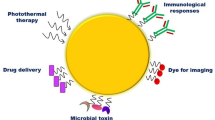
Abstract
Despite various therapeutic or diagnostic developments, cancer is still one of the most lethal diseases due to insufficiently adequate treatments and the delay of the early stage of disease detection. An image-guided drug delivery system (IGDDS), as a real-time noninvasive imaging assessment of therapeutic response, has the strong potential to improve the diagnosis and treatment of cancer because its imaging property offers the quantification of nanomedicine at the intended disease sites, the possible assurance of adequate treatment and elimination of undesirable delay of early-stage diagnosis due to low resolution. One of potential modality that overcomes these challenges could be the nanoconfinement of gold (Au) nanoparticles within other nanoparticles called "Particle-in-Particle (PIP)", which is a strong candidate of cancer treatment because of its "theranostic (therapy + diagnostics)" advantages including imaging (e.g., CT) and therapeutic hyperthermia application. In this review, we will elaborate on the current application of theranostic by nanoconfinement. Then, we will narrow down the gold nanoparticle-mediated theranostic application and its nanoconfinement advantages. Finally, the future direction for maximum nanoconfinement mediated cancer therapy will be included.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Based on statistics from the American Cancer Society, more than 1500 Americans die cancer-related deaths each day despite the availability of chemotherapy, radiotherapy, immunotherapy, gene therapy, and anti-angiogenesis treatments (Street 2019). Despite various therapeutic or diagnostic developments, cancer is still one of the most lethal diseases due to insufficiently adequate treatments and the delay of the early stage of disease detection. An image-guided drug delivery system (IGDDS), as a real-time noninvasive imaging assessment of therapeutic response, has the strong potential to improve the diagnosis and treatment of cancer because its imaging property offers the quantification of nanomedicine at the intended disease sites, the possible assurance of adequate treatment and elimination of undesirable delay of early stage diagnosis due to low resolution (Gabizon et al. 2014; Kaelin 2005; Paci et al. 2014). However, the use of IGDDS is still limited because of the several challenges, including the lack of sufficient sensitivity and resolution of detection and the relatively low therapeutic efficacy for clinical application. In order to improve IGDDS to meet needs of clinical application, currently nanoconfinement induced contrast enhancement (NICE) (Fahmy et al. 2017) and/or nanoconfinement induced therapeutic enhancement (NITE) (Gabizon et al. 2014) has been raised the attention in the field of cancer therapy due to their potential enhanced imaging and therapeutic efficacy. One of potential modalities that encompass both NICE and NITE could be the nanoconfinement of gold (Au) nanoparticles within other nanoparticles called “Particle-in-Particle (PIP)” (Yoo et al. 2018), which is a strong candidate of cancer treatment because of its “theranostic (therapy + diagnostics)” advantages including imaging (e.g., CT) and therapeutic hyperthermia application.
In this review, we will elaborate current application of theranostic or nanoconfinement. Then, we will narrow down the gold nanoparticle mediated theranostic application and its nanoconfinement advantages. Finally, the future direction for maximum nanoconfinement mediated cancer therapy will be included.
Cancer theranostics
Innovative modalities having simultaneous therapy and diagnostics (“Theranostics”) have gained significant interest for real time disease imaging and its specific treatment including cancer. There have been intensive research efforts on theranostics modalities development that show their significant therapeutic and diagnostic capabilities according to their different shapes, sizes, functionalities, targeting mechanisms, and imaging modalities. The ideal design paradigm for maximum efficacy of theranostic nanoparticles includes following key components: diagnosis component for noninvasive imaging, polymer coating for nanoparticle stability and its functional bioconjugation, and drug loading capabilities. These components will be introduced into the next three subchapters as well as current clinical trials of theranostics for the last subchapter.
Diagnosis components for noninvasive imaging
Over the several decades, various diagnosis components for noninvasive imaging have been investigated including magnetic resonance imaging (MRI) (Cademartiri et al. 2007), computed tomography (CT) (Simon 2000), positron emission tomography (PET) (Zhang et al. 2006), single-photon emission computed tomography (SPECT) (Urtasun et al. 1996), ultrasound (US) (Cheng et al. 2005) and fluorescence imaging (Artzi et al. 2011). These quantitative or semi-quantitative imaging modalities have been applied to employ highly sensitive image techniques to find out biodistribution of the labelled drugs or probe in the body. Incorporating radionuclides such as 99mTc, 111In, and 64Cu (Manrique et al. 1999; Spahn et al. 2004) into nanoparticles has been developed for noninvasive biodistribution studies through SPECT. One example of PET imaging contrast is the analog of glucose 2-[18F]-fluoro-2-deoxy-D-glucose (18F-FDG) (Markou et al. 2005). Near infrared (NIR) dyes such as indocyanine green (ICG), IR-783, and IR-780 are one of examples for fluorescence imaging and they show unique optical properties due to their absorption at NIR wavelength (Kuang et al. 2017). Currently, CT contrast agent for clinical application is based on iodine-based contrast agent (Gignac et al. 2016). One of the examples includes iodinated lipid contrast agent (Lipiodol®, Guerbet) (Kweon et al. 2010). It serves as a diagnostic tool to help detect its accumulation in solid tumors to monitor the tumor by CT. Lastly, a significant amount of research for MRI based contrast agent have been developed and among them Gd3+-based MRI contrast agent have been most successful (Iwaki et al. 2012).
