Abstract
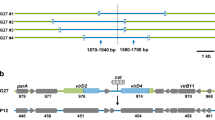
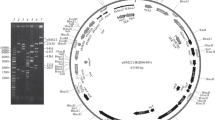
We isolated pHP69, a 9,153 bp plasmid from Helicobacter pylori with a 33.98% (G+C) content. We identified 11 open reading frames (ORFs), including replication initiation protein A (repA), fic (cAMP-induced filamentation protein), mccC, mccB, mobA, mobD, mobB, and mobC, as well as four 22 bp tandem repeat sequences. The nucleic acid and predicted amino acid sequences of these ORFs exhibited significant homology to those of other H. pylori plasmids. pHP69 repA encodes a replication initiation protein and its amino acid sequence is similar to those of replicase proteins from theta-type plasmids. pHP69 contains two types of repeat sequences (R1 and R2), a MOBHEN family mobilization region comprising mobC, mobA, mobB, and mobD, and genes encoding microcin B and C. Among the 36 H. pylori strains containing plasmids, mobA or mccBC are present in 12 or 6, respectively and 3 contain both genes. To examine intrinsic capability of H. pylori for conjugative plasmid transfer, a shuttle vector pBHP69KH containing pHP69 and replication origin of pBR322 was constructed. It was shown that this vector could stably replicate and be mobilized among clinical H. pylori strains and demonstrated to gene transfer by natural plasmid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alm, R.A. and Trust, T.J. 1999. Analysis of the genetic diversity of Helicobacter pylori: the tale of two genomes. J. Mol. Med. 77, 834–846.
Alting-Mees, M.A. and Short, J.M. 1989. pBluescript II: gene mapping vectors. Nucleic Acids Res. 17, 9494.
Backert, S., Kwok, T., and Konig, W. 2005. Conjugative plasmid DNA transfer in Helicobacter pylori mediated by chromosomally encoded relaxase and TraG-like proteins. Microbiology 151, 3493–3503.
Balzer, D., Pansegrau, W., and Lanka, E. 1994. Essential motifs of relaxase (TraI) and TraG proteins involved in conjugative transfer of plasmid RP4. J. Bacteriol. 176, 4285–4295.
Chattoraj, D.K. 2000. Control of plasmid DNA replication by iterons: no longer paradoxical. Mol. Microbiol. 37, 467–476.
Christie, P.J. and Vogel, J.P. 2000. Bacterial type IV secretion: conjugation systems adapted to deliver effector molecules to host cells. Trends Microbiol. 8, 354–360.
de Ungria, M.C., Kolesnikow, T., Cox, P.T., and Lee, A. 1999. Molecular characterization and interstrain variability of pHPS1, a plasmid isolated from the Sydney strain (SS1) of Helicobacter pylori. Plasmid 41, 97–109.
de Ungria, M.C., Tillett, D., Neilan, B.A., Cox, P.T., and Lee, A. 1998. A novel method of extracting plasmid DNA from Helicobacter species. Helicobacter 3, 269–277.
Durfee, T., Nelson, R., Baldwin, S., Plunkett III, G., Burland, V., Mau, B., Petrosino, J.F., Qin, X., Muzny, D.M., Ayele, M., and et al. 2008. The complete genome sequence of Escherichia coli DH10B: Insights into the biology of a laboratory workhorse. J. Bacteriol. 190, 2597–2606.
Francia, M.V., Varsaki, A., Garcillan-Barcia, M.P., Latorre, A., Drainas, C., and de la Cruz, F. 2004. A classification scheme for mobilization regions of bacterial plasmids. FEMS Microbiol. Rev. 28, 79–100.
González-Pastor, J.E., San Millán, J.L., Castilla, M.A., and Moreno, F. 1995. Structure and organization of plasmid genes required to produce the translation inhibitor microcin C7. J. Bacteriol. 177, 7131–7140.
Heuermann, D. and Haas, R. 1995. Genetic organization of a small cryptic plasmid of Helicobacter pylori. Gene 165, 17–24.
Hofreuter, D. and Haas, R. 2002. Characterization of two cryptic Helicobacter pylori plasmids: a putative source for horizontal gene transfer and gene shuffling. J. Bacteriol. 184, 2755–2766.
Hosaka, Y., Okamoto, R., Irinoda, K., Kaieda, S., Koizumi, W., Saigenji, K., and Inoue, M. 2002. Characterization of pKU701, a 2.5-kb plasmid, in a Japanese Helicobacter pylori isolate. Plasmid 47, 193–200.
Kersulyte, D., Velapatino, B., Mukhopadhyay, A.K., Cahuayme, L., Bussalleu, A., Combe, J., Gilman, R.H., and Berg, D.E. 2003. Cluster of type IV secretion genes in Helicobacter pylori’s plasticity zone. J. Bacteriol. 185, 3764–3772.
