Abstract
Reverse total shoulder arthroplasty (RTSA) represents a good solution for the management of patients with fracture of the proximal humerus with associated severe osteoporosis and RC dysfunction. A systematic review of the literature according to the PRISMA guidelines was performed matching the following keywords: “reverse total shoulder arthroplasty”; “reverse total shoulder prostheses”; “fractures”; “fracture of the proximal humerus.” Medline, EMBASE, Google Scholar, and Ovid database have been screened. Ten studies were considered in the qualitative analysis. No randomized prospective controlled trials have been found. A total of 256 patients received a RTSA for the management of fracture of the proximal humerus. There were 28 males (10.8 %) and 228 females (89.2 %). The mean age of patients was 75.5 ± 2.2 years (range 70–78 years). The mean follow-up period was 27.8 ± 21.8 months range (6–86 months). Overall, the mean Constant score was 56.7 ± 7.6 points (range 44–67.8 points), the mean DASH score was 39.9 ± 6 points (range 31.5–46.8 points), the ASES averaged 70.3 ± 6.8 points (range 65–78 points), and the OSS averaged 28.7 points (range 15–56 points). RTSA restores function and relieves pain in patients with proximal humeral fractures. However, no randomized controlled trials are available to support RTSA versus osteosintesis, anatomical prostheses or hemiarthroprotesis. Further studies are needed to evaluate the effectiveness of RTSA in the management of fracture of the proximal humerus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several surgical options are available for the management of complex displaced fractures of the proximal humerus, such as locked or nonlocked screw plates [1], static or dynamic anterograde nailing constructs [2], anatomical prostheses [3, 4], hemiarthroplasty [5–7], and reverse total shoulder arthroplasty (RTSA) [8–10]. Locked screw plates ensure good reduction and stabilization of the fracture [11], but their use is not indicated in case of small bone fragments because of high risk of avascular necrosis [12]. Anterograde nailing constructs with auto-stable screws are useful in case of complex dislocated and displaced fractures because they guarantee a correct fixation of bone fragments, promoting the vascularization of the humeral head and decreasing the risk of necrosis [13]. Nevertheless, their use is not indicated in case of severe osteoporosis. Anatomical shoulder prosthesis or hemiarthroplasties are implanted to treat four-part displaced fractures or fracture dislocations of the proximal humerus, demonstrating good results in terms of pain relief and functional recovery [5, 14]. However, when rotator cuff (RC) is deficient, or the fixation of the tuberosities is not successfully achieved, or nonunion and malunion occur [4], functional outcomes are often disappointing [5, 15].
RTSA represents a good solution for the management of patients with fracture of the proximal humerus with associated severe osteoporosis and RC dysfunction. RTSA was originally developed to treat patients with massive and irreparable RC tears [16–18]. Biomechanically, RTSA changes the vertical balance of the shoulder, lowering the humerus and medializing the center of rotation of the shoulder. Thus, it improves deltoid lever arm to supply RC deficiency, restoring active anterior elevation as well as abduction. The good clinical and functional outcomes led to enlarge RTSA indications to other pathologies such as cuff tear arthropathy (CTA) [19–26], failed RC surgery [22, 27], and fractures of the proximal humerus [4, 11, 15, 28–33].
The use of RTSA is associated with two major problems: the high rate of scapular notching and compromised external rotation that impairs function and patients satisfaction [29, 30, 34]. Moreover, RTSA can be associated with other important complications such as cortical perforation, humeral shaft or glenoid fracture, acromion fracture, neurological injuries, infection, dislocation, loosening of the glenosphere, and mobilization of the glenoid component.
The aim of this study was to systematically review the functional outcomes and complications of the RTSA performed as a primary surgical procedure for the management of fractures of the proximal humerus.
Materials and methods
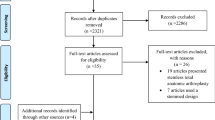
We undertook a systematic review of the literature according to the PRISMA guidelines with a PRISMA checklist and algorithm, and already validated in our setting. Two independent reviewers separately conducted the search. The search was performed on January 14, 2016. All journals were considered, and all relevant studies were analyzed. To qualify for the study, an article had to be published in a peer-reviewed journal. Articles were initially screened for relevance by title and abstract, excluding articles without an abstract and obtaining the full-text article if the abstract did not allow the investigators to assess the defined inclusion and exclusion criteria.
Two investigators separately reviewed the abstract of each publication and then performed a close reading of all papers to minimize selection bias. Moreover, a cross-reference research of the selected articles was performed to obtain other relevant articles. All articles reporting clinical and functional outcomes, as well as complications following RTSA performed as a primary surgery for the management of fractures of the proximal humerus, were taken into account. The following databases were screened: Medline, EMBASE, Google Scholar, and Ovid. Given the linguistic capabilities of the authors, articles in other languages than English, French, Spanish, German, or Italian were excluded. The following key words were matched: “reverse total shoulder arthroplasty”; “reverse total shoulder prostheses”; “fractures”; “fracture of the proximal humerus.”