Although there has been extensive amount of imaging modalities developed in the past few decades, the heterogeneity of tumor and toxicity mediated dose limitation still prevent from the clinical success of disease diagnosis. To overcome this challenge, the development of cross-imaging technologies has been noticed in the biomedical imaging field. For example, CT or MRI is hybridized with SPECT/PET because CT or MRI can provide 3-dimensional structural information while SPECT/PET lacks these features (Cherry 2009; Misri et al. 2012).
Polymer coating for nanoparticle stability and its functional bioconjugation
To target tumor sites effectively, the theranostics systems must have several requirements. First, they should be avoided by body defensive system (e.g., reticuloendothelial system: RES) to target maximum amount at the tumor sites. Second, they need to be maximumly localized in the tumor sites. To overcome these challenges, two major solutions have been introduced in the theranostic research field such as poly(ethylene glycol) (PEG) and targeting ligands coating on the surface of theranostic system (Ardana et al. 2014; Yao et al. 2016).
Once the theranostic systems are systemically administered into the patient’s body, they are passively targeted to tumor sites through the enhanced permeability and retention effect (EPR) effect (Maeda et al. 2000). During the circulation and before the passive entrance of tumor site, they must be survived. PEG allow them to extend their systemic half-life because PEG can empower the stealth property to the theranostic system (Zhang et al. 2017). Thus, prolonged circulation time of the theranostic systems enhance their passive tumor targeting capacity. Within the tumor microenvironment, the theranostic systems must be interacted with the tumor surface to expand their local delivery. By virtue of specific interaction of targeting ligand on the theranostic systems with the overexpressed receptors on the tumor cell surface (Mamot et al. 2003), enhanced tumor targeting efficacy will be achieved. It has the strong potential to result in maximum cancer theranostic outcome.
Currently, many biomarkers have been identified as possible tumor targeting receptors including the folate receptor (Zhang et al. 2012), transferrin receptors (Daniels et al. 2006), integrin (Liapis et al. 1996) and human epidermal receptor 2 (HER-2) (Nahta et al. 2004). The specific interaction between arginine-glycine-aspartic acid (RGD) peptide with avb3 integrin overexpressed on melanoma and glioblastomas allow the theranostic systems to target those tumors cells (Chen et al. 2016). The CD71 receptor or transferrin receptor (TfR) that is abundantly expressed in neuroblastoma and MDA-MB-231 metastatic breast cancer results in enhance tumor localization by transferrin coated theranostic system (Mendes et al. 2018). Lung carcinoma A549 cells overexpress the folate receptor on their cell surface and the folic acid coated theranostic system thus can target those cells specifically (Santra et al. 2011).
Drug loading capabilities
The delivery of drug and/or contrast agent by the theranostic systems is dependent on the carrier property and it is achieved by the chemical conjugation, or physical encapsulation. Biodegradable polymers that is conjugated with drugs or contrast agents is the smallest size of theranostic system. The bigger size (10–200 nm) of carrier (Fig. 1) such as dendrimer, emulsion, polymeric micelle, and liposomes encapsulate and deliver the drugs and/or contrast agents. For example, multifunctional polymeric micelles encapsulate alkynyl-DOTA-Gd as an MRI contrast agent and paclitaxel (6.67w/w% as a loading content) as an anticancer drug (Li et al. 2011a). 89Zr-labeled N-(2-hydroxypropyl)methacrylamined) copolymer (HPMA) is conjugated with doxorubicin to deliver both PET contrast agent and cancer drug (Koziolová et al. 2017). By incorporation of cisplatin and 1,1′-dioctadecyl-3,3,3′-tetramethylindotricarbocyanine iodine (DiR) into liposome, a versatile theranostic liposome for cancer therapy have been explored and the results from in vivo animal study shows efficient theranostic efficacy (Feng et al. 2016). To target and image glioma, the exosomes were loaded with curcumin and superparamagnetic iron oxide nanoparticles and were conjugated neuropilin-1-targeted peptides. The engineered exosomes provided the glioma-targeted images and therapeutic effects in vitro and in vivo (Jia et al. 2018).
