Abstract
Accumulating evidence shows that the anti-cancer potential of the immune response that can be activated by modulation of the immunogenicity of dying cancer cells. This regulated cell death process is called immunogenic cell death (ICD) and constitutes a new innovating anti-cancer strategy with immune-modulatory potential thanks to the release of damage-associated molecular patterns (DAMPs). Some conventional clinically-used chemotherapeutic drugs, as well as preclinically-investigated compounds of natural origins such as anthracyclines, microtubule-destabilizing agents, cardiac glycosides or hypericin derivatives, possess such an immune-stimulatory function by triggering ICD. Here, we discuss the effects of ICD inducers on the release of DAMPs and the activation of corresponding signaling pathways triggering immune recognition. We will discuss potential strategies allowing to overcome resistance mechanisms associated with this treatment approach as well as co-treatment strategies to overcome the immunosuppressive microenvironment. We will highlight the potential role of metronomic immune modulation as well as targeted delivery of ICD-inducing compounds with nanoparticles or liposomal formulations to improving the immunogenicity of ICD inducers aiming at long-term clinical benefits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is by now well-accepted that the immune system efficiently contributes to the positive outcome of chemotherapeutic cancer treatment by activating immunogenic cell death (ICD). A limited number of natural and synthetic compounds with pharmacological interest were described to trigger cell death mechanisms leading to the emission of danger signals. These cellular entities are called damage-associated molecular patterns (DAMPs) that activate the immune system and eventually lead to the destruction of cancer cells. This immune-modulating mechanism which is primarily triggered by the dying cancer cells after cytotoxic stress can be considered as an additional essential component to the three well-established immune-mediated anti-cancer approaches, namely checkpoint inhibition, T cell mediated therapies and dendritic cell (DC)-based methods (Zitvogel et al. 2016). Whereas selected types of cancer, presenting elevated levels of mutations including lung, colon and skin cancer also present intrinsically high levels of immunogenicity, it becomes evident that neoplasms with lower mutational burdens including pediatric cancer (Vogelstein et al. 2013) could be more easily eradicated by augmenting their immunogenicity by compounds that trigger an immunogenic type of cell death. As such ICD can then act in synergy with the before mentioned immune-stimulatory treatment regimens (Messmer et al. 2019).
The basic idea of cancer cells to be used as a “vaccine” came from the original work of Coley, a Memorial Sloan Kettering surgeon who experimentally inoculated sarcoma and lymphoma patients with streptococcal cultures to achieve a cure rate over 10% by this bacterial stimulation of the patients. He is considered a pioneer of the earliest forms of immunotherapies. It was only in the 1960s and 70s that the discovery of T-cells and the emergence of chemotherapeutic drugs led to the hypothesis that the patient’s cancer cells triggered an immune response. The classical “self-/non-self theory” could not explain such an immune response, but it was Mazinger’s “danger” theory that explained how the immune system could become activated after damaged, dying cells liberated so-called alarmins, the danger-associated molecular patterns (DAMPs) eventually leading to the activation of the adaptive arm of the immune system (Matzinger 1994). The most prominent danger signals include calreticulin (CALR) cell surface exposure, the release of high mobility group box 1 (HMGB1) protein, and the liberation of ATP. Such DAMPS are critical for DC and effector T-cell activation (Radogna and Diederich 2018).
So far only a limited number of anti-cancer agents trigger ICD including natural compounds doxorubicin or mitoxantrone (see Chap 3) but also synthetic molecules oxaliplatin and cyclophosphamide. Interestingly only a limited number of such ICD-inducing compounds are well-characterized to this date so that future drug discovery is expected to identify novel yet unexplored ICD inducers (Radogna et al. 2019). Among the therapeutically-useful ICD inducers, natural compounds or hemisynthetic derivatives seem particularly interesting. Nature provides a considerable collection of molecular scaffolds from both terrestrial (Orlikova and Diederich 2012) and marine origins (Schumacher et al. 2011) with potent differentiation (Morceau et al. 2015), anti-inflammatory (Orlikova et al. 2014) or anti-cancer (Cerella et al. 2014; Mazumder et al. 2018a) potential. Moreover, natural compounds were shown to modulate epigenetic mechanisms linked to cancer (Schnekenburger et al. 2014), and immune modulation (Schnekenburger et al. 2019). As a result, over 60% of small-molecule anti-cancer drugs that were developed between 1981 and 2014 were developed from a natural product or were based on a natural pharmacophore (Thornburg et al. 2018). The aim of this review to document the biological anti-cancer activity of these pharmacologically-relevant compounds and their capacity to trigger ICD.
Molecular mechanisms of immunogenic cell death
Even though the molecular structure of the various pharmacological agents strongly differs without an apparent structure–activity relationship, Kroemer and collaborators established a clear picture of the required molecular mechanisms leading to ICD (Galluzzi et al. 2017). Briefly, ICD can be induced by a subset of intracellular stress and inflammatory mechanisms ultimately triggering canonical or non-canonical cell death mechanisms (Diederich and Cerella 2016). The interplay between these cell stress and death modalities is essential and is discussed elsewhere (Radogna and Diederich 2018).
