the anti-oxidant capacity have been extensively shown in peripheral blood, post mortem brain tissues, and in vitro models in several neurodegenerative and polyQ diseases, including SCA2, SCA3, HD, and AD [15, 34,35,36]. It is believed that the redox balance is lost by neural inflammation and/or mitochondrial dysfunction in these pathological entities [13, 37]; aggregation of misfolded protein, a hallmark of various neurodegenerative disorders, can trigger inflammation in the brain, which in turn induces the release of ROS and ultimately oxidative stress [38]. At the cellular level, lipid oxidation impair the integrity and permeability of cell membranes [39, 40], increased levels of LHP and/or MDA have been documented in PolyQ patients [15, 34,35,36] showing that lipid oxidative damage is a common pathological process in several neurodegenerative diseases including SCA7, while carbonyl derivatives induced protein misfolding, originating covalent cross-linking protein aggregates [41]; such alterations disturb both cytoskeleton organization and cell signaling and ultimately lead to cell dysfunction [42, 43]. Interestingly, we detected a marked increase of protein carbonylation in SCA7 patients, in agreement with increased levels of MDA and LHP.
Although the origin of oxidative stress in SCA7 as well as its impact on disease physiology still need to be fully understood, some experimental evidence suggests that nuclear inclusion of mutant ataxin-7 is part of the molecular basis underlying this alteration. It is noteworthy that expression and aggregation of mutant ataxin-7 in a PC12 cell-based model resulted in increased ROS levels and decreased expression of catalase, a key detoxifying enzime [19]. Furthermore, a direct link between mutant ataxin-7 aggregation and oxidative stress was revealed in this study, because treatment with an anti-oxidant agent decreased mutant ataxin-7 toxicity and aggregation while in opposite, oxidative stress induction enhanced mutant ataxin-7 aggregation [19].
Since oxidative stress markers were found to be significantly elevated in SCA7 patients, we evaluated whether the degree of oxidative stress might reflect disease onset and/or severity. It is noteworthy that a more severe oxidative stress was observed in EO patients (most affected patients), compared with AO patients, including increased MDA levels, higher activity of the antioxidant enzymes GPx, GR, and PON-1 and greater CUPRAC index, which implies that oxidative stress might be associated with disease severity. In the same sense, EO patients presented distinctive lower levels of DT and AOPPs in comparison with AO group (Fig. 1c, d), likely meaning that an antioxidant defense mechanism has been established with at least some extent of efficacy. Consistent with this hypotesis, we found significant and positive correlation between stress oxidative markers (MDA, protein carbonyl levels, and GR activity) and disease severity, as evaluated by INAS, SARA, and Klockgether scores. Interestingly, patients bearing the largest number of CAG repeats (62, 54, and 61) with the earliest age at onset of the disease (7, 12, and 7 years, respectively) showed higher SARA (35, 32, and 29, respectively) and INAS (9, 6, and 8, respectively) values as well as greater MDA and carbonyl levels, which clearly suggests that disease severity is related at least to these two stress oxidative markers. SCA7 prognossis and disease progression are currently assessed by clinical scales; nevertheless, application of these clinical scales is subjective and time-consuming and requires extensive examiner training to avoid bias. Thus, identification of non-invasive, specific, and sensitive systemic biomarkers, such as the circulating oxidative stress markers described in this study, would help to follow the natural history of the disease and monitor therapeutic effects. Recently, altered oxidative stress markers that discriminate between patients and presymptomatic carriers were identified in SCA3 [44]. In analogy with this report, further longitudinal studies and analysis in presymptomatic carriers are required to better analyze the oxidant/antioxidant system during SCA7 evolution.
Conclusion
Patients with SCA7 exhibit elevated levels of oxidative stress markers in circulation and the redox system deregulation correlates with disease severity. Collectively, our results indicate a link berween oxidative stress and the pathophysiology of SCA7; however, further studies in different cohort of patients with SCA7 are required to confirm these findings. Assessment of stress oxidative biomarkers might be useful to predict the course of the disease and evaluate therapeutic interventions.