Kim, K.M., Lee, S.G., Park, M.G., Song, J.Y., Kang, H.L., Lee, W.K., Cho, M.J., Rhee, K.H., Youn, H.S., and Baik, S.C. 2007. Gamma-glutamyltranspeptidase of Helicobacter pylori induces mitochondria-mediated apoptosis in AGS cells. Biochem. Biophys. Res. Commun. 355, 562–567.
Kleanthous, H., Clayton, C.L., and Tabaqchali, S. 1991. Characterization of a plasmid from Helicobacter pylori encoding a replication protein common to plasmids in Gram-positive bacteria. Mol. Microbiol. 5, 2377–2389.
Komano. T., Utsumi, R., and Kawamukai, M. 1991. Functional analysis of the fic gene involved in regulation of cell division. Res. Microbiol. 142, 69–77.
Kuipers. E.J., Israel, D.A., Kusters, J.G., and Blasér, M.J. 1998. Evidence for a conjugation-like mechanism of DNA transfer in Helicobacter pylori. J. Bacteriol. 180, 2901–2905.
Lee, W.K., An, Y.S., Kim, K.H., Kim, S.H., Song, J.Y., Ryu, B.D., Choi, Y.J., Baik, S.C., Rhee, K.H., and Cho, M.J. 1997. Construction of a Helicobacter pylori-Escherichia coli shuttle vector for gene transfer in Helicobacter pylori. Appl. Environ. Microbiol. 63, 4866–4871.
Lee, W.K., Cho, M.J., Kim, K.H., Kim, S.K., Park, P.S., Lee, S.Y., Baik, S.C., and Rhee, K.H. 1995. Identification of Helicobacter pylori strains by pulse-field gel electrophoresis and restriction fragment length polymorphism. J. Korean Soc. Microbiol. 30, 641–650.
Marshall, B. and Warren, J.R. 1983. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet 4, 1273–1275.
Minnis, J.A., Taylor, T.E., Knesek, J.E., Peterson, W.L., and Mc-Intire, S.A. 1995. Characterization of a 3.5-kbp plasmid from Helicobacter pylori. Plasmid 34, 22–36.
Occhialini, A., Marais, A., Alm, R., Garcia, F., Sierra, R., and Mégraud, F. 2000. Distribution of open reading frames of plasticity region of strain J99 in Helicobacter pylori strains isolated from gastric carcinoma and gastritis patients in Costa Rica. Infect. Immun. 68, 6240–6249.
Parsonnet, J., Friedman, G.D., Vandersteen, D.P., Chang, Y., Vogelman, J.H., Orentreich, N., and Sibley, R.K. 1991. Helicobacter pylori infection and the risk of gastric carcinoma. N. Engl. J. Med. 325, 1127–1131.
Quiñones, M., Knesek, J.E., and McIntire, S.A. 2001. Sequence and gene expression analyses of plasmid pHPM8 from Helicobacter pylori reveal the presence of two operons with putative roles in plasmid replication and antibiotic activity. Plasmid 46, 223–228.
Rhee, K.H., Cho, M.J., Kim, J.B., Choi, S.K., Park, C.K., Kim, Y.C., Choi, J.H., and Choe, K.J. 1988. A prospective study on the Campylobacter pylori isolated from patients of gastroduodenal inflammatory conditions. J. Korean Soc. Microbiol. 23, 9–16.
Song, J.Y., Choi, S.H., Byun, E.Y., Lee, S.G., Park, Y.H., Park, S.G., Lee, S.K., Kim, K.M., Park, J.U., Kang, H.L., and et al. 2003a. Characterization of a small cryptic plasmid, pHP51, from a Korean isolate of strain 51 of Helicobacter pylori. Plasmid 50, 145–151.
Song, J.Y., Park, S.G., Kang, H.L., Lee, W.K., Cho, M.J., Park, J.U., Baik, S.C., Yoon, H.S., Ko, G.H., and Rhee, K.H. 2003b. pHP489, a Helicobacter pylori small cryptic plasmid, harbors a novel gene coding for a replication initiation protein. Plasmid 50, 236–241.
Vizán, J.L., Hernández-Chico, C., del Castillo, I., and Moreno, T. 1991. The peptide antibiotic microcin B17 induces double-strand cleavage of DNA mediated by E. coli DNA gyrase. EMBO J. 10, 467–476.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s12275-013-0727-y.
Rights and permissions
About this article
Cite this article
Joo, JS., Song, JY., Baik, SC. et al. Genetic organization and conjugal plasmid DNA transfer of pHP69, a plasmid from a Korean isolate of Helicobacter pylori . J Microbiol. 50, 955–961 (2012). https://doi.org/10.1007/s12275-012-2580-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2580-9