Abstract
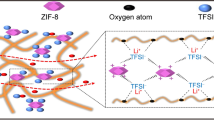
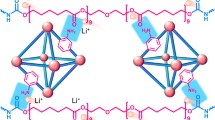
Poly(ethylene oxide) (PEO)-based solid polymer electrolytes (SPEs) are commonly used in lithium metal batteries (LMBs) for their good Li-salt solvating ability and easy processability. However, the relatively low Li-ion conduction ability hinders their further development. In this work, a novel hyperbranched-polyether-type composite solid polymer electrolyte (CSPE) is prepared via a quick cross-linking reaction between aldehyde-terminated polyethylene glycol (PEG) and hyperbranched poly(ethylene imine) (HPEI) in the presence of lithium salt and fluorine-containing Zr-based metal—organic framework (MOF) UiO-66-(F)4. The hydrogen bonds between the fluorine atoms and amino groups in the electrolyte help to the better dispersion of UiO-66-(F)4 in the polymer matrix, which is beneficial to solving the problem of aggregation of nanofillers. Besides, the CSPEs with the functional MOF fillers show improvements in both electrochemical and mechanical properties. Notably, the Li-ion transference number (\(t_{\rm{Li}^+}\)) is considerably enhanced from 0.23 to 0.54. All-solid-state LMBs based on the CSPE also present good cycling performances. A high specific discharge capacity of 141.4 mAh·g−1 is remained after 200 cycles at 0.2 C. This study not only provides an effective synthesis method of the cross-linked hyperbranched polymer electrolyte, but also puts forward a new strategy for uniform dispersion of inorganic fillers in CSPEs.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Li, N. W.; Yin, Y. X.; Yang, C. P.; Guo, Y. G. An artificial solid electrolyte interphase layer for stable lithium metal anodes. Adv. Mater. 2016, 28, 1853–1858.
Zhao, J.; Lu, Z. D.; Wang, H. T.; Liu, W.; Lee, H. W.; Yan, K.; Zhuo, D.; Lin, D. C.; Liu, N.; Cui, Y. Artificial solid electrolyte interphase-protected LixSi nanoparticles: An efficient and stable prelithiation reagent for lithium-ion batteries. J. Am. Chem. Soc. 2015, 137, 8372–8375.
Wang, Z. C.; Sun, Y. Y.; Mao, Y. Y.; Zhang, F. R.; Zheng, L.; Fu, D. S.; Shen, Y. B.; Hu, J. C.; Dong, H. L.; Xu, J. J. et al. Highly concentrated dual-anion electrolyte for non-flammable high-voltage Li-metal batteries. Energy Stor. Mater. 2020, 30, 228–237.
Yu, L.; Chen, S. R.; Lee, H.; Zhang, L. C.; Engelhard, M. H.; Li, Q. Y.; Jiao, S. H.; Liu, J.; Xu, W.; Zhang, J. G. A localized high-concentration electrolyte with optimized solvents and lithium difluoro(oxalate)borate additive for stable lithium metal batteries. ACS Energy Lett. 2018, 3, 2059–2067.
Liu, Y. M.; Qin, X. Y.; Zhou, D.; Xia, H. Y.; Zhang, S. Q.; Chen, G. H.; Kang, F. Y.; Li, B. H. A biscuit-like separator enabling high performance lithium batteries by continuous and protected releasing of NO3− in carbonate electrolyte. Energy Stor. Mater. 2020, 24, 229–236.
Sun, C. W.; Liu, J.; Gong, Y. D.; Wilkinson, D. P.; Zhang, J. J. Recent advances in all-solid-state rechargeable lithium batteries. Nano Energy 2017, 33, 363–386.
Yan, W. Q.; Gao, X. W.; Jin, X.; Liang, S. S.; Xiong, X. S.; Liu, Z. C.; Wang, Z. G.; Chen, Y. H.; Fu, L. J.; Zhang, Y. et al. Nonporous gel electrolytes enable long cycling at high current density for lithium-metal anodes. ACS Appl. Mater. Interfaces 2021, 13, 14258–14266.