Current clinical trials of theranostics as cancer treatment
In this section, several current clinical trials of theranostics as cancer treatment in US will be introduced according to the phase of each study (Table 1). The information for this section is based on the US National Library of Medicine. Currently (September 2019), total 12 clinical trials of theranostics for cancer therapy are active and among them there is only one (US National Library of Medicine 2000a) early phase of theranostics clinical trial. In this trials, radioactive drugs such as 131-Iodine and 90-Yttrium are used for radio therapy against neuroendocrine tumors and they can be used contrast agents for SPECT/CT. In phase 1 stage, there are active five clinical trials (US National Library of Medicine 2000c) of theranostics against thyroid cancer (2 trials), meningioma, neuroblastoma, and hepatocellular carcinoma. First thyroid cancer trial uses the radiolabeled gastrin analogs (117Lu-DOTA-(dGlu)6-Ala-Tyr-Gly-Trp-Nle-Asp-Phe-NH2) as a radionuclide therapy. In this trial, the patient’s neck/thorax/abdomen are scanned by CT. In the next thyroid cancer trial, radiolabeled peptide drug (111In-CP04(DOTA-(dGlu)6-Ala-Tyr-Gly-Trp-Met-Asp-Phe-NH2) and SPECT/CT images are the therapeutic and diagnostic components. For the trials of meningioma, Cu-64 SARTATE and Cu-67 SARTATE are PET imaging and radionuclide therapeutic components, respectively. Both Cu-64 SARTATE and Cu-67 SARTATE also used for clinical trials of neuroblastoma. The last phase 1 trial of theranostics is the assessment of 177Lu-DOTATATE for radionuclide therapy and [68 Ga]DOTATATE for PET imaging of hepatocellular carcinoma. In phase 2, six clinical theranostic trials (US National Library of Medicine 2000b) are active including prostate cancer using 117Lu-PSMA as a prostate specific membrane antigen (PSMA) therapy and PET imaging, non-small cell lung cancer using 18F-2-18F-fluoro-2-deoxy-D-glucose as a tyrosine kinase receptor inhibitor and PET imaging contrast agent, neuroendocrine tumor using 90Y-DOTATOC for radiotherapy and 68 Ga-DOTATOC for PET/CT imaging, meningioma using Cu-64 SARTATE for radionuclide therapy and Cu-67 SARTATE for PET imaging, neuroblastoma using Cu-64 SARTATE for radionuclide therapy and Cu-67 SARTATE for PET imaging, and hepatocellular carcinoma using 177Lu-DOTATATE for radionuclide therapy and [68 Ga]DOTATATE for PET imaging. Up to now, there is no phase 3 and phase 4 clinical trials of theranostics that are active now.
Nanoconfinement advantages for cancer treatment
Nanoconfinement defines the physical confinement within a nanosized region, and it is a very new concept in therapeutic and diagnostic fields (Cipolla et al. 2016). Most of the current studies related to nanoconfinement have dealt with the physical change or enhancement after a confinement of agent within nanosized frame or capsules. For example, nanoconfined β-sheet allow silk to enhance its stiffness, strength, and toughness (Keten et al. 2010). From the study by Zuo et al. the glass transition temperature of ionic liquids was found to be dependent on the confinement of ionic liquid components (Zuo et al. 2019). On the contrary, there is very few researches available about nanoconfinement mediated imaging (1 paper from Pubmed by the key word of “nanoconfinement & contrast enhancement”) or therapeutic enhancement (0 research from Pubmed by the key word of “nanoconfinement & therapeutic enhancement), indicating its infant stage of research.
In our studies, we utilized this unique nanoconfinement phenomenon in the theranostic enhancement application. Nanoconfined imaging agent within nanoparticles showed 5–10 folds enhancement of imaging intensity, compared with its commercial counterpart (Fahmy et al. 2017). It provides us various advantages in the clinical application including potential dose reduction, minimum toxicity, enhanced imaging resolution, and disease detection enhancement for the early stage of disease detection. Also, gold nanoparticle encapsulated within PLGA-PEG micelle (Particle in Particle: PIP) was developed. The PIP platform contains aggregated gold nanoparticles which may allow increasing the surface area and increase the number of the nanoconfinement of gold compared to free gold nanoparticles in a given volume. Thus, this system enhances the generation of heat because the exothermic effect of the nanoparticle is depending on the surface area of each nanoparticle. On the basis of this principle, PIP revealed various advantages from nanoconfinement (Yoo et al. 2018): (i) the absorbance of PIP is able to shift to near-infra red (NIR) wavelength for the deep penetration purpose; (ii) it produces multi-folds heat production compared with individual gold nanoparticles or its clusters; and (iii) spherical gold PIP is able to hold its shape after multiple NIR applications without any loss of absorbance in NIR and heat production while gold nanorod PIP shows its shapes after multiple NIR application that leads to the loss of absorbance and heat production. Thus, nanoconfinement mediated therapeutic and diagnostic enhancement has a strong potential to relieve the burden of current clinical cancer treatment challenge. In this regard, we will introduce several examples of both nanoconfinement induced contrast or therapeutic enhancement at the following subchapters although very few researches are existed.