We included articles that reported about patients with fracture of the proximal humerus managed with RTSA; presented a sufficient description of fracture with imaging (radiographs or CT scan); had an adequate description of the type of fracture (or used Neer, Duparc, OTA or AO-Muller classifications); clearly described the type of RTSA implanted; reported a sufficient description of the surgical intervention; completely described the clinical condition of the patients (using clinical scores, measuring ROM); had an appropriate description of the follow-up period; and presented a detailed report of the complications, outcome measures, and outcome scores. Missing data pertaining to these parameters warranted exclusion from this systematic review.
Literature reviews, case reports, case series, conference abstracts, posters, studies on animals, cadavers or in vitro, biomechanical reports, tumoral studies, technical notes, letters to editors, and instructional course were excluded. We also excluded articles about RTSA for revision of proximal humerus fracture sequelae.
Furthermore, we exclude studies with follow-up shorter than 6 months and with no information on surgical intervention, complications, diagnosis, imaging, clinical outcomes, radiographic outcomes, and statistical analysis.
The inclusion and exclusion criteria are summarized in (Table 1) (INCL-EXCL criteria). The categorical variables were reported as frequency with percentage. Continuous variable data were reported as mean ± standard deviation or range as minimum and maximum values. In all studies, P values <0.5 were considered statistically significant.
Results
The literature search and the inclusion–exclusion criteria lead to 13 articles eligible for the study. Of those articles, three were excluded because they reported outcomes of the same sample of patients at different follow-up times [29, 35]. For this reason, ten studies were considered in the qualitative analysis. Of these, 3 were cohort studies (27.2 %) [30, 31, 36] with historical control, 2 prospective cohort studies (18.2 %) [32], 3 retrospective cohort studies (36.4 %) [30, 33, 37, 38], and 2 studies (18.2 %) [29, 39] with unclear design.
Demographic results
A total of 256 patients received RTSA for the management of fracture of the proximal humerus. All prostheses were implanted as primary surgery. There were 28 males (10.8 %) and 228 females (89.2 %). The male-to-female ratio was 0.1.
The mean age of patients was 75.5 ± 2.2 years (range 70–78 years). The right shoulder was involved in 53 cases (45.7 %) and the left shoulder in 63 cases (54.3 %). In other 154 patients (59.1 %), the shoulder side was not reported. The dominant arm was injured in 48 (18.5 %) patients while in 211 patients (81.5 %), these data were not reported.
The mean follow-up period was 27.8 ± 21.8 months range (6–86 months). A total of 12 patients (3.5 %) were lost to follow-up due to death, moved to other cities, or unknown reasons.
Imaging assessment
Fractures of the proximal humerus were diagnosed in all patients using radiographs and/or CT scans. Radiographs in anteroposterior view with the arm in neutral position were performed in all patients (100 %) while anteroposterior view with the arm externally and internally rotated was performed in 103 patients (39.8 %). The radiographic scapular-lateral view was performed in 205 patients (79.2 %) while Lamy’s view was performed in 95 patients (36.7 %). CT scans were performed in 109 patients (42.1 %).
Postoperative scapular notching was graded according to Sirveaux’s classification in 114 patients (44 %), Valenti’s classification in 35 patients (13.5 %), and Nerot’s classification in 39 patients (15 %). In other 71 patients (27.5 %), scapular notching was assessed postoperatively with radiographs, but not graded with a specific classification system.
Fractures classification
Fractures of the proximal humerus were classified according to the following classification systems: Neer [40], Duparc [41], AO-Muller, and OTA [42].
A total of 115 fractures (44.4 %) were classified according to Neer’s classification system: there were 2 type II fractures (1.7 %); 15 type III fractures (13 %); 98 type IV fractures (85.3 %). Twenty fractures (7.7 %) were classified according to Duparc’s classification: there were 3 type II fractures (15 %); 13 type III fractures (65 %); 4 type III fractures (20 %). Nineteen fractures (7.4 %) were graded as “grade C” according to AO-Muller’s classification. Twenty fractures (7.7 %) were classified according to OTA classification: there were 5 B2 fractures (25 %); 7 C2 fractures (35 %); 8 C3 fractures (40 %). Eighty-five fractures (32.8 %) were classified as complex comminuted fractures of the proximal humerus. Moreover, there were 40 fracture dislocations (12.7 %).