A common denominator is certainly the induction of unfolded protein response (UPR) leading to CALR exposure at the outer leaflet of the plasma membrane followed by activation of endoplasmic reticulum (ER) chaperones. As an example, the anthracycline doxorubicin, a prototypical ICD inducer of natural origins kills cancer cells accompanied by ER stress-mediated CALR/protein disulfide isomerase family A member 3 (PDIA3, ERp57) exposure in endometrial carcinoma. This stress response then directly or indirectly triggered the maturation and chemotaxis of T lymphocytes leading to phagocytosis even of drug-resistant endometrial carcinoma cells culminating in potent immunogenic apoptosis (Xu et al. 2017).
In an attempt to determine molecular markers allowing to predict ICD-induction potential Bezu et al. performed a systematic analysis of clinically used anti-cancer agents to reveal that among all steps of the ER stress signaling pathways, exclusively the phosphorylation of eukaryotic translation initiation factor 2 subunit alpha (EIF2S1, eIF2α) correlated with CALR exposure under the control of eukaryotic translation initiation factor 2 alpha kinase 3 (EIF2AK3). Subsequently, the authors observed EIF2S1-dependent stress granule formation followed by autophagy induction. The authors concluded that EIF2S1 phosphorylation “constitutes a pathognomonic characteristic of ICD” (Bezu et al. 2018).
In addition to the stress-induced exposure of protein on the outer plasma membrane, as a result of the onset of cellular damage, selected proteins are secreted into the microenvironment namely the DAMPs high-mobility group protein HMGB1 and annexin 1 (Schnekenburger et al. 2019). HMGB1 possesses immuno stimulatory functions by binding to TLR4 but is also able to bind to other pattern recognition receptors (PRR). Ultimately, in ICD, the HMGB1-TLR4 axis triggers the synthesis of proinflammatory cytokines including type I interferons (IFN).
Stress-inducing compounds can ultimately lead to the onset of cell death mechanisms triggering a progressive loss of ATP, which is considered a hallmark of ICD if released in the extracellular environment. Premortem autophagy facilitates the release of ATP under these conditions and acts as a chemoattractant for DCs. Indeed, Castoldi et al. used anthracyclines to activate ICD and improved the ICD-mediated immune response by a 48-h fasting regimen. Such a food deprivation sequence is known to lead to induction of autophagy. In agreement with this observation, the systemic administration of the autophagy inhibitor dimethyl alpha-ketoglutarate reversed this positive effect. To achieve an immune activation, an intact immune system is required as well as the capacity of cells to rely on functional autophagy (Castoldi et al. 2019). Indeed, the anthracycline mitoxantrone appeared more toxic on tumors evolving in mice presenting a partial deficiency of the autophagic capacity (ATG4B−/−). The authors concluded that autophagy is rate-limiting for therapeutically relevant anti-cancer immune responses yet has a significant role in reducing the toxicity of chemotherapy.
The autophagic cellular response often leads necroptosis or other forms of controlled necrosis, and Lin et al. demonstrated the impact of autophagy in the immunogenicity of necroptosis (Lin et al. 2018) considered to be more highly immunogenic compared to other types of cell death including canonical apoptosis (Garg et al. 2013a). Eventually, under ICD stress, additional research revealed the release of interleukin 1β (IL1B) upon inflammasome signaling as well as the activation of TLR3, resulting in a type I IFN response that stimulates the production of C-X-C‐chemokine ligand 10 (CXCL10) (Galluzzi et al. 2017). An overview of the different steps of ICD and DAMPs release is represented in Fig. 1.
Induction of immunogenic cell death. After treatment with ICD inducers, cancer cells undergo cellular stress mechanisms (autophagy, endoplasmic reticulum stress) that leads to the release of danger associated molecular patterns (DAMPs). Briefly, cells expose so-called “find me signals” including calreticulin on their outer plasma membrane to increase uptake of dying cells. Dying cells release DAMPs like HMGB1 that binds to dendritic cell receptors including Toll-like receptor 2 and/or other pattern recognition receptors leading to dendritic cell maturation and antigen presentation to cytotoxic CD8+ T cells. Type I interferon (INF)-dependent chemokine production and cytokine stimulation including interleukin 1β (IL1B) trigger T cell priming. Antigen-specific immune response via the recruitment of T cells eventually generates a protective immune memory against the originally-targeted cancer cells. (Image credits (license): Colon tumor: Emmanuelm (CC-BY-3.0); mouse liver showing apoptotic cells indicated by arrows: Laboratory of Experimental Pathology, Division of Intramural Research, NIEHS (NIH) (public domain); macrophage: Obli (CC BY 2.0); Dendritic cell: National Institutes of Health (NIH) (public domain); T lymphocyte: NIAID/NIH (public domain))
Natural compound ICD-inducers
Whereas most chemotherapeutic treatments lead to suppression of the immune system, selected anti-cancer agents and some conventional chemotherapeutic drugs possess an immunostimulatory function by leading to ICD. A limited number of research teams not only screened for the cytotoxic capacity of given compounds but also assessed the ability of anti-cancer agents to trigger the appearance of an ICD phenotype. Menger et al. (2012) screened over 200 cytotoxic clinically-used but also preclinical experimental drugs by using a robotized system to identify exposure of CALR and PDIA3, development of autophagic nucleation points, the release of ATP and HMGB1 as well as the development of apoptosis versus necrosis. Another high-content screening aimed to identify HMGB1-releasing agents. The authors identified known ICD inducers, microtubule disruptors, and epigenetic modifiers (Liu et al. 2017). Altogether these approaches underline the importance of a coordinated search for novel and potent activations of ICD with immunomodulatory potential. Selected anti-cancer agents were shown to be particularly interesting:
Anthracyclines
The Italian Farmitalia Research Laboratories discovered the anti-cancer anthracycline glycosides daunorubicin (Fig. 2a, Table 1) and doxorubicin or adriamycin, the first broad-spectrum clinically-used chemotherapeutic agent (Arcamone 2009). This compound family was extracted initially from Streptomyces peucetius, from a soil sample of the region of Castel del Monte in Apulia, Italy. Daunorubicin allowed the semisynthetic synthesis of doxorubicin, one of the most used anti-cancer compounds known to trigger a mitotic catastrophe. Besides their potent cytotoxicity, anthracyclines were later shown to trigger an immunogenic effect. In 1988, Rosenberg and collaborators observed that a combination of IL2 and doxorubicin led to an in vivo reduction of lung metastases of weakly immunogenic MCA-105 sarcoma. The synergetic effect was supposed to be due to cytotoxic activity on the one hand, but also via increased susceptibility of tumor immune lysis and decreased immune suppressor activity (Papa et al. 1988).