Nanoconfinement induced contrast enhancement
Although nanoconfinement is well recognized phenomenon, the contrast enhancement by nanoconfinement in Pubmed shows two literatures (Ho and Wang 2019; Law et al. 2014). Both papers are not directly related with imaging enhancement. However, there are few researches appeared in the Google and they are 2 papers and 1 patent related with NICE. The study by Tuyen Duong et al. (Nguyen et al. 2018) elaborates that nanoconfinement of iron oxide within poly(l-lactic-co-glycolic acid) (PLGA) (PLGA/SPION). Then, PLGA/SPIONs are encapsulated within liposome to make hybrid nanoconstruct that enhances magnetic relaxivity and thus it is able to image mouse osteosarcoma tumor by MRI. Also, another study demonstrates that the confinement of magnetic resonance contrast agents within polymer nanocontainers enable an increased relaxivity compared to an aqueous solution of the contrast agent. It applies successfully in vivo to yield enhanced contrast in MRI imaging (Malzahn et al. 2016).
The only patent (Fahmy et al. 2017) that described NICE is about the CT or MRI contrast agent. Specifically, iodine CT contrast agent or iron oxide MRI contrast agent are encapsulated into the polymeric nanoparticle to make Particle-in-Particle system and their CT or MRI contrasts are significantly enhanced (Fig. 2). It allows for the use of lower concentration of contrast agent which avoids the toxicity associated with high concentration of such agents.
Nanoconfinement induced therapeutic enhancement (NITE)
Compared with NICE, NITE has been actively studied. Although there is no literature appeared in the Pubmed using “nanoconfinement” and “therapeutic enhancement”, a couple of researches in the Google were found. The first study that was done by our lab is about the gold nanoparticle encapsulated within PLGA-PEG micelles, called Particle-in-Particle (PIP) system as an anticancer hyperthermia (Yoo et al. 2018) (Fig. 3). The results from this study show the significant heat production from PIP that suppress various cancer cells (Yoo et al. 2018). Also, iron oxide nanoparticle cluster encapsulated PLGA nanoparticles demonstrate higher transverse relaxivity, leading to enhance the pharmacological activity (Ragheb et al. 2013).
Gold nanoparticle and its PIP for nanoconfinement theranostic application
Gold nanomaterials such as spherical nanoparticles (AuNP), nanorods (AuNR), nanocages, and nanoshells (AuNS) have been designated as multi-functional probes due to their unique properties of optical quenching, X-ray absorption, and surface-enhanced raman scattering (SERS) (Boisselier and Astruc 2009; Huang et al. 2006). These sensing properties are applied to single diagnostic imaging techniques, but additional gold nanomaterial properties like size controllability, excellent biocompatibility, easy surface modification, and photothermal effects can be further used towards multi-purpose theranostic agents (Boisselier and Astruc 2009; Chithrani et al. 2006).
The gold nanoparticles have been used in cancer imaging as various different ways. First, since gold has a significant X-ray attenuation coefficient than other elements in human bodies, the accumulation of gold nanoparticles in tumors is able to increase X-ray attenuation for CT images of tumors. Thus, targeting labelled gold nanoparticles can be potential CT contrast agents (Aydogan et al. 2010). As a second example of gold nanoparticle for cancer imaging, it can be used as a light scattering-based imaging contrast agent. Generally, gold nanoparticles can scatter light according to their shape and size. Gold nanoparticles with greater size of 10 nm can be visualized by dark-field scattering microscopy and they are more photostable than a fluorescent dye (Huang et al. 2006). The third example of cancer imaging by gold nanoparticles include their application of the photoacoustic imaging (PAI) (Wang and Lu 2017). It is based on the acoustic waves generated by the thermal expansion of gold nanoparticles induced by optical excitation. The images of PAI are derived from the ultrasonic waves detection after pulsed laser irradiation. Gold nanoparticles have strong potential for PAI due to their photothermal conversion ability and tunable optical properties. The gold nanorods are able to visualize the mice ovarian cancer using PAI (Jokerst et al. 2012).
Most of gold nanoparticles as therapeutic purpose have been applied as photothermal therapy because of their production of heat upon external energy source such as laser. The extent of heat generation is directly proportional to the incident excitation power and the surface properties, shape, and size of gold nanoparticles. For example, spherical gold nanoparticles produce heat upon 500–600 nm wavelength of laser (Bernardy et al. 2010), while gold nanorod does upon near-infrared (NIR) laser (Dickerson et al. 2008).
The theranostic applications of gold with modifications have been demonstrated. Glycol chitosan AuNP (GC-AuNP) with matrix metalloproteinase (MMP)-sensitive peptide probes have been used for multimodal tumor imaging: (1) contrasted CT images of the tumor and (2) optical images of tumor region by fluorescence Cy5.5 (Sun et al. 2011). Nucleic acids integrated AuNS was demonstrated as theranostic probes for phototriggered siRNA delivery (Lee et al. 2014). PEG-coated AuNR (PEG-AuNR) was reported as a CT contrast agent as well as a therapeutic photothermal agent (Von Maltzahn et al. 2009). When NIR laser or high-intensity focused ultrasound was applied to raise the temperature of the medium (higher than the melting temperature of the phase-change material), the encapsulated molecules were released from the inside of the Au nanocage (Li et al. 2011b; Rey et al. 2010).
