Abstract
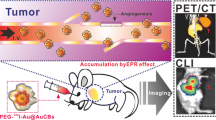
Nanoparticles with effective tumor accumulation and efficient renal clearance have attracted significant interests for clinical applications. We prepared 2.5 nm tyrosine based carbon dots (TCDs) with phenolic hydroxyl groups on the surface for directly 125I labeling. The 125I labeled polyethylene glycol (PEG) functionalized TCDs (125I-TCDPEGs) showed excellent radiochemical stability both in vitro and in vivo. Due to the enhanced permeability and retention effect, these 125I-TCDPEGs demonstrated a tumor accumulation around 4%–5% of the injected dose per gram (ID/g) for U87MG, 4T1, HepG2 and MCF7 tumor-bearing mice at 1 h post-injection. Meanwhile, the 125I-TCDPEGs also could be fast renally excreted, with less than 0.6% ID/g left in the liver and spleen within 24 h. These radioactive carbon dots not only can be used for cellular fluorescence imaging due to their intrinsic optical property, but are also effective single photon emission computed tomography (SPECT) imaging agents for tumor. Together with their excellent biocompatibility and stability, we anticipate these 125I-TCDPEGs of great potential for early tumor diagnosis in clinic. What’s more, our TCDPEGs are also proved to be feasible carriers for other iodine isotopes such as 127I and 131I for different biomedical application.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Charlton, K. A sustainable future for nuclear imaging. Nat. Rev. Phys. 2019, 1, 530–532.
Zeglis, B. M.; Holland, J. P.; Lebedev, A. Y.; Cantorias, M. V.; Lewis, J. S. Radiopharmaceuticals for imaging in oncology with special emphasis on positron-emitting agents In Nuclear Oncology: Pathophysiology and Clinical Applications. Strauss, H. W.; Mariani, G.; Volterrani D.; Larson S. M., Eds.; Springer: New York, 2013; pp 35–78.
Dunphy, M. P.; Lewis, J. S. Radiopharmaceuticals in preclinical and clinical development for monitoring of therapy with PET. J. Nucl. Med. 2009, 50 Suppl 1, 106S–121S.
Park, S. M.; Aalipour, A.; Vermesh, O.; Yu, J. H.; Gambhir, S. S. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2017, 2, 17014.
Sun, X. L.; Cai, W. B.; Chen, X. Y. Positron emission tomography imaging using radiolabeled inorganic nanomaterials. Acc. Chem. Res. 2015, 48, 286–294.
Pampaloni, M. H.; Nardo, L. PET/MRI radiotracer beyond 18F-FDG. PET Clin. 2014, 9, 345–349.
Mushtaq, S.; Jeon, J.; Shaheen, A.; Jang, B. S.; Park, S. H. Critical analysis of radioiodination techniques for micro and macro organic molecules. J. Radioanal. Nucl. Chem. 2016, 309, 859–889.
Cavina, L.; van der Born, D.; Klaren, P. H. M.; Feiters, M. C.; Boerman, O. C.; Rutjes, F. P. J. T. Design of radioiodinated pharmaceuticals: Structural features affecting metabolic stability towards in vivo deiodination. Eur. J. Org. Chem. 2017, 2017, 3387–3414.
Seevers, R. H.; Counsell, R. E. Radioiodination techniques for small organic molecules. Chem. Rev. 1982, 82, 575–590.
Bailey, G. S. Labeling of peptides and proteins by radioiodination. In Basic Protein and Peptide Protocols. Walker, J. M., Ed.; Springer: Humana Press, 1994; pp 441–448.
Dewanjee, M. K. Methods of radioiodination reactions with several oxidizing agents. In Radioiodination: Theory, Practice, and Biomedical Applications. Dewanjee, M. K., Ed.; Springer: Boston, MA, 1992; pp 129–218.
Yi, X.; Xu, M. Y.; Zhou, H. L.; Xiong, S. S.; Qian, R.; Chai, Z. F.; Zhao, L.; Yang, K. Ultrasmall hyperbranched semiconducting polymer nanoparticles with different radioisotopes labeling for cancer theranostics. ACS Nano2018, 12, 9142–9151.