Chemical structures of immunogenic cell death-inducing molecular scaffolds. Structures were drawn by using ChemDraw Professional 16.0. (Image credits (license): Streptomyces sp. (public domain); Taxus brevifolia: Jason Hollinger (CC BY 2.0); Colchicum autumnale: Lestat (Jan Mehlich (CC BY-SA 3.0); Halichondria okadai: https://openi.nlm.nih.gov, Daisuke Uemura (public domain); Digitalis purpurea: (public domain); Dolabella auricularia: Brocken Inaglory (CC BY-SA 3.0); Maytenus boaria: Pepe Robles (public domain); Strophanthus gratus: Prenn (CC BY-SA 3.0); Digitalis lanata: Haplochromis (CC BY 2.5); Drimia maritima: Javier martin (public domain); Hypericum perforatum: Bjoertvedt (CC BY-SA 3.0); Scutellaria baicalensis: Doronenko (CC BY-SA 3.0); Capsicum annuum: Ryan Bushby (CC BY 2.5))
Doxorubicin was described later to trigger a type of cell death that was dependent of the pan-caspase inhibitor zVAD-FMK. Moreover, the immunogenic effect induced by doxorubicin required DCs and CD8+ T-cells, as their inhibition abrogated the immunogenicity (Casares et al. 2005). Similar results were obtained by treating murine neuro-2a neuroblastoma cells (Inoue et al. 2014). When the authors co-cultured CD8α+ T-cells with doxorubicin-treated neuro-2a cells and bone marrow DCs, CD8α+ T-cells became responsive to anti-CD3/CD28 antibody stimulation accompanied by increased proliferation and IFNγ secretion. Besides in solid tumors, immunogenic apoptosis was also described in human acute myeloid leukemia (AML) after daunorubicin treatment of primary human AML cells leading to CALR exposure and release of heat shock protein (HSP) 70 and HSP90 (Fredly et al. 2011).
Doxorubicin also triggered the release of DAMPs in melanoma cells including CALR exposure, ATP secretion, up-regulation of type I IFN genes and HMGB1 secretion. In this instance, the ER stress pathway leading to eukaryotic translation initiation factor 2 alpha kinase 3 (EIF2AK3), protein kinase RNA-like endoplasmic reticulum kinase (PERK)) activation and phosphorylation of EIF2S1 was dispensable in melanoma ICD induction. The authors instead identified eukaryotic translation initiation factor 2 alpha kinase 2 (EIF2AK2, PKR) and eukaryotic translation initiation factor 2 alpha kinase 4 (EIF2AK4, GCN2) as critical mediators of EIF2S1 phosphorylation regulating the translocation of CALR to the melanoma outer membrane (Giglio et al. 2018). Another study involved Wnt family member 5A (WNT5A), produced by BRAF mutant melanoma cells. This regulator is involved in DC tolerance and tumor fibrosis by preventing antigen presentation and T-cell infiltration altogether contributing to an immunosuppressive tumor microenvironment in metastatic melanoma. Liu et al. demonstrated that “WNT5A trapping” transformed the immunosuppressive tumor microenvironment to facilitate ICD mediated by doxorubicin (Liu et al. 2018b).
Mitoxantrone is an anthracenedione anthracycline derivative used in cancer therapy and against multiple sclerosis. Mitoxantrone-treated, autophagy-deficient CT26 colorectal carcinoma cells released lower amounts of ATP compared to autophagy-competent CT26 (Michaud et al. 2011). Similarly, mitoxantrone reduced the growth of autophagy-competent but not of autophagy-deficient melanomas. This antiproliferative effect was abolished by depletion of CD4+ or CD8+ T-cells (Michaud et al. 2014). Recent results demonstrated that vaccination with mitoxantrone-treated primary colon cancer cells stimulated tumor-infiltrating lymphocytes in colorectal liver metastasis (Qin et al. 2019).