In our study, we have demonstrated comprehensively that nanoconfinement of imaging agent or gold leads to the significant enhancement of image intensity and temperature elevation. However, these study results are not optimized for ideal gold-based theranostics, and they are from a segregated individual study of each aspect such as diagnostics and therapeutics. Thus, we need to develop and optimize a gold-based theranostics that integrates these elements within one simple nanoparticle system to maximize the chance of its clinical application.
Future direction and summary
Although versatility of gold nanoparticles allow them to have strong potential to be an theranostics candidate, they still have limits to be developed into an ideal theranostics for clinical application because of the following unmet requirements: unsatisfactory imaging resolution to distinguish disease from surrounding tissues, insufficient heat production for hyperthermia, and low sensitivity against NIR for deep tissue penetration capability (Deatsch and Evans 2014; Wang et al. 2015). From the previous chapter about the clinical trials of theranostics, there are also no nanoparticle related trials but mostly drug and/or contrast imaging conjugates. Thus, it is of substantial clinical importance to develop a new gold-based theranostics that can support high enough imageable system and produce sufficient hyperthermia.
One of potential solution will be taking advantage of nanoconfinement of gold PIP as an innovative theranostic cancer treatment because it is able to produce multifold thermal energy as a hyperthermia cancer therapy and it also can enhance cancer imaging capability by nanoconfinement. The other potential solution could be decoration of specific tumor targeting antibody that is produced by cancer exosome. Since cancer exosomes resemble with mother cancer cell surface, the injection of these cancer exosome will be able to produce the potential antibody candidates that specifically target exosome surface. Thus, these antibodies also can specifically target mother cancer cell surface.
In summary, combining both nanoconfinement advantage of PIP system and cancer cell exosome mediated antibody coating has strong potential of clinical transfer of theranostic cancer treatment.
References
Ardana A, Whittaker AK, Thurecht KJ (2014) PEG-based hyperbranched polymer theranostics: optimizing chemistries for improved bioconjugation. Macromolecules 47:5211–5219. https://doi.org/10.1021/ma501196h
Artzi N, Oliva N, Puron C, Shitreet S, Artzi S, Bon Ramos A, Groothuis A, Sahagian G, Edelman ER (2011) In vivo and in vitro tracking of erosion in biodegradable materials using non-invasive fluorescence imaging. Nat Mater 10:890. https://doi.org/10.1038/nmat3095
Aydogan B, Li J, Rajh T, Chaudhary A, Chmura SJ, Pelizzari C, Wietholt C, Kurtoglu M, Redmond P (2010) AuNP-DG: deoxyglucose-labeled gold nanoparticles as X-ray computed tomography contrast agents for cancer imaging. Mol Imag Biol 12:463–467. https://doi.org/10.1007/s11307-010-0299-8
Bernardy N, Romio AP, Barcelos EI, Pizzol CD, Dora CL, Lemos-Senna E, Araujo PH, Sayer C (2010) Nanoencapsulation of quercetin via miniemulsion polymerization. J Biomed Nanotechnol 6:181–186. https://doi.org/10.1166/jbn.2010.1107
Boisselier E, Astruc D (2009) Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem Soc Rev 38:1759–1782. https://doi.org/10.1039/B806051G
Cademartiri F, Palumbo AA, Maffei E (2007) Non invasive imaging of coronary arteries with 64-slice CT and 1.5 T MRI: challenging invasive techniques. Acta Bio Med Atenei Parmensis 78:6–15
Chen H, Niu G, Wu H, Chen X (2016) Clinical application of radiolabeled RGD peptides for PET imaging of integrin αvβ3. Theranostics 6:78. https://doi.org/10.7150/thno.13242
Cheng LK, Sands G, French R, Withy S, Wong S, Legget M, Smith W, Pullan AJ (2005) Rapid construction of a patient-specific torso model from 3D ultrasound for non-invasive imaging of cardiac electrophysiology. Med Biol Eng Compu 43:325–330. https://doi.org/10.1007/BF02345808
Cherry SR (2009) Multimodality imaging: beyond pet/ct and spect/ct. Semin Nucl Med 39:348–353. https://doi.org/10.1053/j.semnuclmed.2009.03.001
Chithrani BD, Ghazani AA, Chan WC (2006) Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett 6:662–668. https://doi.org/10.1021/nl052396o
Cipolla D, Wu H, Salentinig S, Boyd B, Rades T, Vanhecke D, Petri-Fink A, Rothin-Rutishauser B, Eastman S, Redelmeier T (2016) Formation of drug nanocrystals under nanoconfinement afforded by liposomes. RSC Adv 6:6223–6233. https://doi.org/10.1039/C5RA25898G
Daniels TR, Delgado T, Helguera G, Penichet ML (2006) The transferrin receptor part II: targeted delivery of therapeutic agents into cancer cells. Clinical Immunol 121:159–176. https://doi.org/10.1016/j.clim.2006.06.006
Deatsch AE, Evans BA (2014) Heating efficiency in magnetic nanoparticle hyperthermia. J Magn Magn Mater 354:163–172. https://doi.org/10.1016/j.jmmm.2013.11.006
Dickerson EB, Dreaden EC, Huang X, El-Sayed IH, Chu H, Pushpanketh S, Mcdonald JF, El-Sayed MA (2008) Gold nanorod assisted near-infrared plasmonic photothermal therapy (PPTT) of squamous cell carcinoma in mice. Cancer Lett 269:57–66. https://doi.org/10.1016/j.canlet.2008.04.026
Fahmy T, Sinusas A, Kim D (2017) Compositions for nanoconfinement induced contrast enhancement and methods of making and using thereof. Google Patents.