References
David G, Abbas N, Stevanin G, Durr A, Yvert G, Cancel G, et al. Cloning of the SCA7 gene reveals a highly unstable CAG repeat expansion. Nat Genet. 1997;17(1):65–70.
David G, Durr A, Stevanin G, Cancel G, Abbas N, Benomar A, et al. Molecular and clinical correlations in autosomal dominant cerebellar ataxia with progressive macular dystrophy (SCA7). Hum Mol Genet. 1998;7(2):165–70.
Enevoldson TP, Sanders MD, Harding AE. Autosomal dominant cerebellar ataxia with pigmentary macular dystrophy. A clinical and genetic study of eight families. Brain. 1994;117(Pt 3):445–60.
Garden GA, La Spada AR. Molecular pathogenesis and cellular pathology of spinocerebellar ataxia type 7 neurodegeneration. Cerebellum. 2008;7(2):138–49.
Velazquez-Perez L, Cerecedo-Zapata CM, Hernandez-Hernandez O, Martinez-Cruz E, Tapia-Guerrero YS, Gonzalez-Pina R, et al. A comprehensive clinical and genetic study of a large Mexican population with spinocerebellar ataxia type 7. Neurogenetics. 2015;16(1):11–21.
Salas-Vargas J, Mancera-Gervacio J, Velazquez-Perez L, Rodrigez-Labrada R, Martinez-Cruz E, Magana JJ, et al. Spinocerebellar ataxia type 7: a neurodegenerative disorder with peripheral neuropathy. Eur Neurol. 2015;73(3–4):173–8.
David G, Giunti P, Abbas N, Coullin P, Stevanin G, Horta W, et al. The gene for autosomal dominant cerebellar ataxia type II is located in a 5-cM region in 3p12-p13: genetic and physical mapping of the SCA7 locus. Am J Hum Genet. 1996;59(6):1328–36.
van de Warrenburg BP, Frenken CW, Ausems MG, Kleefstra T, Sinke RJ, Knoers NV, et al. Striking anticipation in spinocerebellar ataxia type 7: the infantile phenotype. J Neurol. 2001;248(10):911–4.
McMahon SJ, Pray-Grant MG, Schieltz D, Yates JR 3rd, Grant PA. Polyglutamine-expanded spinocerebellar ataxia-7 protein disrupts normal SAGA and SLIK histone acetyltransferase activity. Proc Natl Acad Sci U S A. 2005;102(24):8478–82.
Palhan VB, Chen S, Peng GH, Tjernberg A, Gamper AM, Fan Y, et al. Polyglutamine-expanded ataxin-7 inhibits STAGA histone acetyltransferase activity to produce retinal degeneration. Proc Natl Acad Sci U S A. 2005;102(24):8472–7.
Yang H, Liu S, He WT, Zhao J, Jiang LL, Hu HY. Aggregation of Polyglutamine-expanded Ataxin 7 protein specifically sequesters ubiquitin-specific protease 22 and deteriorates its deubiquitinating function in the Spt-Ada-Gcn5-acetyltransferase (SAGA) complex. J Biol Chem. 2015;290(36):21996–2004.
Janer A, Werner A, Takahashi-Fujigasaki J, Daret A, Fujigasaki H, Takada K, et al. SUMOylation attenuates the aggregation propensity and cellular toxicity of the polyglutamine expanded ataxin-7. Hum Mol Genet. 2010;19(1):181–95.
Cornelius N, Wardman JH, Hargreaves IP, Neergheen V, Bie AS, Tumer Z, et al. Evidence of oxidative stress and mitochondrial dysfunction in spinocerebellar ataxia type 2 (SCA2) patient fibroblasts: effect of coenzyme Q10 supplementation on these parameters. Mitochondrion. 2017;34:103–14.