Results by Ladoire et al. further investigated the interconnection of autophagy and ICD in anthracycline-treated patient cohorts. They assessed double positivity for the presence of microtubule associated protein 1 light chain 3 alpha (MAP1LC3A, LC3B+) puncta, witnessing an active autophagic flux, and the presence of nuclear HMGB1 is a marker for improved breast cancer survival (Ladoire et al. 2015), especially in patients with a poor prognosis.
Mitoxantrone stimulated the production of CXCL8 and its mouse ortholog CXCL2 which are involved in the translocation of CALR to the outer plasma membrane. The knockdown of CXCL8/CXCL2 receptors reduced mitoxantrone-induced CALR exposure in both human and murine cancer cells, as well as the capacity to trigger an immune response in vivo. Conversely, exogenous CXCL2 improved the immunogenicity of dying cells depending on CALR (Sukkurwala et al. 2014b). Use of mitoxantrone allowed to elucidate fundamental regulatory mechanisms of CALR exposure. Indeed, colorectal cancer cells expressing low levels of miR-27a exposed more ecto-CALR on their surface and released higher amounts of ATP and HMGB1 compared cancer cells that express higher miR-27a levels. miR-27a is known to downregulate the EIF2AK3/phosphoinositide 3-kinase (PI3K)-dependent UPR. Altogether the authors suggested miR-27a as a master regulator of ICD (Colangelo et al. 2016).
Besides, the use of mitoxantrone allowed to dissect the role of CD47 regulator BCL2 Interacting Protein (BNIP)3 in melanoma. It is well known that the hypoxia-responsive protein BNIP3 regulates the expression of CD47, an immunosuppressive ‘don’t eat me’ signal that prevents clearance by phagocytes. In response to mitoxantrone-induced ICD, BNIP3 increased ATP secretion, phagocytic clearance, and the vaccination potential of the dying melanoma cells (Romano et al. 2018).
Microtubule-destabilizing agents
Colchicine (Fig. 2b, Table 1) from Colchicum autumnale is known since 1500 BC (Graham and Roberts 1953) and was the first compound identified as a microtubule-destabilizing agent (MDA) whereas the vinca alkaloids vinblastine and vincristine from the periwinkle Catharanthus roseus were discovered about 50 years ago and were described to have strong anti-cancer potential (van Der Heijden et al. 2004). The MDA paclitaxel (Fig. 2b) was discovered in the 1960s resulting from an NCI-funded screening program. Later it could be isolated from the bark of Taxus brevifolia, the Pacific yew (Weaver 2014). Named Taxol, this MDA became the first-line drug against ovarian, breast, bladder, prostate, and lung cancers. To overcome considerable toxicity and adverse effects, the FDA also approved the MDAs epothilone (Fig. 2b, Table 1) (Ixempra), eribulin (Halaven) a derivative from halichondrin B synthesized by the sponge Halichondria okadai (Hirata and Uemura 1986) (Fig. 2c), auristatin E (Johansson et al. 2017) (Fig. 2D) from the sea hare Dolabella auricularia (used as an antibody–drug conjugate: Brentuximab vedotin, Adcetris), and maytansine (Fig. 2d) (Kupchan et al. 1972) from Maytenus (Kumar 2017) (used as an antibody–drug conjugate: Trastuzumab emtansine, Kadcyla). These compounds are used as single chemotherapeutic agents or as antibody–drug conjugates (ADCs) (Steinmetz and Prota 2018).
More recently, MDAs were also described to modulate immune functions (Wen et al. 2011). Paclitaxel was mostly described as the active component of specific types of formulations, selected MDAs were investigated as single drugs to assess their ICD-inducing capacity. The effect of colchicine and derivatives were assessed on mouse B16F10 (B16) melanoma cells leading to ICD with the release of DAMPs HSP70, HSP90, and HMGB1 without affecting CALR exposure. DC vaccines pulsed with colchicine-treated tumor cell lysates reduced tumor growth, increased cytotoxic T-lymphocyte activity against tumors, and increased survival of treated mice. CD8+ and natural killer (NK) cells, not CD4+ cells, were described as the effector cells responsible for the observed immune-mediated anti-cancer effect (Wen et al. 2011). Epothilones extracted from myxobacterium Sorangium cellulosum (Cragg and Pezzuto 2016) improved the immune recognition of ovarian cancer cells by increasing the expression of human leukocyte antigen (HLA) Class I and inflammatory cytokines (Pellicciotta et al. 2011). The same compound triggered CALR exposure in immortalized non-transformed cell types, in malignant murine and human cell lines and tumor protein (TP)53-deficient primary mouse mammary gland cells (Senovilla et al. 2012).
Cardiac glycosides
Cardiac glycosides (CGs) are a well-known class of pharmacologically active compounds essentially because of their use against obstructive heart diseases. As an inhibitor of the essential Na/K ATPase, this compound category was also described to have potent anti-cancer potential (Schneider et al. 2017; Diederich et al. 2017; Slingerland et al. 2013) via induction of apoptosis or necrosis accompanied by induction of autophagy/mitophagy (Radogna et al. 2016) in solid tumors.