Song, M. L.; Liu, N.; He, L.; Liu, G.; Ling, D. S.; Su, X. H.; Sun, X. L. Porous hollow palladium nanoplatform for imaging-guided trimodal chemo-, photothermal-, and radiotherapy. Nano Res. 2018, 11, 2196–2808.
Yi, X.; Yang, K.; Liang, C.; Zhong, X. Y.; Ning, P.; Song, G. S.; Wang, D. L.; Ge, C. C.; Chen, C. Y.; Chai, Z. F. et al. Imaging-guided combined photothermal and radiotherapy to treat subcutaneous and metastatic tumors using iodine-131-doped copper sulfide nanoparticles. Adv. Funct. Mater. 2015, 25, 4689–4699.
Phillips, E.; Penate-Medina, O.; Zanzonico, P. B.; Carvajal, R. D.; Mohan, P.; Ye, Y. P.; Humm, J.; Gönen, M.; Kalaigian, H.; Schoder, H. et al. Clinical translation of an ultrasmall inorganic optical-PET imaging nanoparticle probe. Sci. Transl. Med. 2014, 6, 260ra149.
Zhou, M.; Li, J. J.; Liang, S.; Sood, A. K.; Liang, D.; Li, C. CuS nanodots with ultrahigh efficient renal clearance for positron emission tomography imaging and image-guided photothermal therapy. ACS Nano2015, 9, 7085–7096.
Wen, L.; Chen, L.; Zheng, S. M.; Zeng, J. F.; Duan, G. X.; Wang, Y.; Wang, G. L.; Chai, Z. F.; Li, Z.; Gao, M. Y. Ultrasmall biocompatible WO3-x nanodots for multi-modality imaging and combined therapy of cancers. Adv. Mater. 2016, 28, 5072–5079.
Shen, S. D.; Jiang, D. W.; Cheng, L.; Chao, Y.; Nie, K. Q.; Dong, Z. L.; Kutyreff, C. J.; Engle, J. W.; Huang, P.; Cai, W. B. et al. Renal-clearable ultrasmall coordination polymer nanodots for chelator-free 64Cu-labeling and imaging-guided enhanced radiotherapy of cancer. ACS Nano2017, 11, 9103–9111.
Chen, L.; Chen, J. Y.; Qiu, S. S.; Wen, L.; Wu, Y.; Hou, Y.; Wang, Y.; Zeng, J. F.; Feng, Y.; Li, Z. et al. Biodegradable nanoagents with short biological half-life for SPECT/PAI/MRI multimodality imaging and PTT therapy of tumors. Small2018, 14, 1702700.
Lu, N.; Fan, W. P.; Yi, X.; Wang, S.; Wang, Z. T.; Tian, R.; Jacobson, O.; Liu, Y. J.; Yung, B. C.; Zhang, G. F. et al. Biodegradable hollow mesoporous organosilica nanotheranostics for mild hyperthermia-induced bubble-enhanced oxygen-sensitized radiotherapy. ACS Nano2018, 12, 1580–1591.
Chen, D. Q.; Zhang, G. Q.; Li, R. M.; Guan, M. R.; Wang, X. Y.; Zou, T. J.; Zhang, Y.; Wang, C. R.; Shu, C. Y.; Hong, H. et al. Biodegradable, hydrogen peroxide, and glutathione dual responsive nanoparticles for potential programmable paclitaxel release. J. Am. Chem. Soc. 2018, 140, 7373–7376.
Feng, H.; Qian, Z. S. Functional carbon quantum dots: A versatile platform for chemosensing and biosensing. Chem. Rec. 2018, 18, 491–505.
Roy, P.; Chen, P. C.; Periasamy, A. P.; Chen, Y. N.; Chang, H. T. Photoluminescent carbon nanodots: Synthesis, physicochemical properties and analytical applications. Mater. Today2015, 18, 447–458.
Yang, S. T.; Cao, L.; Luo, P. G.; Lu, F. S.; Wang, X.; Wang, H. F.; Meziani, M. J.; Liu, Y. F.; Qi, G.; Sun, Y. P. Carbon dots for optical imaging in vivo. J. Am. Chem. Soc. 2009, 131, 11308–11309.