Type of prostheses
The mean time from injury to surgery was 10 ± 4.4 days (range 3–15 days) in 182 patients (71 %), while in 74 patients (29 %), these data were not reported.
Delta III® reverse shoulder prostheses (DePuy Orthopaedics, Warsaw, IN, USA) [32, 35, 38, 39] were implanted in 141 patients (52 %), Aequalis® reverse prostheses (Tornier SAS, Montbonnot, France) [30, 37] in 50 patients (18.4 %), SMR Modular Shoulder System® (Lima-LTO, San Daniele del Friuli, Italy) [5, 36] in 28 patients (10.3 %), Arrow® reverse shoulder prosthesis (FhOrthopaedics, Mulhouse) [33] in 29 patients (11.1 %), Encore® reverse shoulder prosthesis (Encore Medical, Austin, Texas, USA) in 13 patients [31] (4.8 %), and the Zimmer® anatomical shoulder reversed prostheses (Zimmer, Warsaw, IN, USA) [38] in 9 patients (3.4 %). Table 2 summarize surgical approaches and type of prostheses implanted.
A total of 151 RTSAs (58.3 %) were cemented. Of these, 43 (28.5 %) received gentamicin-loaded cement. The remaining 108 RTSAs (41.7 %) were implanted without cement.
Immobilization and rehabilitation
The postoperative immobilization period was reported for 214 patients (82.6 %), and it averaged 20 ± 15.6 days (range 0–42 days). The same group of patients (82.6 %) started the rehabilitation protocol at an average of 10 ± 9 days (range 2–30 days) from surgery.
Outcomes assessment
Clinical outcomes were evaluated using the Constant–Murley shoulder score [43] in 219 patients (84.5 %), the Disability of Arm, Shoulder, and Hand score (DASH) [44] in 123 patients (47.5 %), the American Shoulder and Elbow Surgeons score (ASES) [45] in 60 patients (23.2 %), and the Oxford Shoulder score (OSS) [46] in 10 patients (3.9 %).
Functional outcomes were assessed measuring active ROM of the operated shoulder in 206 patients (79.5 %). The following movements were considered: anterior elevation; abduction; external rotation with the arm in adduction; external rotation with the arm at 90° of abduction; internal rotation (Table 3).
Clinical outcomes
Overall, the mean Constant score was 56.7 ± 7.6 points (range 44–67.8 points), the mean DASH score was 39.9 ± 6 points (range 31.5–46.8 points), the ASES averaged 70.3 ± 6.8 points (range 65–78 points), and the OSS averaged 28.7 points (range 15–56 points).
Functional outcomes
The mean anterior elevation was 113.4° ± 14.7° (range 95.7°–139°), the mean abduction was 97° ± 12.1 (range 86°–112.5°), the mean external rotation with the arm in adduction was 11.8° ± 10° (range 0°–27°), the mean external rotation with the arm at 90° of abduction was 35.5° ± 9.1° (range 25°–49°), and the mean internal rotation was 38° ± 13.1°.
Complications
Complications were reported in all studies. We divided complications as follows: medical complications, intraoperative complications, and postoperative complications. Moreover, we evaluated radiographic complications considering tuberosity repair and scapular notching.
Medical complications occurred in 15 patients (5.8 %). Seven patients present infection (2.6 %), 1 sustained a deep venous thrombus (0.4), 3 developed a reflex sympathetic dystrophy syndrome (1.2 %), 3 a complex pain regional syndrome (1.2 %), and 1 patient had a lymphedema (0.4 %).
Intraoperative complications occurred in 12 patients (4.6 %). 1 (0.4 %) brachial plexopathy, 1 (0.4 %) deltoid paresis, 1 (0.4 %) radial nerve injury, 1 (0.4 %) ulnar nerve injury, 3 (1.2 %) median nerve injury and 3 (1.2 %) axillary nerve injury, 1 (0.4 %) fracture of the glenoid and 1 (0.4 %) fracture of the acromion.
Postoperative complications occurred in 9 patients (3.5 %). Five patients had a dislocation. Of these, 3 were anterior dislocations (60 %). One patient (0.4 %) had humeral stem loosening, 1 patient (0.4 %) developed hematoma and another patient (0.4 %) developed the separation of the muscular flap following the superolateral approach. Nine patients (2.9 %) underwent revision surgery for the management of complications.
Scapular notching was the most common radiographic complication, occurring in 82 patients (31.7 %), followed by 55 cases (21.2 %) of tuberosity malunion/nonunion or resorption, and ectopic ossification in 28 patients (10.8 %).