Feng L, Gao M, Tao D, Chen Q, Wang H, Dong Z, Chen M, Liu Z (2016) Cisplatin-prodrug-constructed liposomes as a versatile theranostic nanoplatform for bimodal imaging guided combination cancer therapy. Adv Func Mater 26:2207–2217. https://doi.org/10.1002/adfm.201504899
Gabizon A, Bradbury M, Prabhakar U, Zamboni W, Libutti S, Grodzinski P (2014) Cancer nanomedicines: closing the translational gap. Lancet 384:2175–2176. https://doi.org/10.1016/S0140-6736(14)61457-4
Gignac PM, Kley NJ, Clarke JA, Colbert MW, Morhardt AC, Cerio D, Cost IN, Cox PG, Daza JD, Early CM (2016) Diffusible iodine-based contrast-enhanced computed tomography (diceCT): an emerging tool for rapid, high-resolution, 3-D imaging of metazoan soft tissues. J Anat 228:889–909. https://doi.org/10.1111/joa.12449
Ho TA, Wang Y (2019) Enhancement of oil flow in shale nanopores by manipulating friction and viscosity. Physical Chemistry Chemical Physics. https://doi.org/10.1039/C9CP01960J
Huang X, El-Sayed IH, Qian W, El-Sayed MA (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128:2115–2120. https://doi.org/10.1021/ja057254a
Iwaki S, Hanaoka K, Piao W, Komatsu T, Ueno T, Terai T, Nagano T (2012) Development of hypoxia-sensitive Gd3+-based MRI contrast agents. Bioorg Med Chem Lett 22:2798–2802. https://doi.org/10.1016/j.bmcl.2012.02.071
Jia G, Han Y, An Y, Ding Y, He C, Wang X, Tang Q (2018) NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials 178:302–316. https://doi.org/10.1016/j.biomaterials.2018.06.029
Jokerst JV, Cole AJ, Van De Sompel D, Gambhir SS (2012) Gold nanorods for ovarian cancer detection with photoacoustic imaging and resection guidance via Raman imaging in living mice. ACS Nano 6:10366–10377. https://doi.org/10.1021/nn304347g
Kaelin WG Jr (2005) The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer 5:689–698. https://doi.org/10.1038/nrc1691
Keten S, Xu Z, Ihle B, Buehler MJ (2010) Nanoconfinement controls stiffness, strength and mechanical toughness of β-sheet crystals in silk. Nat Mater 9:359. https://doi.org/10.1038/NMAT2704
Koziolová E, Goel S, Chytil P, Janoušková O, Barnhart TE, Cai W, Etrych T (2017) A tumor-targeted polymer theranostics platform for positron emission tomography and fluorescence imaging. Nanoscale 9:10906–10918. https://doi.org/10.1039/C7NR03306K
Kuang Y, Zhang K, Cao Y, Chen X, Wang K, Liu M, Pei R (2017) Hydrophobic IR-780 dye encapsulated in cRGD-conjugated solid lipid nanoparticles for NIR imaging-guided photothermal therapy. ACS Appl Mater Interfaces 9:12217–12226. https://doi.org/10.1021/acsami.6b16705
Kweon S, Lee H-J, Hyung WJ, Suh J, Lim JS, Lim S-J (2010) Liposomes coloaded with iopamidol/lipiodol as a RES-targeted contrast agent for computed tomography imaging. Pharm Res 27:1408–1415. https://doi.org/10.1007/s11095-010-0135-5
Law JJ, Guven A, Wilson LJ (2014) Relaxivity enhancement of aquated Tris (β-diketonate) gadolinium (III) chelates by confinement within ultrashort single-walled carbon nanotubes. Contrast Media Mol Imaging 9:409–412. https://doi.org/10.1002/cmmi.1603
Lee SJ, Lee A, Hwang SR, Park J-S, Jang J, Huh MS, Jo D-G, Yoon S-Y, Byun Y, Kim SH (2014) TNF-α gene silencing using polymerized siRNA/thiolated glycol chitosan nanoparticles for rheumatoid arthritis. Mol Ther 22:397–408. https://doi.org/10.1038/mt.2013.245
Li X, Qian Y, Liu T, Hu X, Zhang G, You Y, Liu S (2011a) Amphiphilic multiarm star block copolymer-based multifunctional unimolecular micelles for cancer targeted drug delivery and MR imaging. Biomaterials 32:6595–6605. https://doi.org/10.1016/j.biomaterials.2011.05.049