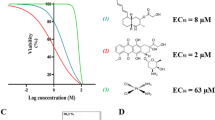
In 2012, Menger et al. demonstrated that CGs digoxin and digitoxin (Fig. 2e, Table 1), as well as ouabain and lanatoside C (Fig. 2f), exacerbated the anti-cancer effect of DNA-damaging agents only in immunocompetent mice. Moreover, a combination of chemotherapy and CGs led to a vaccination effect of syngeneic mice against injection of the same cells. The authors validated that the co-administration of digoxin to patients undergoing chemotherapy positively impacted the overall survival of breast, colorectal, head and neck, and hepatocellular carcinoma patients. These observations were especially true when patients were treated with chemotherapeutic agents unable to trigger ICD (Menger et al. 2012). In a similar screening, Sukkurwala et al. used the collection of anti-cancer compounds of the National Cancer Institute (NCI) Mechanistic Diversity Set and assessed the ability of over 800 compounds to reveal ICD hallmarks in vitro. The CGs scillaren A and proscillaridin (Fig. 2g), lanatoside, and digitoxigenin appeared to be the most potent ICD inducers also in this study (Sukkurwala et al. 2014a).
Hypericin derivatives
Physical stresses including ionizing irradiation, UVC light and essentially photodynamic Therapy (PDT) with hypericin, hydrostatic pressure and hyperthermia were described to activate anti-cancer immunity by induction of ICD (Adkins et al. 2014). Hypericin is a cytotoxic anthraquinone derivative that was extracted from Saint John’s wort, Hypericum perforatum (Kubin et al. 2005; Davids et al. 2008) (Fig. 2h, Table 1). Hypericin-mediated photodynamic therapy (HPDT) was shown to induce ICD (Garg et al. 2012) by using a pathway that differs from ICD induced by anthracyclines. Indeed, HPDT-induced CALR exposure independently of the presence of PDIA3. As the onset of efficient autophagy was crucial for anthracycline-induced ICD, a reduction of autophagy by ATG5 knockdown did not affect HPDT-induced ATP secretion. Moreover, cells with such a reduced autophagic capacity still exposed enhanced CALR induction on the outer membrane, induced DC maturation, IL6 secretion, CD4+/CD8+ T-cell proliferation, and IFNγ production (Garg et al. 2013b).
ER stress is nevertheless induced by HPDT and accompanied by activation of EIF2AK3 and the pro-apoptotic transcription factor DNA damage inducible transcript 3 (DDIT3, CHOP), induction of BCL2 like 11 (BCL2L11, Bim) and phorbol-12-myristate-13-acetate-induced protein 1 (PMAIP1, Noxa) and decrease anti-apoptotic myeloid cell leukemia (MCL)1 (Verfaillie et al. 2013). Altogether, for the treatment of high-grade glioma by HPDT, reactive oxygen species (ROS) and DAMPs release/presentation were shown to be required as their inhibition reduced immunogenicity. Moreover, the DC-associated MYD88 innate immune signal transduction adaptor, as well as CD8+ T-cells are essential (Garg et al. 2016).
Shikonin
Shikonin (Fig. 2h, Table 1), an anti-inflammatory and anti-cancer phytochemical (Chen et al. 2003) was initially used as a traditional Chinese remedy extracted from “Zicao”, the dried root of Lithospermum erythrorhizon. Chen et al. investigated the potential of shikonin as an adjuvant for DC-based cancer vaccines based on the potential of shikonin to trigger ICD. Indeed, it was shown that shikonin kills cancer cells by activating various types of cell death including controlled necrosis as well as extrinsic receptor-mediated and intrinsic and mitochondrial apoptosis. Shikonin-induced cell death abundantly releases DAMPs in the resultant tumor cell lysates. Chan et al. showed that the combined treatment with DAMPs and lipopolysaccharides (LPS) activates DCs and enhances the priming of Th1/Th17 effector T-cells. The authors showed that shikonin-tumor cell lysate-loaded DC vaccines induced cytotoxicity of splenocytes against target tumor cells, inhibition of tumor growth, and increased animal survival (Chen et al. 2012). Besides, shikonin efficiently induced ICD of 4T1 mouse mammary gland cells. DC vaccines pulsed with shikonin-treated tumor cell lysates suppressed rapamycin-enhanced metastasis and immunosuppressive regulatory T-cell expansion (Lin et al. 2015).
From a mechanistic point of view, shikonin was shown to target heterogeneous nuclear ribonucleoprotein A1 (HNRNPA1) in mammary carcinoma cells. The direct binding is primarily involved in shikonin-stimulated ICD as well as inhibition of post-transcriptional mRNA processing, including the nuclear export of newly synthesized mRNAs (Yin et al. 2016) most likely leading to a deregulation of de novo protein translation. In addition to apoptosis, shikonin treatment of 4T1 mouse cells can also trigger Receptor-interacting serine/threonine-protein kinase (RIPK)1/3-dependent necroptosis concomitant with enhanced autophagy. Shikonin-induced autophagy contributes to DAMPs upregulation. Interestingly, essentially DAMPs presented at the outer plasma membrane of dying cells CALR, HSP70 and 90 activated cocultured DCs. Interruption of the autophagic flux by chloroquine further upregulated these DAMPs and further activated DCs. The enhanced immunogenicity of tumor cells and vaccine efficacy due to the combination of shikonin and autophagy inhibitor chloroquine constitutes an innovative strategy for developing cancer vaccines (Lin et al. 2018).