Liu, B. H.; Jia, Y. K.; Yuan, C. H.; Wang, L. B.; Gao, X.; Yin, S.; Xu, J. Safety issues and mechanisms of lithium-ion battery cell upon mechanical abusive loading: A review. Energy Stor. Mater. 2020, 24, 85–112.
Perea, A.; Dontigny, M.; Zaghib, K. Safety of solid-state Li metal battery: Solid polymer versus liquid electrolyte. J. Power Sources 2017, 359, 182–185.
Castillo, J.; Qiao, L. X.; Santiago, A.; Judez, X.; De Buruaga, A. S.; Jiménez-Martín, G.; Armand, M.; Zhang, H.; Li, C. M. Perspective of polymer-based solid-state Li-S batteries. Energy Mater. 2022, 2, 200003.
Janek, J.; Zeier, W. G. A solid future for battery development. Nat. Energy 2016, 1, 16141.
Fan, L.; Wei, S. Y.; Li, S. Y.; Li, Q.; Lu, Y. Y. Recent progress of the solid-state electrolytes for high-energy metal-based batteries. Adv. Energy Mater. 2018, 8, 1702657.
Zhang, Q. Q.; Liu, K.; Ding, F.; Liu, X. J. Recent advances in solid polymer electrolytes for lithium batteries. Nano Res. 2017, 10, 4139–4174.
Wan, J. Y.; Xie, J.; Kong, X.; Liu, Z.; Liu, K.; Shi, F. F.; Pei, A.; Chen, H.; Chen, W.; Chen, J. et al. Ultrathin, flexible, solid polymer composite electrolyte enabled with aligned nanoporous host for lithium batteries. Nat. Nanotechnol. 2019, 14, 705–711.
Mindemark, J.; Lacey, M. J.; Bowden, T.; Brandell, D. Beyond PEO—Alternative host materials for Li+-conducting solid polymer electrolytes. Prog. Polym. Sci. 2018, 81, 114–143.
Zhang, Y. H.; Lu, W.; Cong, L. N.; Liu, J.; Sun, L. Q.; Mauger, A.; Julien, C. M.; Xie, H. M.; Liu, J. Cross-linking network based on poly(ethylene oxide): Solid polymer electrolyte for room temperature lithium battery. J. Power Sources 2019, 420, 63–72.
Waletzko, R. S.; Korley, L. T. J.; Pate, B. D.; Thomas, E. L.; Hammond, P. T. Role of increased crystallinity in deformation-induced structure of segmented thermoplastic polyurethane elastomers with PEO and PEO-PPO-PEO soft segments and HDI hard segments. Macromolecules 2009, 42, 2041–2053.
Xi, J. Y.; Qiu, X. P.; Zheng, S. X.; Tang, X. Z. Nanocomposite polymer electrolyte comprising PEO/LiClO4 and solid super acid: Effect of sulphated-zirconia on the crystallization kinetics of PEO. Polymer 2005, 46, 5702–5706.
Idris, N. H.; Senin, H. B.; Arof, A. K. Dielectric spectra of LiTFSI-doped chitosan/PEO blends. Ionics 2007, 13, 213–217.
Yan, X. J.; Peng, B.; Hu, B. W.; Chen, Q. PEO-urea-LiTFSI ternary complex as solid polymer electrolytes. Polymer 2016, 99, 44–48.
Gorecki, W.; Jeannin, M.; Belorizky, E.; Roux, C.; Armand, M. Physical properties of solid polymer electrolyte PEO(LiTFSI) complexes. J. Phys.:Condens. Matter 1995, 7, 6823–6832.
Xiao, Z. L.; Zhou, B. H.; Wang, J. R.; Zuo, C.; He, D.; Xie, X. L.; Xue, Z. G. PEO-based electrolytes blended with star polymers with precisely imprinted polymeric pseudo-crown ether cavities for alkali metal ion batteries. J. Membr. Sci. 2019, 576, 182–189.
Lu, Y. Y.; Tikekar, M.; Mohanty, R.; Hendrickson, K.; Ma, L.; Archer, L. A. Stable cycling of lithium metal batteries using high transference number electrolytes. Adv. Energy Mater. 2015, 5, 1402073.
Deng, K. R.; Qin, J. X.; Wang, S. J.; Ren, S.; Han, D. M.; Xiao, M.; Meng, Y. Z. Effective suppression of lithium dendrite growth using a flexible single-ion conducting polymer electrolyte. Small 2018, 14, 1801420.