According to Sirveaux classification, there were 22 grade I (26.8 %), 7 grade II (8.5 %), 5 grade III (6.1 %), and 7 grade IV cases (8.5 %) of scapular notching. According to Valenti’s classification, there were 5 grade II (6.1 %), 4 grade III (4.9 %), and 3 grade IV cases (3.7 %) of scapular notching. According to Nerot’s classification, there were 7 grade I (8.5 %), 6 grade II (7.3 %), and 3 grade III cases (3.7 %) of scapular notching. In the remaining 13 patients (15.9 %), the scapular notching was not graded, even if it was present.
Discussion
Complex comminuted displaced fractures of the proximal humerus represent one of the most difficult situations to treat in shoulder surgery, remaining a major problem in orthopedics.
In the last decade, the use of RTSA, initially developed to manage massive and irreparable RC tears in old patients with or without glenohumeral arthritis, has been extended to trauma. After our literature search, we had to exclude 3 studies [29, 35, 47] because they reported outcomes of the same sample of patients with different lengths of follow-up, and our inclusion and exclusion criteria allowed us to consider only 10 articles for this systematic review.
No randomized prospective control trials have been found in the literature on the topic. This finding represents an important limitation to define the real efficacy of RTSA in the management of fractures of the proximal humerus.
Patients with complex fractures of the proximal humerus are often elderly. The mean age of patients of the studies included in this review was 75.5 years, ranging from 70 to 78 years, with a standard deviation of 2.2 years. At this age, patients usually present a severe deficiency of the RC due to fatty degeneration or muscle atrophy [48]. Moreover, since 89.2 % of patients affected by fracture of the proximal humerus were women, many of them may present osteoporosis. For these reasons, different fixation techniques produce poor clinical and functional outcomes, with high failure rate, and the RTSA seems to be the best surgical option for these patients. However, Bufquin et al. [39] found that elderly patients had a lower Constant score compared to younger patients, but this difference was not statistically significant.
Not many studies compared functional results of reverse prosthesis and hemiarthroplasty in complex fractures of the proximal humerus. Young et al. [36] found no statistical difference in OSS and ASES between reverse prosthesis group and hemiarthroplasty group. On the other hand, Gallinet et al. [30] reported higher postoperative Constant score after RTSA than after hemiarthroplasty, and in the study of Boyle et al. [5], the RSA group had a significantly better five-year OSS than the hemiarthroplasty group (41.5 vs. 32.3; P = 0.022). We found 8.1 % of perioperative complication in patients with proximal humerus fractures managed with RTSA. However, the complication rate of RTSA is lower than the overall complication rates of hemiarthroplasty that has been reported between 11.6 % [49] and 19 % [50].
Several studies demonstrated that the tuberosity union quality is crucial, to provide good functional results in patients with fractures of the proximal humerus managed with hemiarthroplasty [4, 5, 15]. Indeed, poor outcomes are reported when tuberosity fixation is not possible during surgery, when malunion or nonunion occurs, or when the consolidation of tuberosity is not achieved in its anatomical position [38]. Tuberosity nonunion or malunion is less debilitating in patients managed with RTSA because the implant design allows active elevation thanks to the deltoid. For this reason, RTSA can be considered in the setting of acute three-part and four-part proximal humerus fractures and fracture/dislocations that demonstrate poor potential for tuberosity healing (i.e., comminuted tuberosities, osteoporotic bone) [51]. However, secure tuberosity fixation should be attempted in each case, since successful union can result in the preservation of external rotation [39]. Gallinet et al. [38] reported better results in terms of mobility (anterior elevation, abduction, external and internal rotation ROM), as well as total Constant score and DASH score when tuberosity repair was performed. Moreover, patients with anatomical consolidation of the tuberosity achieved better results compared to patients in which nonunion or malunion of tuberosities occurred. Valenti et al. [33] reported similar results, showing that patients who had their greater tuberosity and lesser tuberosity reinserted had a significantly better external rotation with the arm at the side. On the other hand, clinical results reported by Bufquin et al. [39] seemed not to be influenced by the healing of the tuberosities. Klein et al. [32] used the juxta-articular approach without acromial osteotomy, removing tuberosities without reattaching the RC. The mean Constant score was 68.75, and the external rotation ROM was 25° at a mean follow-up of 33.3 months. Cazeneuve et al. [29] used the anterolateral approach without reattaching the tuberosity. The mean Constant score was 53 at a mean follow-up of 86 months.
Scapular notching represents another important complication of RTSA, limiting functional results and its use in trauma. Scapular notching has been found in 31.7 % of cases. It is frequently observed 1 year after prosthesis implantation [52], and its incidence increases over the years [35, 47, 53]. Furthermore, the extent of scapular notching progresses with the length of follow-up and determines a low Constant score when it is associated with abnormal humeral images (41 points vs. 57 points in patients with isolated scapular notching) [35, 47, 53]. Sadoghi et al. [27] did not identified any correlation between scapular notching and clinical outcomes at 24–42 months of follow-up, while after 60 months scapular notching was positively correlated with pain section of the Constant score and active ROM. However, no other studies assessed the relationship between scapular notching and clinical or functional outcomes. Another frequent complication affecting outcomes is ectopic ossification, which occurs in 10.8 % of cases. Patients who did not develop ectopic ossifications had statistically significantly better internal rotation ROM.