Other compounds
A limited number of publications identified addition natural compounds as ICD inducers accompanied by DAMPs release. The immunogenic potential of these compounds will need to be further explored in the future to assess their therapeutic and immunomodulatory potential fully:
Septacidin (Fig. 2h, Table 1) produced by Streptomyces fibriatus is an l-heptopyranose with anti-cancer, antifungal, and analgesic activities (Tang et al. 2018). Moreover, septacidin promotes CALR exposure, ATP secretion and HGMB1 release from human osteosarcoma U2OS cells and murine fibrosarcoma MCA205 cells. Moreover, MCA205 cells killed by septacidin protected immunocompetent mice against a re-challenge with the same living cancer cells in a T lymphocyte-dependent manner (Sukkurwala et al. 2014a).
Wogonin (Fig. 2i, Table 1) is a flavonoid-like compound extracted initially from Scutellaria baicalensis. This compound triggers ER stress and ROS production, eventually leading to phosphorylation of EIF2AK3 and EIF2S1 further activating PI3K/AKT and relocating CALR/Annexin A1 to the outer cell membrane. Interestingly, calcineurin like EF-hand protein 1 (CHP1, p22), a Ca2+-binding protein, was shown to be necessary for CALR translocation. DAMPs, HMGB1 and ATP were released from wogonin-treated mouse gastric carcinoma MFC cells and activated DCs and cytokine release. Immunization with cells treated with wogonin significantly inhibited tumor growth in mice (Yang et al. 2012).
Vitamin E succinate (Fig. 2j, Table 1), also called α-tocopheryl acid succinate, was shown to target cancer cell mitochondria leading to ROS-dependent cell death within 30–60 min (Weber et al. 2003). Tumor growth suppression by this compound is not only due to cytotoxicity but also to activation of DCs (Ramanathapuram et al. 2005).
Capsaicin (Fig. 2i, Table 1) extracted from Capsicum induces pre- and early apoptotic cell surface exposure of CALR, HSP90, and HSP70 and ATP release in gastric cancer cell models. CALR exposure was prevented by inhibition of ER-Golgi traffic by brefeldin A. At late apoptotic stages, HMGB1, HSP90, and HSP70 were released (D’eliseo et al. 2013). As a result, capsaicin activated the phagocytosis of human osteosarcoma MG-63 cells by DCs, leading to IFNγ secretion (Jin et al. 2016).
Similarly, n3-polyunsaturated fatty acid docosahexaenoic acid (22:6n3, DHA) (Fig. 2j, Table 1) induces CALR exposure after treatment of human PaCa-44 pancreatic and EJ bladder cancer cell lines. Involvement of ROS was demonstrated as CALR exposure was inhibited by the natural antioxidant glutathione, the pan-caspase zVAD-FMK, and caspase-8-specific zIETD-FMK inhibitors (Molinari et al. 2011). Similarly, DHA promoted immunogenic apoptosis in multiple myeloma cells, with the emission of CALR, HSP90 and HMGB1 combined with the activation of pro-apoptotic autophagy and the activation of DCs by DHA-induced apoptotic MM cells (D’eliseo et al. 2017).
Resistance mechanisms
As for all anti-cancer treatments, potential resistance mechanisms against ICD inducers need to be investigated and the underlying mechanisms better understood. Considering that conventional chemotherapeutic agents like doxorubicin were used for over 40 years, conventional multi-drug resistance mechanisms that were described for such compounds are now also likely to reduce the potential of ICD inducers.
Classically, chemotherapeutic agents lead to multidrug resistance (MDR) accompanied by extrusion of chemotherapeutic agents including doxorubicin. Such mechanisms partially abolish the induction of ICD, and conversely, the inhibition of MDR contributes to improved ICD induction. As an example, zoledronic acid is a clinically used compound against bone complications in cancer and at the same time restores sensitivity towards doxorubicin improving ICD induction even in multidrug-resistant human cancer cells (Riganti et al. 2013). This aminobisphosphonate inhibits the mevalonate pathway and as a result, blocked signaling pathways downstream of RAS- and RHOA to eventually inhibit hypoxia inducible factor 1 subunit alpha (HIF1A)-driven P-glycoprotein expression. As less doxorubicin gets extruded, efficiently increased doxorubicin concentrations restored ICD that required the activation of DCs and CD8+ T-cell recruitment as previously described.
A different type of resistance can be triggered by DAMPs release itself. Loi et al. observed the importance of the cell surface receptor CD73 on the ICD-inducing effect of doxorubicin. Indeed, CD73 expression seemed inversely correlated to the successful ICD activation by doxorubicin (Loi et al. 2013). This effect was partially strong in breast cancer patients that overexpressed CD73. Interestingly, inhibition of CD73 reestablished doxorubicin-mediated immune modulation.