Chen, L.; Fan, L. Z. Dendrite-free Li metal deposition in all-solidstate lithium sulfur batteries with polymer-in-salt polysiloxane electrolyte. Energy Stor. Mater. 2018, 15, 37–45.
Xue, R. J.; Angell, C. A. High ionic conductivity in PEO. PPO block polymer + salt solutions. Solid State Ion. 1987, 25, 223–230.
Ghelichi, M.; Qazvini, N. T.; Jafari, S. H.; Khonakdar, H. A.; Farajollahi, Y.; Scheffler, C. Conformational, thermal, and ionic conductivity behavior of PEO in PEO/PMMA miscible blend: Investigating the effect of lithium salt. J. Appl. Polym. Sci. 2013, 129, 1868–1874.
Cui, Z. Y.; Xu, Y. Y.; Zhu, L. P.; Wang, J. Y.; Xi, Z. Y.; Zhu, B. K. Preparation of PVDF/PEO-PPO-PEO blend microporous membranes for lithium ion batteries via thermally induced phase separation process. J. Membr. Sci. 2008, 325, 957–963.
Young, W. S.; Albert, J. N. L.; Schantz, A. B.; Epps, T. H. Mixed-salt effects on the ionic conductivity of lithium-doped PEO-containing block copolymers. Macromolecules 2011, 44, 8116–8123.
Marzantowicz, M.; Dygas, J. R.; Krok, F.; Tomaszewska, A.; Florjańczyk, Z.; Zygadło-Monikowska, E.; Lapienis, G. Star-branched poly(ethylene oxide) LiN(CF3SO2)2: A promising polymer electrolyte. J. Power Sources 2009, 194, 51–57.
Zheng, Y. W.; Li, X. W.; Li, C. Y. A novel de-coupling solid polymer electrolyte via semi-interpenetrating network for lithium metal battery. Energy Stor. Mater. 2020, 29, 42–51.
Khurana, R.; Schaefer, J. L.; Archer, L. A.; Coates, G. W. Suppression of lithium dendrite growth using cross-linked polyethylene/poly(ethylene oxide) electrolytes: A new approach for practical lithium-metal polymer batteries. J. Am. Chem. Soc. 2014, 136, 7395–7402.
Jayathilaka, P. A. R. D.; Dissanayake, M. A. K. L.; Albinsson, I.; Mellander, B. E. Effect of nano-porous Al2O3 on thermal, dielectric and transport properties of the (PEO)9LiTFSI polymer electrolyte system. Electrochim. Acta 2002, 47, 3257–3268.
Lin, D. C.; Liu, W.; Liu, Y. Y.; Lee, H. R.; Hsu, P. C.; Liu, K.; Cui, Y. High ionic conductivity of composite solid polymer electrolyte via in situ synthesis of monodispersed SiO2 nanospheres in poly(ethylene oxide). Nano Lett. 2016, 16, 459–465.
Lin, C. W.; Hung, C. L.; Venkateswarlu, M.; Hwang, B. J. Influence of TiO2 nano-particles on the transport properties of composite polymer electrolyte for lithium-ion batteries. J. Power Sources 2005, 146, 397–401.
Munichandraiah, N.; Scanlon, L. G.; Marsh, R. A.; Kumar, B.; Sircar, A. K. Influence of zeolite on electrochemical and physicochemical properties of polyethylene oxide solid electrolyte. J. Appl. Electrochem. 1995, 25, 857–863.
Li, W. W.; Zhang, S. P.; Wang, B. R.; Gu, S.; Xu, D.; Wang, J. N.; Chen, C. H.; Wen, Z. Y. Nanoporous adsorption effect on alteration of the Li+ diffusion pathway by a highly ordered porous electrolyte additive for high-rate all-solid-state lithium metal batteries. ACS Appl. Mater. Interfaces 2018, 10, 23874–23882.
Xi, J. Y.; Qiu, X. P.; Cui, M. Z.; Tang, X. Z.; Zhu, W. T.; Chen, L. Q. Enhanced electrochemical properties of PEO-based composite polymer electrolyte with shape-selective molecular sieves. J. Power Sources 2006, 156, 581–588.