Major strength of the present study is the use of the PRISMA statement with a PRISMA checklist and algorithm. Using PRISMA guidelines, as we did in our previous study [54], the risk of errors during data extraction and evaluation is substantially reduced. Another important strength is that, in order to avoid bias, all articles were screened blindly by two independent reviewers who extracted the same data from the papers. Furthermore, the restricted inclusion and exclusion criteria of the present systematic review allowed us to consider in our qualitative analysis only high-quality studies conducted in a strict scientific fashion (using objective methods of outcome assessment). In this manner, we reduced the risk of bias that is common in systematic reviews studies.
Some limitations of our study must be underlined. Firstly, the main limitation is represented by the lack of randomized prospective control trials included in our qualitative analysis. Secondly, the results that we report should be considered with caution taking into account the nature of the present study. Thirdly, we include studies with a minimum follow-up of 6 months, without stratifying results in relationship with the length of follow-up. Fourthly, we include only articles reporting about RTSA performed as primary surgery, without considering studies reporting the use of RTSA performed as a revision surgery. Furthermore, we include only 10 studies reporting about 259 patients. We know that this sample of patients is not enough to consider the outcomes found after our qualitative analysis as univocal. Finally, the lack of uniformity in reporting type of fracture, classification of scapular notching, and clinical and functional outcomes not allowed us to perform a statistical analysis considering all these parameters.
Further studies conducted in a more strict scientific fashion are necessary to understand the prognostic factors, as well as strength and limits, of the use of RTSA for the management of fractures of the proximal humerus.
Conclusions
Following encouraging results of RTSA [69], its use has been enlarged to trauma. RTSA can be considered a useful solution for the management of fractures of the proximal humerus in patients with RC dysfunction and/or severe osteoporosis. Nevertheless, there is a lack of uniformity in the studies in reporting type of fracture, classification of scapular notching, and clinical and functional outcomes. Moreover, no randomized prospective control trials are available in the literature about the topic, limiting the real understanding of the issue.
References
Neer CS II (1970) Displaced proximal humeral fractures. II. Treatment of three-part and four-part displacement. J Bone Joint Surg Am 52:1090–1103
Scarlat M, Cuny C, Irrazi M, Slimani S, Turell P (2000) Enclouage des fractures proximales de l’humérus par clou Telegraph à verrouillage autostable. Technique et premiers résultats, à propos de 66 cas (communication de la 75e réunion annuelle de la SOFCOT). Rev Chir Orthop 86(Suppl 2):54
Doursounian L, Grimberg J, Cazeau C, Touzard RC (1996) A new method of osteosynthesis in proximal humeral fractures: a new internal fixation device. Apropos of 17 cases followed over more than 2 years. Revue de chirurgie orthopedique et reparatrice de l’appareil moteur 82:743–752
Boileau P, Trojani C, Walch G, Krishnan SG, Romeo A, Sinnerton R (2001) Shoulder arthroplasty for the treatment of the sequelae of fractures of the proximal humerus. J Shoulder Elb Surg 10:299–308
Boyle MJ, Youn SM, Frampton CM, Ball CM (2013) Functional outcomes of reverse shoulder arthroplasty compared with hemiarthroplasty for acute proximal humeral fractures. J Shoulder Elb Surg 22:32–37
Tanner MW, Cofield RH (1983) Prosthetic arthroplasty for fractures and fracture-dislocations of the proximal humerus. Clin Orthop Relat Res 179:116–128
Giovale M, Mangano T, Roda E, Repetto I, Cerruti P, Kuqi E, Franchin F (2014) Shoulder hemiarthroplasty for complex humeral fractures: a 5 to 10-year follow-up retrospective study. Musculoskel Surg 98:27–33