On the one hand, this observation adds CD73 as a potential therapeutic target in triple negative breast cancer, on the other side, it allowed recognizing that DAMPs emission could also antagonize the stimulatory immune modulation DAMPs were described to provide. CD73 is known to convert ATP to adenosine leading to inhibition of the immune response via inhibition of T-cell activation by the adenosinergic pathway. Cotreatments with a CD73-targeting antibody could contribute to reactivate further ICD in patients or cell models overexpressing CD73. Similar results were shown for ectopeptidase CD39. Interestingly, this mechanism described for doxorubicin could potentially be accurate for other ICD inducers insofar they trigger ATP secretion.
Garg et al. discovered a defect in the capacity of selected cell models to expose CALR. After treatment with mitoxantrone or hypericin-photodynamic therapy, the authors observed a reduced phagocytic clearance of AY27 bladder urothelial cell carcinoma cells due to low endogenous CALR protein levels “CALRlow.” Similarly, selected cancer patients also possessed such a phenotype. Conversely, the “CALRhigh”-phenotype of other patients was corresponding to an improved clinical response to therapy with paclitaxel for example (Garg et al. 2015).
Paclitaxel-induced ICD induction can be abrogated by immunosuppressive mechanisms controlled by programmed cell death receptor 1 (PD-1) and programmed cell death ligand 1 (PD-L1) (PD-1/PD-L1) immune checkpoint pathways. Tang et al. used checkpoint inhibitors as a countermeasure to this type of resistance mechanism. A rationally designed hybrid micelle delivering siRNA against PD-L1 and paclitaxel simultaneously to B16F10 cells reduced the expression of PD-L1 and the mTOR pathway marker P-S6K. This synergistic combination treatment reduced tumor growth by a combined effect involving cytotoxicity, activating cytotoxic T-cell immunity and inhibiting the mTOR pathway (Tang et al. 2019).
Targeted delivery of immunogenic cell death inducers
To overcome drug resistance mechanisms, co-treatments but essentially the targeted delivery of ICD inducers was shown to induce the potential of selected compounds further. Various examples of vectorization, encapsulation, and nano-formulation, as well as cotreatments with compounds reducing or preventing resistance, also contribute to improving the immunogenicity of the ICD inducing compound family.
Liu et al. suggested a nanoparticle delivery system to target desmoplastic melanoma and trigger the release of mitoxantrone and celastrol (pristimerin) (Fig. 2i), a cytotoxic quinone methide triterpenoid from the root of Tripterygium wilfordii (Thunder god vine) (Byun et al. 2009). Both compounds synergized to induce ICD (Liu et al. 2018a). Considering the potential problems of drug delivery and resistance mechanisms of some potent ICD-inducers such as doxorubicin, Rios-Doria et al. suggested the use of liposomal formulation called “Doxil.” Interestingly ICD-inducing doxorubicin liposomal formulation efficiently synergizes with other cancer immunotherapies to lead to an increased anti-cancer effect in mice (Rios-Doria et al. 2015). Similarly, pegylated liposomal doxorubicin triggers ICD in patients with ovarian, fallopian tube, or primary peritoneal carcinoma (clinical trial NCT 01666444). Here the authors efficiently combined this liposomal preparation to motolimod, a TLR8 agonist that stimulates innate immune responses. This combination treatment did not improve the clinical outcome of the patients compared to placebo but a statistically significant difference of overall survival of motolimod-treated patients was noticed on the basis of injection site reaction and in vitro immune responses to understand how to better identify patients for treatment with immunomodulatory agents in novel combinations and/or delivery approaches (Monk et al. 2017). Besides the formulation, selected papers also describe improved delivery methods. The ultrasound-controlled release of ICD inducers into nuclei using doxorubicin-liposome-microbubble complex was used by Huang et al. to enhance the induction of ICD in mouse LL/2 lung and colon CT26 tumor models (Huang et al. 2018). The authors measured elevated levels of activated CD8+ T-lymphocytes but lower amounts of inhibitor regulatory T-cells in such tumors. A strongly immunosuppressive microenvironment also characterizes glioblastoma. Here the authors approached this type of cancer by using a DC-mediated delivery of doxorubicin-polyglycerol-nanodiamond composites (Nano-DOX). These are potent DAMPs inducers triggering a DC-mediated immune response against glioblastoma. Nano-DOX-treated cells activated both DAMPs production and antigen release. DC-mediated delivery of Nano-DOX to stimulate immunogenicity of glioblastoma cells (Li et al. 2018). An interesting combination associated indoximod, which inhibits the indoleamine-pyrrole 2,3-dioxygenase (IDO1) pathway to doxorubicin to generate a novel DOX/IND-liposome. This carrier could be effectively combined with PD-1 blocking antibodies to even more efficiently eradicate lung metastases by combining checkpoint inhibition to ICD induction thus reactivating the microenvironment (Lu et al. 2018). The authors measured improved recruitment of CD8+ cytotoxic T lymphocytes, reduction of regulatory T-cells with an increase in CD8+/forkhead box P3 (FOXP3)+ T-cell ratios. Another nanoparticle formulation, namely “Chimeric Polypeptide Doxorubicin” (CP-Dox) efficiently increased the infiltration of leukocytes, slowed tumor growth and prevented metastasis in poorly immunogenic mouse 4T1 mammary carcinoma cells. The immune-mediated activity of CP-Dox depends here on CD8+ T-cells and the release of IFNγ.