Jamal, H.; Khan, F.; Hyun, S.; Min, S. W.; Kim, J. H. Enhancement of the ionic conductivity of a composite polymer electrolyte via surface functionalization of SSZ-13 zeolite for all-solid-state Li-metal batteries. J. Mater. Chem. A 2021, 9, 4126–4137.
Yuan, C. F.; Li, J.; Han, P. F.; Lai, Y. Q.; Zhang, Z. A.; Liu, J. Enhanced electrochemical performance of poly(ethylene oxide) based composite polymer electrolyte by incorporation of nano-sized metal-organic framework. J. Power Sources 2013, 240, 653–658.
Yu, J. M.; Guo, T. L.; Wang, C.; Shen, Z. H.; Dong, X. Y.; Li, S. H.; Zhang, H. G.; Lu, Z. D. Engineering two-dimensional metal-organic framework on molecular basis for fast Li+ conduction. Nano Lett. 2021, 21, 5805–5812.
Fu, X. W.; Hurlock, M. J.; Ding, C. F.; Li, X. Y.; Zhang, Q.; Zhong, W. H. MOF-enabled ion-regulating gel electrolyte for long-cycling lithium metal batteries under high voltage. Small 2022, 18, 2106225.
Huo, H. Y.; Wu, B.; Zhang, T.; Zheng, X. S.; Ge, L.; Xu, T. W.; Guo, X. X.; Sun, X. L. Anion-immobilized polymer electrolyte achieved by cationic metal-organic framework filler for dendrite-free solid-state batteries. Energy Stor. Mater. 2019, 18, 59–67.
Wang, H. C.; Wang, Q.; Cao, X.; He, Y. Y.; Wu, K.; Yang, J. J.; Zhou, H. H.; Liu, W.; Sun, X. M. Thiol-branched solid polymer electrolyte featuring high strength, toughness, and lithium ionic conductivity for lithium-metal batteries. Adv. Mater. 2020, 22, 2001259.
Hu, Z. G.; Peng, Y. W.; Kang, Z. X.; Qian, Y. H.; Zhao, D. A modulated hydrothermal (MHT) approach for the facile synthesis of UiO-66-type MOFs. Inorg. Chem. 2015, 54, 4862–4868.
Ameloot, R.; Aubrey, M.; Wiers, B. M.; Gómora-Figueroa, A. P.; Patel, S. N.; Balsara, N. P.; Long, J. R. Ionic conductivity in the metal-organic framework UiO-66 by dehydration and insertion of lithium tert-butoxide. Chem.—Eur. J. 2013, 19, 5533–5536.
Ji, P. F.; Drake, T.; Murakami, A.; Oliveres, P.; Skone, J. H.; Lin, W. B. Tuning Lewis acidity of metal-organic frameworks via perfluorination of bridging ligands: Spectroscopic, theoretical, and catalytic studies. J. Am. Chem. Soc. 2018, 140, 10553–10561.
Rey, I.; Lassègues, J. C.; Grondin, J.; Servant, L. Infrared and Raman study of the PEO-LiTFSI polymer electrolyte. Electrochim. Acta 1998, 43, 1505–1510.
Yang, J. J.; Shao, L.; Wang, X.; Yang, Y.; Tian, Z. Y.; Chen, W. X.; Zhang, G.; Shen, C. Effect of intermolecular interactions on the performance of UiO-66-laden solid composite polymer electrolytes. J. Alloys Compd. 2020, 845, 155179.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 52073285 and 11975238) and the authors also express gratitude for the help from the analysis and testing center at the University of Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
12274_2022_4523_MOESM1_ESM.pdf
Enhancing Li-ion conduction and mechanical properties via addition of fluorine-containing metal—organic frameworks in all-solid-state cross-linked hyperbranched polymer electrolytes
Rights and permissions
About this article
Cite this article
Wen, W., Zeng, Q., Chen, P. et al. Enhancing Li-ion conduction and mechanical properties via addition of fluorine-containing metal—organic frameworks in all-solid-state cross-linked hyperbranched polymer electrolytes. Nano Res. 15, 8946–8954 (2022). https://doi.org/10.1007/s12274-022-4523-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-022-4523-z