Abstract
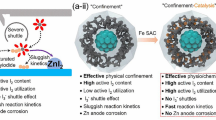
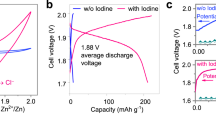
Rechargeable metal-iodine batteries are an emerging attractive electrochemical energy storage technology that combines metallic anodes with halogen cathodes. Such batteries using aqueous electrolytes represent a viable solution for the safety and cost issues associated with organic electrolytes. A hybrid-electrolyte battery architecture has been adopted in a lithium-iodine battery using a solid ceramic membrane that protects the metallic anode from contacting the aqueous electrolyte. Here we demonstrate an eco-friendly, low-cost zinc-iodine battery with an aqueous electrolyte, wherein active I2 is confined in a nanoporous carbon cloth substrate. The electrochemical reaction is confined in the nanopores as a single conversion reaction, thus avoiding the production of I3− intermediates. The cathode architecture fully utilizes the active I2, showing a capacity of 255 mAh·g−1 and low capacity cycling fading. The battery provides an energy density of ∼ 151 Wh·kg−1 and exhibits an ultrastable cycle life of more than 1,500 cycles.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dunn, B.; Kamath, H.; Tarascon, J.-M. Electrical energy storage for the grid: A battery of choices. Science 2011, 334, 928–935.
Sun, Y. M.; Liu, N.; Cui, Y. Promises and challenges of nanomaterials for lithium-based rechargeable batteries. Nature Energy 2016, 1, 16071.
Feng, N. N.; He, P.; Zhou, H. S. Critical challenges in rechargeable aprotic Li–O2 batteries. Adv. Energy Mater. 2016, 6, 1502303.
Hu, Z.; Liu, Q. N.; Chou, S.-L.; Dou, S.-X. Advances and challenges in metal sulfides/selenides for next-generation rechargeable sodium-ion batteries. Adv. Mater. 2017. DOI: 10.1002/adma.201700606.
Park, M.; Ryu, J.; Wang, W.; Cho, J. Material design and engineering of next-generation flow-battery technologies. Nat. Rev. Mater. 2016, 2, 16080.
Zhao, Y.; Ding, Y.; Li, Y. T.; Peng, L. L.; Byon, H. R.; Goodenough, J. B.; Yu, G. H. A chemistry and material perspective on lithium redox flow batteries towards highdensity electrical energy storage. Chem. Soc. Rev. 2015, 44, 7968–7996.
Broadhead, J. A new lithium-non-lithium non-aqueous secondary battery. In Eighth International Power Sources Symposium, Internat. Power Sources Symposium Committee, Croydon, Surrey, UK, 1972; pp 287–298.
Wang, Y. L.; Sun, Q. L.; Zhao, Q. Q.; Cao, J. S.; Ye, S. H. Rechargeable lithium/iodine battery with superior high-rate capability by using iodine-carbon composite as cathode. Energy Environ. Sci. 2011, 4, 3947–3950.
Zhao, Q.; Lu, Y. Y.; Zhu, Z. Q.; Tao, Z. L.; Chen, J. Rechargeable lithium-iodine batteries with iodine/nanoporous carbon cathode. Nano Lett. 2015, 15, 5982–5987.
Gong, D. C.; Wang, B.; Zhu, J. Y.; Podila, R.; Rao, A. M.; Yu, X. Z.; Xu, Z.; Lu, B. N. An iodine quantum dots based rechargeable sodium-iodine battery. Adv. Energy Mater. 2017, 7, 1601885.
Tian, H. J.; Gao, T.; Li, X. G.; Wang, X. W.; Luo, C.; Fan, X. L.; Yang, C. Y.; Suo, L. M.; Ma, Z. H.; Han, W. Q. et al. High power rechargeable magnesium/iodine battery chemistry. Nat. Commun. 2017, 8, 14083.
Tian, H. J.; Zhang, S. L.; Meng, Z.; He, W.; Han, W.-Q. Rechargeable aluminum/iodine battery redox chemistry in ionic liquid electrolyte. ACS Energy Lett. 2017, 2, 1170–1176.
