Abstract
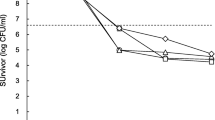
In this study, we investigated the effect of cryptic prophage regions in a blaNDM-1-bearing plasmid, which was identified in a patient from South Korea, on the survival of bacteria against adverse environmental conditions. First, we conjugated the intact plasmid and plasmids with deleted cryptic prophages into Escherichia coli DH5α. The E. coli transconjugants carrying the plasmid with intact cryptic prophages showed increased survival during treatment with a high concentration of NaCl, high and low temperatures, an oxidative stressor (H2O2), and an immunological stressor (human serum). By contrast, the transconjugants carrying the plasmid with a single-cryptic prophage knockout did not show any change in survival rates. mRNA expression analyses revealed that the genes encoding sigma factor proteins were highly upregulated by the tested stressors and affected the expression of various proteins (antioxidant, cell osmosis-related, heat shock, cold shock, and universal stress proteins) associated with the specific defense against each stress. These findings indicate that a bacterial strain carrying a plasmid with intact carbapenemase gene and cryptic prophage regions exhibited an increased resistance against simulated environmental stresses, and cryptic prophages in the plasmid might contribute to this enhanced stress resistance. Our study indicated that the coselection of antibiotic resistance and resistance to other stresses may help bacteria to increase survival rates against adverse environments and disseminate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
26 April 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s12275-023-00049-1
References
Battesti, A., Majdalani, N., and Gottesman, S. 2011. The RpoS-mediated general stress response in Escherichia coli. Annu. Rev. Microbiol.65, 189–213.
Bondy-Denomy, J. and Davidson, A.R. 2014. When a virus is not a parasitic: the beneficial effects of prophages on bacterial fitness. J. Microbiol.52, 235–242.
Čikoš, Š, Bukovská, A., and Koppel, J. 2007. Relative quantification of mRNA: comparison of methods currently used for realtime PCR data analysis., 113.
Dolejska, M., Villa, L., Poirel, L., Nordmann, P., and Carattoli, A. 2013. Complete sequencing of an IncHI1 plasmid encoding the carbapenemase NDM-1, the ArmA 16S RNA methylase and a resistance-nodulation-cell division/multidrug efflux pump. J. Antimicrob. Chemother.68, 34–39.
Falgenhauer, L., Yao, Y., Fritzenwanker, M., Schmiedel, J., Imirzalioglu, C., and Chakraborty, T. 2014. Complete genome sequence of phage-like plasmid pECOH89, encoding CTX-M-15. Genome Announc.2, e00356–14.
Feiner, R., Argov, T., Rabinovich, L., Sigal, N., Borovok, I., and Herskovits, A.A. 2015. A new prospective on lysogeny: prophages as active regulatory switches of bacteria. Nat. Rev. Microbiol.13, 641–650.
Fortier, L.C. and Sekulovic, O. 2013. Importance of prophages to evolution and virulence of bacterial pathogens. Virulence4, 354–365.
Händel, N., Hoeksema, M., Freijo Mata, M., Brul, S., and ter Kuile, B.H. 2011. Effects of stress, reactive oxygen species, and the SOS response on de novo acquisition of antibiotic resistance in Escherichia coli. Antimicrob. Agents Chemother.60, 1319–1327.
Johnson, C.M. and Grossman, A.D.. Integrative and conjugative elements (ICEs): What they do and how they work. Ann. Rev. Genet.49, 577–601.
Kim, S.Y. and Ko, K.S. 2019. Effects of prophage regions in a plasmid carrying a carbapenemase gene on survival against antibiotic stress. Int. J. Antimicrob. Agents53, 89–94.
Kim, S.Y., Shin, J., Shin, S.Y., and Ko, K.S. 2013. Characteristics of carbapenem-resistant Enterobacteriaceae isolates from Korea. Diagn. Microbiol. Infect. Dis.76, 486–490.
Liu, X., Li, Y., Guo, Y., Zeng, Z., Li, B., Wood, T.K., Cai, X., and Wang, X. 2015. Physiological function of rac prophage during biofilm formation and regulation of rac excision in Escherichia coli K-12. Sci. Rep.5, 16074.
Magaziner, S.J., Zeng, Z., Chen, B., and Salmond, G.P.C. 2019. The prophages of Citrobacter rodentium represent a conserved family of horizontally acquired mobile genetic elements associated with enteric evolution towards pathogenicity. J. Bacteriol.201, e00638–18.
Murakami, K., Ono, T., Viducic, D., Kayama, S., Mori, M., Hirota, K., Nemoto, K., and Miyake, Y.. Role for rpoS gene of Pseudomonas aeruginosa in antibiotic tolerance. FEMS Microbiol. Lett.242, 161–167.
Oliver, A., Coque, T.M., Alonso, D., Valverde, A., Baquero, F., and Cantón, R. 2005. CTX-M-10 linked to a phage-related element is widely disseminated among Enterobacteriaceae in a Spanish hospital. Antimicrob. Agents Chemother.49,.
Shin, J. and Ko, K.S. 2015. A plasmid bearing the blaCTX-M-15 gene and phage P1-like sequence from a sequence type 11 Klebsiella pneumoniae isolate. Antimicrob. Agents Chemother.59, 6608–6610
Siu, L.K., Fung, C.P., Chang, F.Y., Lee, N., Yeh, K.M., Koh, T.H., and Ip, M. 2011. Molecular typing and virulence analysis of serotype K1 Klebsiella pneumoniae strains isolated from liver abscess patients and stool samples from noninfectious subjects in Hong Kong, Singapore, and Taiwan. J. Clin. Microbiol.49, 3761–3765.
Wang, X., Kim, Y., Ma, Q., Hong, S.H., Pokusaeva, K., Sturino, J.M., and Wood, T.K. 2010. Cryptic prophages help bacteria cope with adverse environments. Nat. Commun.1, 147.
Wang, X., Kim, Y., and Wood, T.K. 2009. Control and benefits of CP4-57 prophage excision in Escherichia coli biofilms. ISME J.3, 1164–1179.
Wang, X. and Wood, T.K. 2011. IS5 inserts upstream of the master motility operon flhDC in a quasi-Lamarckian way. ISME J.5, 1517–1525.
Yamomoto, K., Yamanaka, Y., Shimada, T., Sarkar, P., Yoshida, M., Bhardwaj, N., Watanabe, H., Taira, Y., Chatterji, D., and Ishihama, A. 2018. Altered distribution of RNA polymerase lacking on the omega subunit within the prophages along the Escherichia coli K-12 genome. mSystems3, e00172–17
Acknowledgments
This work was supported by the Basic Science Research Program of the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (grant no. NRF-2019R1A2C2004879).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
12275_2020_9605_MOESM1_ESM.pdf
Cryptic prophages in a blaNDM-1-bearing plasmid increase bacterial survival against high NaCl concentration, high and low temperatures, and oxidative and immunological stressors
About this article
Cite this article
Kim, S.Y., Ko, K.S. RETRACTED ARTICLE: Cryptic prophages in a blaNDM-1-bearing plasmid increase bacterial survival against high NaCl concentration, high and low temperatures, and oxidative and immunological stressors. J Microbiol. 58, 483–488 (2020). https://doi.org/10.1007/s12275-020-9605-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-020-9605-6