Li W, Cai X, Kim C, Sun G, Zhang Y, Deng R, Yang M, Chen J, Achilefu S, Wang LV (2011b) Gold nanocages covered with thermally-responsive polymers for controlled release by high-intensity focused ultrasound. Nanoscale 3:1724–1730. https://doi.org/10.1039/C0NR00932F
Liapis H, Flath A, Kitazawa S (1996) Integrin alpha V beta 3 expression by bone-residing breast cancer metastases. Diagn Mol Pathol 5:127–135. https://doi.org/10.1097/00019606-199606000-00008
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65:271–284. https://doi.org/10.1016/S0168-3659(99)00248-5
Malzahn K, Ebert S, Schlegel I, Neudert O, Wagner M, Schütz G, Ide A, Roohi F, Münnemann K, Crespy D (2016) Design and control of nanoconfinement to achieve magnetic resonance contrast agents with high relaxivity. Adv Healthcare Mater 5:567–574. https://doi.org/10.1002/adhm.201500748
Mamot C, Drummond DC, Greiser U, Hong K, Kirpotin DB, Marks JD, Park JW (2003) Epidermal growth factor receptor (EGFR)-targeted immunoliposomes mediate specific and efficient drug delivery to EGFR-and EGFRvIII-overexpressing tumor cells. Can Res 63:3154–3161. https://doi.org/10.1158/0008-5472.CAN-05-1093
Manrique A, Faraggi M, Vera P, Vilain D (1999) 201T1 and 99mTc-MIBI gated SPECT in patients with large perfusion defects and left ventricular dysfunction: comparison with equilibrium radionuclide angiography. J Nucl Med 40:805
Markou A, Manning P, Kaya B, Datta SN, Bomanji JB, Conway GS (2005) [18F] fluoro-2-deoxy-d-glucose ([18F] FDG) positron emission tomography imaging of thymic carcinoid tumor presenting with recurrent Cushing’s syndrome. Eur J Endocrinol 152:521–525. https://doi.org/10.1530/eje.1.01839
Mendes M, Sousa JJ, Pais A, Vitorino C (2018) Targeted theranostic nanoparticles for brain tumor treatment. Pharmaceutics 10:181. https://doi.org/10.3390/pharmaceutics10040181
Misri R, Saatchi K, Häfeli UO (2012) Nanoprobes for hybrid SPECT/MR molecular imaging. Nanomedicine 7:719–733. https://doi.org/10.2217/nnm.12.32
Nahta R, Hung M-C, Esteva FJ (2004) The HER-2-targeting antibodies trastuzumab and pertuzumab synergistically inhibit the survival of breast cancer cells. Can Res 64:2343–2346. https://doi.org/10.1158/0008-5472.CAN-03-3856
Nguyen TDT, Pitchaimani A, Ferrel C, Thakkar R, Aryal S (2018) Nano-confinement-driven enhanced magnetic relaxivity of SPIONs for targeted tumor bioimaging. Nanoscale 10:284–294. https://doi.org/10.1039/C7NR07035G
Paci A, Veal G, Bardin C, Levêque D, Widmer N, Beijnen J, Astier A, Chatelut E (2014) Review of therapeutic drug monitoring of anticancer drugs part 1–cytotoxics. Eur J Cancer 50:2010–2019. https://doi.org/10.1016/j.ejca.2014.04.014
Ragheb RR, Kim D, Bandyopadhyay A, Chahboune H, Bulutoglu B, Ezaldein H, Criscione JM, Fahmy TM (2013) Induced clustered nanoconfinement of superparamagnetic iron oxide in biodegradable nanoparticles enhances transverse relaxivity for targeted theranostics. Magn Reson Med 70:1748–1760. https://doi.org/10.1002/mrm.24622
Rey DA, Strickland AD, Kirui D, Niamsiri N, Batt CA (2010) In vitro self-assembly of gold nanoparticle-coated poly (3-hydroxybutyrate) granules exhibiting plasmon-induced thermo-optical enhancements. ACS Appl Mater Interfaces 2:1804–1810. https://doi.org/10.1021/am100306m
Santra S, Kaittanis C, Santiesteban OJ, Perez JM (2011) Cell-specific, activatable, and theranostic prodrug for dual-targeted cancer imaging and therapy. J Am Chem Soc 133:16680–16688. https://doi.org/10.1021/ja207463b
Simon BA (2000) Non-invasive imaging of regional lung function using X-ray computed tomography. J Clin Monit Comput 16:433–442. https://doi.org/10.1023/A:1011444826908
Spahn I, Coenen HH, Qaim SM (2004) Enhanced production possibility of the therapeutic radionuclides 64Cu, 67Cu and 89Sr via (n, p) reactions induced by fast spectral neutrons. Radiochim Acta 92:183–186. https://doi.org/10.1524/ract.92.3.183.30489