Zhao, Y.; Wang, L. N.; Byon, H. R. High-performance rechargeable lithium-iodine batteries using triiodide/iodide redox couples in an aqueous cathode. Nat. Commun. 2013, 4, 1896.
Zhao, Y.; Byon, H. R. High-performance lithium-iodine flow battery. Adv. Energy Mater. 2013, 3, 1630–1635.
Yamamoto, T.; Hishinuma, M.; Yamamoto, A. Zn|ZnI2| iodine secondary battery using iodine-nylon-6 adduct as positive electrode, and its charge-discharge performance. Inorg. Chim. Acta 1984, 86, L47–L49.
Li, B.; Nie, Z. M.; Vijayakumar, M.; Li, G. S.; Liu, J.; Sprenkle, V.; Wang, W. Ambipolar zinc-polyiodide electrolyte for a high-energy density aqueous redox flow battery. Nat. Commun. 2015, 6, 6303.
Lee, J.; Srimuk, P.; Fleischmann, S.; Ridder, A.; Zeiger, M.; Presser, V. Nanoconfinement of redox reactions enables rapid zinc iodide energy storage with high efficiency. J. Mater. Chem. A 2017, 5, 12520–12527.
Puri, B. R.; Bansal, R. C. Iodine adsorption method for measuring surface area of carbon blacks. Carbon 1965, 3, 227–230.
Moser, J. R. Solid state lithium-iodine primary battery. U.S. Patent 3,660,163, May 2, 1972.
See, K. A.; Gerbec, J. A.; Jun, Y.-S.; Wudl, F.; Stucky, G. D.; Seshadri, R. A high capacity calcium primary cell based on the Ca–S system. Adv. Energy Mater. 2013, 3, 1056–1061.
Kiefer, W.; Bernstein, H. J. The UV-laser excited resonance Raman spectrum of the I3 − ion. Chem. Phys. Lett. 1972, 16, 5–9.
Pang, Q.; Liang, X.; Kwok, C. Y.; Nazar, L. F. Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 2016, 1, 16132.
Aurbach, D.; Pollak, E.; Elazari, R.; Salitra, G.; Kelley, C. S.; Affinito, J. On the surface chemical aspects of very high energy density, rechargeable Li–sulfur batteries. J. Electrochem. Soc. 2009, 156, A694–A702.
Xu, J. T.; Ma, J. M.; Fan, Q. H.; Guo, S. J.; Dou, S. X. Recent progress in the design of advanced cathode materials and battery models for high-performance lithium-X (X = O2, S, Se, Te, I2, Br2) batteries. Adv. Mater. 2017, 29, 1606454.
Reddy, T. B. Linden’s Handbook of Batteries; 4th ed. The McGraw-Hill Companies, Inc.: New York, 2010; pp15.10–15.11.
Dong, X. L.; Chen, L.; Su, X. L.; Wang, Y. G.; Xia, Y. Y. Flexible aqueous lithium-ion battery with high safety and large volumetric energy density. Angew. Chem., Int. Ed. 2016, 55, 7474–7477.
Suo, L. M.; Borodin, O.; Sun, W.; Fan, X. L.; Yang, C. Y.; Wang, F.; Gao, T.; Ma, Z. H.; Schroeder, M.; von Cresce, A. et al. Advanced high-voltage aqueous lithium-ion battery enabled by “water-in-Bisalt” electrolyte. Angew. Chem., Int. Ed. 2016, 55, 7136–7141.
Wang, F.; Suo, L. M.; Liang, Y. J.; Yang, C. Y.; Han, F. D.; Gao, T.; Sun, W.; Wang, C. S. Spinel LiNi0.5Mn1.5O4 cathode for high-energy aqueous lithium-ion batteries. Adv. Energy Mater. 2017, 7, 1600922.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Nos. 21171128 and 21603162), Tianjin Sci. & Tech. Program (No. 17JCYBJC21500), and the Fundamental Research Funds of Tianjin University of Technology.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bai, C., Cai, F., Wang, L. et al. A sustainable aqueous Zn-I2 battery. Nano Res. 11, 3548–3554 (2018). https://doi.org/10.1007/s12274-017-1920-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1920-9