Baudi P, Campochiaro G, Serafini F, Gazzotti G, Matino G, Rovesta C, Catani F (2014) Hemiarthroplasty versus reverse shoulder arthroplasty: comparative study of functional and radiological outcomes in the treatment of acute proximal humerus fracture. Musculoskel Surg 98:19–25
Grassi FA, Zorzolo I (2014) Reverse shoulder arthroplasty without subscapularis repair for the treatment of proximal humeral fractures in the elderly. Musculoskel Surg 98:5–13
Iacobellis C, Berizzi A, Biz C, Camporese A (2015) Treatment of proximal humeral fractures with reverse shoulder arthroplasty in elderly patients. Musculoskel Surg 99:39–44
Kristiansen B, Christensen SW (1986) Plate fixation of proximal humeral fractures. Acta Orthop Scand 57:320–323
Ehlinger M, Gicquel P, Clavert P, Bonnomet F, Kempf JF (2004) A new implant for proximal humeral fracture: experimental study of the basket plate. Revue de chirurgie orthopedique et reparatrice de l’appareil moteur 90:16–25
Cuny C, Darbelley L, Touchard O, Irrazi M, Beau P, Berrichi A, Empereur F (2003) Proximal 4-part humerus fractures treated by antegrade nailing with self-stabilizing screws: 31 cases. Revue de chirurgie orthopedique et reparatrice de l’appareil moteur 89:507–514
Compito CA, Self EB, Bigliani LU (1994) Arthroplasty and acute shoulder trauma. Reasons for success and failure. Clin Orthop Relat Res 307:27–36
Naranja RJ Jr, Iannotti JP (2000) Displaced three- and four-part proximal humerus fractures: evaluation and management. J Am Acad Orthop Surg 8:373–382
de Boer FA, van Kampen PM, Huijsmans PE (2016) The influence of subscapularis tendon reattachment on range of motion in reversed shoulder arthroplasty: a clinical study. Musculoskelet Surg. doi:10.1007/s12306-016-0401-8
Sabesan VJ, Lombardo D, Josserand D, Buzas D, Jelsema T, Petersen-Fitts GR, Wiater JM (2016) The effect of deltoid lengthening on functional outcome for reverse shoulder arthroplasty. Musculoskelet Surg. doi:10.1007/s12306-016-0400-9
Sabesan VJ, Lombardo DJ, Shahriar R, Petersen-Fitts GR, Wiater JM (2016) The effect of glenosphere size on functional outcome for reverse shoulder arthroplasty. Musculoskelet Surg. doi:10.1007/s12306-015-0396-6
Boileau P, Watkinson D, Hatzidakis AM, Hovorka I, Award Neer (2005) The Grammont reverse shoulder prosthesis: results in cuff tear arthritis, fracture sequelae, and revision arthroplasty. J Shoulder Elb Surg 15(2006):527–540
Ramirez MA, Ramirez J, Murthi AM (2012) Reverse total shoulder arthroplasty for irreparable rotator cuff tears and cuff tear arthropathy. Clin Sports Med 31:749–759
Smith CD, Guyver P, Bunker TD (2012) Indications for reverse shoulder replacement: a systematic review. J Bone Joint Surg Br 94:577–583
Khan WS, Longo UG, Ahrens PM, Denaro V, Maffulli N (2011) A systematic review of the reverse shoulder replacement in rotator cuff arthropathy, rotator cuff tears, and rheumatoid arthritis. Sports Med Arthrosc 19:366–379
Naveed MA, Kitson J, Bunker TD (2011) The Delta III reverse shoulder replacement for cuff tear arthropathy: a single-centre study of 50 consecutive procedures. J Bone Joint Surg Br 93:57–61
Nolan BM, Ankerson E, Wiater JM (2011) Reverse total shoulder arthroplasty improves function in cuff tear arthropathy. Clin Orthop Relat Res 469:2476–2482
Frankle M, Levy JC, Pupello D, Siegal S, Saleem A, Mighell M, Vasey M (2006) The reverse shoulder prosthesis for glenohumeral arthritis associated with severe rotator cuff deficiency. a minimum two-year follow-up study of sixty patients surgical technique. J Bone Joint Surg Am 88(Suppl 1 Pt 2):178–190
Vanhove B, Beugnies A (2004) Grammont’s reverse shoulder prosthesis for rotator cuff arthropathy. A retrospective study of 32 cases. Acta Orthop Belg 70:219–225
Sadoghi P, Leithner A, Vavken P, Holzer A, Hochreiter J, Weber G, Pietschmann MF, Muller PE (2011) Infraglenoidal scapular notching in reverse total shoulder replacement: a prospective series of 60 cases and systematic review of the literature. BMC Musculoskelet Disord 12:101
Anakwenze OA, Zoller S, Ahmad CS, Levine WN (2014) Reverse shoulder arthroplasty for acute proximal humerus fractures: a systematic review. J Shoulder Elb Surg 23:e73–e80
Cazeneuve JF, Cristofari DJ (2010) The reverse shoulder prosthesis in the treatment of fractures of the proximal humerus in the elderly. J Bone Joint Surg Br 92:535–539