Huang, P.; Lin, J.; Wang, X. S.; Wang, Z.; Zhang, C. L.; He, M.; Wang, K.; Chen, F.; Li, Z. M.; Shen, G. X. et al. Light-triggered theranostics based on photosensitizer-conjugated carbon dots for simultaneous enhanced-fluorescence imaging and photodynamic therapy. Adv. Mater. 2012, 24, 5104–5110.
Du, J. J.; Xu, N.; Fan, J. L.; Sun, W.; Peng, X. J. Carbon dots for in vivo bioimaging and theranostics. Small2019, 15, 1805087.
Zheng, M.; Ruan, S. B.; Liu, S.; Sun, T. T.; Qu, D.; Zhao, H. F.; Xie, Z. G.; Gao, H. L.; Jing, X. B.; Sun, Z. C. Self-targeting fluorescent carbon dots for diagnosis of brain cancer cells. ACS Nano2015, 9, 11455–11461.
Opacic, T.; Paefgen, V.; Lammers, T.; Kiessling, F. Status and trends in the development of clinical diagnostic agents. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1441.
Aherne, G. W.; James, S. L.; Marks, V. The radioiodination of bleomycin using iodogen. Clin. Chim. Acta1982, 119, 341–343.
Spetz, J.; Rudqvist, N.; Forssell-Aronsson, E. Biodistribution and dosimetry of free 211At, 125I–and 131I–in rats. Cancer Biother. Radiopharm. 2013, 28, 657–664.
Cranley, K.; Bell, T. K. 125I thyroid intakes: Consideration of thyroid radiation dose, and air and water concentration limits. Int. J. Appl. Radiat. Isot. 1979, 30, 161–163.
Zhu, A. Z.; Yoon, Y.; Liang, Z. X.; Voll, R.; Goodman, M. E.; Goodman, M. M.; Shim, H. Abstract 5227: Detection of metastatic potential by a novel small molecule F-18 PET imaging agent. Cancer Res. 2011, 71, 5227.
Foss, C. A.; Mease, R. C.; Fan, H.; Wang, Y. H.; Ravert, H. T.; Dannals, R. F.; Olszewski, R. T.; Heston, W. D.; Kozikowski, A. P.; Pomper, M. G. Radiolabeled small-molecule ligands for prostate-specific membrane antigen: In vivo imaging in experimental models of prostate cancer. Clin. Cancer Res. 2005, 11, 4022–4028.
Mankoff, D. A.; Link, J. M.; Linden, H. M.; Sundararajan, L.; Krohn, K. A. Tumor receptor imaging. J. Nucl. Med. 2008, 49 Suppl 2, 149S–163S.
Tang, S. H.; Peng, C. Q.; Xu, J.; Du, B. J.; Wang, Q. X.; Vinluan III, R. D.; Yu, M.X.; Kim, M. J.; Zheng, J. Tailoring renal clearance and tumor targeting of ultrasmall metal nanoparticles with particle density. Angew. Chem., Int. Ed. 2016, 55, 16039–16043.
Zhao, R. B.; Keen, L.; Kong, X. D. Clinical translation and safety regulation of nanobiomaterials In Nanobiomaterials: Classification, Fabrication and Biomedical Applications. Wang X. M.; Ramalingam M.; Kong, X. D., Eds.; Wiley Germany: Verlag, 2017; pp 459–479.
Acknowledgements
X. S. acknowledges the National Key Research and Development Program of China (No. 2016YFA0203600), the National Natural Science Foundation of China (Nos. 81971738 and 81571743), the Project Program of State Key Laboratory of Natural Medicines, and the China Pharmaceutical University (No. SKLNMZZRC05).
Author information
Authors and Affiliations
Corresponding author
Additional information
Notes
The authors declare no competing financial interest.
Electronic Supplementary Material
12274_2019_2549_MOESM1_ESM.pdf
Radioiodinated tyrosine based carbon dots with efficient renal clearance for single photon emission computed tomography of tumor
Rights and permissions
About this article
Cite this article
Liu, N., Shi, Y., Guo, J. et al. Radioiodinated tyrosine based carbon dots with efficient renal clearance for single photon emission computed tomography of tumor. Nano Res. 12, 3037–3043 (2019). https://doi.org/10.1007/s12274-019-2549-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-019-2549-7