Street W (2019) Cancer facts & figures 2019. American Cancer Society, Atlanta
Sun IC, Eun DK, Koo H, Ko CY, Kim HS, Yi DK, Choi K, Kwon IC, Kim K, Ahn CH (2011) Tumor-targeting gold particles for dual computed tomography/optical cancer imaging. Angew Chem Int Ed 50:9348–9351. https://doi.org/10.1002/anie.201102892
Urtasun R, Parliament M, Mcewan A, Mercer J, Mannan R, Wiebe L, Morin C, Chapman J (1996) Measurement of hypoxia in human tumours by non-invasive SPECT imaging of iodoazomycin arabinoside. Br J Cancer Supplement 27:S209
US National Library of Medicine. Early Phase 1 (2000a) https://clinicaltrials.gov/ct2/results?term=theranostics&cond=cancer&age_v=&gndr=&type=&rslt=&phase=4&Search=Apply. Accessed 4 Dec 2019
US National Library of Medicine. Phase 2 (2000b) https://clinicaltrials.gov/ct2/results?term=theranostics&cond=cancer&age_v=&gndr=&type=&rslt=&phase=1&Search=Apply. Accessed 4 Dec 2019
US National Library of Medicine. Phase 1 (2000c) https://clinicaltrials.gov/ct2/results?term=theranostics&cond=cancer&age_v=&gndr=&type=&rslt=&phase=0&Search=Apply. Accessed 4 Dec 2019
Von Maltzahn G, Park J-H, Agrawal A, Bandaru NK, Das SK, Sailor MJ, Bhatia SN (2009) Computationally guided photothermal tumor therapy using long-circulating gold nanorod antennas. Can Res 69:3892–3900. https://doi.org/10.1158/0008-5472.CAN-08-4242
Wang S, Lu G (2017) Applications of gold nanoparticles in cancer imaging and treatment. Noble and precious metals-properties, nanoscale effects and applications. IntechOpen, London. https://doi.org/10.5772/intechopen.70901
Wang Y, Yang T, Ke H, Zhu A, Wang Y, Wang J, Shen J, Liu G, Chen C, Zhao Y (2015) Smart Albumin-Biomineralized Nanocomposites for Multimodal Imaging and Photothermal Tumor Ablation. Adv Mater 27:3874–3882. https://doi.org/10.1002/adma.201500229
Yao VJ, D’angelo S, Butler KS, Theron C, Smith TL, Marchio S, Gelovani JG, Sidman RL, Dobroff AS, Brinker CJ (2016) Ligand-targeted theranostic nanomedicines against cancer. J Control Release 240:267–286. https://doi.org/10.1016/j.jconrel.2016.01.002
Yoo E, Choi JH, Hoang NH, Lee JS, Vuong S, Hur B, Han P, Oh KT, Fahmy T, Kim D (2018) Particle-in-particle platform for nanoconfinement-induced oncothermia. ACS Appl Bio Mater 1:1927–1941. https://doi.org/10.1021/acsabm.8b00490
Zhang Z, Machac J, Helft G, Worthley SG, Tang C, Zaman AG, Rodriguez OJ, Buchsbaum MS, Fuster V, Badimon JJ (2006) Non-invasive imaging of atherosclerotic plaque macrophage in a rabbit model with F-18 FDG PET: a histopathological correlation. BMC Nucl Med 6:3. https://doi.org/10.1186/1471-2385-6-3
Zhang Y, Liu J-M, Yan X-P (2012) Self-assembly of folate onto polyethyleneimine-coated CdS/ZnS quantum dots for targeted turn-on fluorescence imaging of folate receptor overexpressed cancer cells. Anal Chem 85:228–234. https://doi.org/10.1021/ac3025653
Zhang K-L, Zhou J, Zhou H, Wu Y, Liu R, Wang L-L, Lin W-W, Huang G, Yang H-H (2017) Bioinspired, “active” stealth magneto-nanomicelles for theranostics combining efficient MRI and enhanced drug delivery. ACS Appl Mater Interfaces 9:30502–30509. https://doi.org/10.1021/acsami.7b10086
Zuo Y, Zhang Y, Huang R, Min Y (2019) The effect of nanoconfinement on the glass transition temperature of ionic liquids. Phys Chem Chem Phys 21:22–25. https://doi.org/10.1039/C8CP06479B
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kang, C., Kim, D. Nanoconfinement-mediated cancer theranostics. Arch. Pharm. Res. 43, 110–117 (2020). https://doi.org/10.1007/s12272-020-01217-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01217-2