Gallinet D, Clappaz P, Garbuio P, Tropet Y, Obert L (2009) Three or four parts complex proximal humerus fractures: hemiarthroplasty versus reverse prosthesis: a comparative study of 40 cases. Orthop Traumatol Surg Res OTSR 95:48–55
Reitman RD, Kerzhner E (2011) Reverse shoulder arthoplasty as treatment for comminuted proximal humeral fractures in elderly patients. Am J Orthop 40:458–461
Klein M, Juschka M, Hinkenjann B, Scherger B, Ostermann PA (2008) Treatment of comminuted fractures of the proximal humerus in elderly patients with the Delta III reverse shoulder prosthesis. J Orthop Trauma 22:698–704
Valenti P, Katz D, Kilinc A, Elkholti K, Gasiunas V (2012) Mid-term outcome of reverse shoulder prostheses in complex proximal humeral fractures. Acta Orthop Belg 78:442–449
Russo R, Della Rotonda G, Cautiero F, Ciccarelli M (2015) Reverse shoulder prosthesis to treat complex proximal humeral fractures in the elderly patients: results after 10-year experience. Musculoskel Surg 99(Suppl 1):17–23
Cazeneuve JF, Cristofari DJ (2009) Delta III reverse shoulder arthroplasty: radiological outcome for acute complex fractures of the proximal humerus in elderly patients. Orthop Traumatol Surg Res OTSR 95:325–329
Young SW, Segal BS, Turner PC, Poon PC (2010) Comparison of functional outcomes of reverse shoulder arthroplasty versus hemiarthroplasty in the primary treatment of acute proximal humerus fracture. ANZ J Surg 80:789–793
Lenarz C, Shishani Y, McCrum C, Nowinski RJ, Edwards TB, Gobezie R (2011) Is reverse shoulder arthroplasty appropriate for the treatment of fractures in the older patient? Early observations. Clin Orthop Relat Res 469:3324–3331
Gallinet D, Adam A, Gasse N, Rochet S, Obert L (2013) Improvement in shoulder rotation in complex shoulder fractures treated by reverse shoulder arthroplasty. J Shoulder Elb Surg 22:38–44
Bufquin T, Hersan A, Hubert L, Massin P (2007) Reverse shoulder arthroplasty for the treatment of three- and four-part fractures of the proximal humerus in the elderly: a prospective review of 43 cases with a short-term follow-up. J Bone Joint Surg Br 89:516–520
Neer CS II (1987) Displaced proximal humeral fractures. Part I. Classification and evaluation. By Charles S. Neer, I, 1970. Clin Orthop Relat Res 223:3–10
Duparc J (1995) Classification of articular fractures of the upper extremity of the humerus. Acta Orthop Belg 61(Suppl 1):65–70
Garnavos C, Kanakaris NK, Lasanianos NG, Tzortzi P, West RM (2012) New classification system for long-bone fractures supplementing the AO/OTA classification. Orthopedics 35:e709–e719
Constant CR, Murley AH (1987) A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res 214:160–164
Hudak PL, Amadio PC, Bombardier C (1996) Development of an upper extremity outcome measure: the DASH (disabilities of the arm, shoulder and hand) [corrected]. The upper extremity collaborative group (UECG). Am J Ind Med 29:602–608
Michener LA, McClure PW, Sennett BJ (2002) American Shoulder and Elbow Surgeons Standardized Shoulder Assessment Form, patient self-report section: reliability, validity, and responsiveness. J Shoulder Elb Surg 11:587–594
Dawson J, Fitzpatrick R, Carr A (1996) Questionnaire on the perceptions of patients about shoulder surgery. J Bone Joint Surg Br 78:593–600
Cazeneuve JF, Hassan Y, Kermad F, Brunel A (2008) Delta III reverse-ball-and-socket total shoulder prosthesis for acute complex fractures of the proximal humerus in elderly population. Eur J Orthop Surg Traumatol 18:81–86
Goutallier D, Postel JM, Bernageau J, Lavau L, Voisin MC (1994) Fatty muscle degeneration in cuff ruptures. Pre- and postoperative evaluation by CT scan. Clin Orthop Relat Res 304:78–83
Aldinger PR, Raiss P, Rickert M, Loew M (2010) Complications in shoulder arthroplasty: an analysis of 485 cases. Int Orthop 34:517–524
Edwards TB, Kadakia NR, Boulahia A, Kempf JF, Boileau P, Nemoz C, Walch G (2003) A comparison of hemiarthroplasty and total shoulder arthroplasty in the treatment of primary glenohumeral osteoarthritis: results of a multicenter study. J Shoulder Elb Surg 12:207–213
Jones KJ, Dines DM, Gulotta L, Dines JS (2013) Management of proximal humerus fractures utilizing reverse total shoulder arthroplasty. Curr Rev Musculoskelet Med 6:63–70