Guevara-Garcia M, Gil-del Valle L, Velasquez-Perez L, Garcia-Rodriguez JC. Oxidative stress as a cofactor in spinocerebellar ataxia type 2. Redox Rep. 2012;17(2):84–9.
Klepac N, Relja M, Klepac R, Hecimovic S, Babic T, Trkulja V. Oxidative stress parameters in plasma of Huntington's disease patients, asymptomatic Huntington's disease gene carriers and healthy subjects : a cross-sectional study. J Neurol. 2007;254(12):1676–83.
Miyata R, Hayashi M, Tanuma N, Shioda K, Fukatsu R, Mizutani S. Oxidative stress in neurodegeneration in dentatorubral-pallidoluysian atrophy. J Neurol Sci. 2008;264(1–2):133–9.
Stack EC, Matson WR, Ferrante RJ. Evidence of oxidant damage in Huntington's disease: translational strategies using antioxidants. Ann N Y Acad Sci. 2008;1147:79–92.
Zhou L, Wang H, Wang P, Ren H, Chen D, Ying Z, et al. Ataxin-3 protects cells against H2O2-induced oxidative stress by enhancing the interaction between Bcl-X(L) and Bax. Neuroscience. 2013;243:14–21.
Ajayi A, Yu X, Lindberg S, Langel U, Strom AL. Expanded ataxin-7 cause toxicity by inducing ROS production from NADPH oxidase complexes in a stable inducible spinocerebellar ataxia type 7 (SCA7) model. BMC Neurosci. 2012;13:86.
Denny-Brown D, Dawson DM, Tyler HR. Handbook of neurological examination and case recording, vol. viii. 3rd ed. Cambridge: Harvard University Press; 1982. 87 p.
Gomez-Coello A, Valadez-Jimenez VM, Cisneros B, Carrillo-Mora P, Parra-Cardenas M, Hernandez-Hernandez O, et al. Voice Alterations in Patients With Spinocerebellar Ataxia Type 7 (SCA7): Clinical-Genetic Correlations. J Voice. 2017;31(1):123 e1–5.
Magana JJ, Gomez R, Maldonado-Rodriguez M, Velazquez-Perez L, Tapia-Guerrero YS, Cortes H, et al. Origin of the spinocerebellar ataxia type 7 gene mutation in Mexican population. Cerebellum. 2013;12(6):902–5.
Schmitz-Hubsch T, du Montcel ST, Baliko L, Berciano J, Boesch S, Depondt C, et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology. 2006;66(11):1717–20.
Schmitz-Hubsch T, Coudert M, Bauer P, Giunti P, Globas C, Baliko L, et al. Spinocerebellar ataxia types 1, 2, 3, and 6: disease severity and nonataxia symptoms. Neurology. 2008;71(13):982–9.
Klockgether T, Lüdtke R, Kramer B, Abele M, Bürk K, Schöls L, et al. The natural history of degenerative ataxia: a retrospective study in 466 patients. Brain. 1998;Pt 4:589–600.
el-Saadani M, Esterbauer H, el-Sayed M, Goher M, Nassar AY, Jurgens G. A spectrophotometric assay for lipid peroxides in serum lipoproteins using a commercially available reagent. J Lipid Res. 1989;30(4):627–30.
Gerard-Monnier D, Erdelmeier I, Regnard K, Moze-Henry N, Yadan JC, Chaudiere J. Reactions of 1-methyl-2-phenylindole with malondialdehyde and 4-hydroxyalkenals. Analytical applications to a colorimetric assay of lipid peroxidation. Chem Res Toxicol. 1998;11(10):1176–83.
Bernal A, Mendez JD, Rosado AA. Rapid colorimetric assay for dry weight. Arch Invest Med (Mex). 1981;12(1):83–8.
Hanasand M, Omdal R, Norheim KB, Goransson LG, Brede C, Jonsson G. Improved detection of advanced oxidation protein products in plasma. Clin Chim Acta. 2012;413(9–10):901–6.
Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R. Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta. 2003;329(1–2):23–38.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265–75.
Aviram M, Rosenblat M, Bisgaier CL, Newton RS, Primo-Parmo SL, La Du BN. Paraoxonase inhibits high-density lipoprotein oxidation and preserves its functions. A possible peroxidative role for paraoxonase. J Clin Invest. 1998;101(8):1581–90.
Apak R, Guclu K, Ozyurek M, Karademir SE, Altun M. Total antioxidant capacity assay of human serum using copper(II)-neocuproine as chromogenic oxidant: the CUPRAC method. Free Radic Res. 2005;39(9):949–61.
Chang KH, Chen WL, Wu YR, Lin TH, Wu YC, Chao CY, et al. Aqueous extract of Gardenia jasminoides targeting oxidative stress to reduce polyQ aggregation in cell models of spinocerebellar ataxia 3. Neuropharmacology. 2014;81:166–75.
Pacheco LS, da Silveira AF, Trott A, Houenou LJ, Algarve TD, Bello C, et al. Association between Machado-Joseph disease and oxidative stress biomarkers. Mutat Res. 2013;757(2):99–103.
Sorolla MA, Reverter-Branchat G, Tamarit J, Ferrer I, Ros J, Cabiscol E. Proteomic and oxidative stress analysis in human brain samples of Huntington disease. Free Radic Biol Med. 2008;45(5):667–78.
Borza LRA. Review on the cause-effect relationship between oxidative stress and toxic proteins in the pathogenesis of neurodegenerative diseases. Rev Med Chir Soc Med Nat Iasi. 2014;118(1):19–27.
Liu Z, Zhou T, Ziegler AC, Dimitrion P, Zuo L. Oxidative stress in neurodegenerative diseases: from molecular mechanisms to clinical applications. Oxidative Med Cell Longev. 2017;2017:2525967.
Chen CT, Green JT, Orr SK, Bazinet RP. Regulation of brain polyunsaturated fatty acid uptake and turnover. Prostaglandins Leukot Essent Fat Acids. 2008;79(3–5):85–91.
Floyd RA, Carney JM. Free radical damage to protein and DNA: mechanisms involved and relevant observations on brain undergoing oxidative stress. Ann Neurol. 1992;32(Suppl):S22–7.
Chondrogianni N, Stratford FL, Trougakos IP, Friguet B, Rivett AJ, Gonos ES. Central role of the proteasome in senescence and survival of human fibroblasts: induction of a senescence-like phenotype upon its inhibition and resistance to stress upon its activation. J Biol Chem. 2003;278(30):28026–37.
Petersen DR, Doorn JA. Reactions of 4-hydroxynonenal with proteins and cellular targets. Free Radic Biol Med. 2004;37(7):937–45.
Zarkovic N. 4-hydroxynonenal as a bioactive marker of pathophysiological processes. Mol Asp Med. 2003;24(4–5):281–91.
de Assis AM, Saute JAM, Longoni A, Haas CB, Torrez VR, Brochier AW, et al. Peripheral oxidative stress biomarkers in spinocerebellar Ataxia type 3/Machado-Joseph disease. Front Neurol. 2017;8:485.
Acknowledgments
This work was supported by SEP-CONACyT (grant number CB-2015-01-258043) to JJ-M. This study is dedicated to the patients and the members of SCA7-affected families. We also thank Yessica Tapia-Guerrero, Joan Jano-Ito, and César Cerecedo for technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
An informed consent form was signed by all subjects prior to examination and the research protocol was approved by the National Rehabilitation Institute (INR, Mexico City) Research and Ethical Committee.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Torres-Ramos, Y., Montoya-Estrada, A., Cisneros, B. et al. Oxidative Stress in Spinocerebellar Ataxia Type 7 Is Associated with Disease Severity. Cerebellum 17, 601–609 (2018). https://doi.org/10.1007/s12311-018-0947-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-018-0947-0