Cazeneuve JF, Cristofari DJ (2014) Grammont reversed prosthesis for acute complex fracture of the proximal humerus in an elderly population with 5–12 years follow-up. Orthop Traumatol Surg Res OTSR 100:93–97
Cazeneuve JF, Cristofari DJ (2006) Grammont reversed prosthesis for acute complex fracture of the proximal humerus in an elderly population with 5–12 years follow-up. Revue de chirurgie orthopedique et reparatrice de l’appareil moteur 92:543–548
Longo UG, Franceschetti E, Petrillo S, Maffulli N, Denaro V (2011) Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc 19:428–437
Garrigues GE, Johnston PS, Pepe MD, Tucker BS, Ramsey ML, Austin LS (2012) Hemiarthroplasty versus reverse total shoulder arthroplasty for acute proximal humerus fractures in elderly patients. Orthopedics 35:e703–e708
Grassi FA, Murena L, Valli F, Alberio R (2009) Six-year experience with the Delta III reverse shoulder prosthesis. J Orthop Surg 17:151–156
Ji JH, Jeong JY, Song HS, Ok JH, Yang SJ, Jeon BK, Kim TG, Moon YS, Kim YS (2013) Early clinical results of reverse total shoulder arthroplasty in the Korean population. J Shoulder Elb Surg 22:1102–1107
Kilic M, Berth A, Blatter G, Fuhrmann U, Gebhardt K, Rott O, Zenz P, Irlenbusch U (2010) Anatomic and reverse shoulder prostheses in fracture sequelae of the humeral head. Acta Orthop Traumatol Turc 44:417–425
Levy J, Frankle M, Mighell M, Pupello D (2007) The use of the reverse shoulder prosthesis for the treatment of failed hemiarthroplasty for proximal humeral fracture. J Bone Joint Surg Am 89:292–300
Martinez AA, Bejarano C, Carbonel I, Iglesias D, Gil-Albarova J, Herrera A (2012) The treatment of proximal humerus nonunions in older patients with reverse shoulder arthroplasty. Injury 43(Suppl 2):S3–S6
Martinez AA, Calvo A, Bejarano C, Carbonel I, Herrera A (2012) The use of the Lima reverse shoulder arthroplasty for the treatment of fracture sequelae of the proximal humerus. J Orthop Sci Off J Jpn Orthop Assoc 17:141–147
Postacchini R, Castagna A, Borroni M, Cinotti G, Postacchini F, Gumina S (2012) Total shoulder arthroplasty for the treatment of failed hemiarthroplasty in patients with fracture of the proximal humerus. J Shoulder Elb Surg 21:1542–1549
Rasmussen JV, Jakobsen J, Brorson S, Olsen BS (2012) The danish shoulder arthroplasty registry: clinical outcome and short-term survival of 2137 primary shoulder replacements. Acta Orthop 83:171–173
Wall B, Nove-Josserand L, O’Connor DP, Edwards TB, Walch G (2007) Reverse total shoulder arthroplasty: a review of results according to etiology. J Bone Joint Surg Am 89:1476–1485
Wall B, Walch G (2007) Reverse shoulder arthroplasty for the treatment of proximal humeral fractures. Hand Clin 23:425–430
Wellmann M, Struck M, Pastor MF, Gettmann A, Windhagen H, Smith T (2013) Short and midterm results of reverse shoulder arthroplasty according to the preoperative etiology. Arch Orthop Trauma Surg 133:463–471
Willis M, Min W, Brooks JP, Mulieri P, Walker M, Pupello D, Frankle M (2012) Proximal humeral malunion treated with reverse shoulder arthroplasty. J Shoulder ElbSurg 21:507–513
Cazeneuve JF, Cristofari DJ (2011) Long term functional outcome following reverse shoulder arthroplasty in the elderly. Orthop Traumatol Surg Res 97(6):583–589. doi:10.1016/j.otsr.2011.03.025
Berton A, Gulotta LV, Petrillo S, Florio P, Longo UG, Denaro V, Kontaxis A (2015) The effect of humeral version on teres minor muscle moment arm, length, and impingement in reverse shoulder arthroplasty during activities of daily living. J Shoulder Elbow Surg 24(4):578–586. doi:10.1016/j.jse.2014.08.019
Terragnoli F, Zattoni G, Damiani L, Cabrioli A, Li Bassi G (2007) Treatment of proximal humeral fractures with reverse prostheses in elderly patients. J Orthop Traumatol 8(2):71–76
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Longo, U.G., Petrillo, S., Berton, A. et al. Reverse total shoulder arthroplasty for the management of fractures of the proximal humerus: a systematic review. Musculoskelet Surg 100, 83–91 (2016). https://doi.org/10.1007/s12306-016-0409-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12306-016-0409-0