Moreover, intra-tumor myeloid cells were repolarized towards an anti-cancer phenotype. The authors underline that anti-cancer immunotherapies present improved potential if used as with nanoparticle-packaged chemotherapy combinations (Mastria et al. 2018). Extracellular vesicles are naturally-occurring cargo delivery entities useful as drug vehicles. Garofalo et al. investigated human lung cancer cell-derived EVs for the systemic delivery of oncolytic virus combined to paclitaxel, leading to enhanced anti-tumor effects in nude mice. The authors demonstrated the ability of such vesicles to induce tumor-associated inflammatory reactions, which were characterized by ICD and CD3+/CD4+/CD8+ T-cell infiltration (Garofalo et al. 2019).
Metronomic immunogenic cell death induction
An exciting field of research tries to understand better how the dose and timing of a conventional cytotoxic agent eventually impact the anti-tumor immune responses. Wu and Waxman (2018) proposed a novel strategy to improve effective immunogenic chemotherapy that relies on a “modified metronomic schedule” for drug delivery called “medium-dose intermittent chemotherapy (MEDIC).” The authors observed positive responses when an immunogenic chemotherapeutic agent was administered “intermittently and at an intermediate dose”. The aim of the authors was to damage cancer cells by repeated cytotoxic damage to tumors combined to a schedule potentially compatible with activation of anti-tumor immune responses (Wu and Waxman 2018). Similarly, Banerjee et al. used a metronomic treatment approach based on a paclitaxel formulation of solid lipid nanoparticles that triggers ICD, modified with Tyr-3-octreotide (PSM), an octapeptide that mimics natural somatostatin. They used this therapeutic approach to treat melanoma that expresses elevated levels of somatostatin receptors. Altogether, metronomic PSM exhibits anti-melanoma activities without adverse toxicity. The authors conclude that this metronomic immune modulation via PSM could also be used for other types of cancer besides melanoma (Banerjee et al. 2019).
Combination treatments
As mentioned in this review, ICD induction can be achieved by single agents but is more efficient as a combination treatment. Accordingly, recent literature provides multiple examples of co-treatments involving ICD-inducing natural products. As previously mentioned, DC vaccine preparations based on tumor cells pretreated with chloroquine and shikonin further enhanced the anti-metastatic activity of 4T1 mouse breast cancer. This combination strategy allowed a reduction of the effective doxorubicin concentration. The authors concluded that the improved immunogenicity due to the shikonin and chloroquine cotreatment would allow the development of future cancer vaccines by combinational drug treatment (Lin et al. 2018).
Another combination therapy was also shown to be efficient. Oncolytic peptide LTX-315 possesses potent immunomodulatory properties triggers ICD with the release of DAMPs and leads to “reshaping” of the tumor microenvironment. Indeed, the authors showed that this compound could turn “cold” tumors into “hot” ones by increasing tumor-infiltrating lymphocytes. LTX-315 combined to Dox in the case of triple-negative breast cancer leading to a robust combinatory effect (Camilio et al. 2019). To improve the capacity of anthracycline-induced ICD induction, ZnCl2 was described to enhance the therapeutic effects of antineoplastic agents by promoting CALR exposure leading to improved anti-cancer effects even in TP53-deficient chemoresistant glioblastoma (Cirone et al. 2013).
Future orientation and conclusions
Compounds like mitoxantrone trigger ICD and anti-cancer immune responses. Fan et al. attempted to use the concept of ICD induction to convert it into a biotechnologically-useful tool. They suggested turning “immunogenically dying tumor cells into a powerful platform for cancer vaccination” in mouse melanoma and colon carcinoma. They modified cancer cells with adjuvant-loaded nanoparticles that target and deliver the drug triggering ICD. The resulting dying cells were carrying these nanoparticles trigger antigen-specific CD8α+ T-cells in vivo. This approach could be combined with immune checkpoint blockade to achieve consistent tumor regression. According to the authors, this approach “open new doors to personalized cancer immunotherapy” (Fan et al. 2017).
To date, only a small quantity of anti-cancer chemicals or natural compounds were discovered to induce ICD. Therefore, in the future, it will be important to identify novel compounds with the potential to induce ICD. Here it will become essential to explore the potential of natural compounds to contribute to ICD induction whether alone or in combination. Altogether, many phytochemicals were already described to act as “cellular stressors” triggering various modes of cell death. Research needs to focus on these phytochemicals and investigate whether or not the cell death modality induced by these phytochemical stressors triggers the appearance of an ICD phenotype beyond looking for pure cytotoxic agents (Mazumder et al. 2018b; Lee et al. 2018; Ji et al. 2018). We believe that immune modulation by phytochemical will constitute a most promising innovative avenue of natural compound drug discovery, alone or in combination with checkpoint inhibitors.
References
Adkins I, Fucikova J, Garg AD, Agostinis P, Spisek R (2014) Physical modalities inducing immunogenic tumor cell death for cancer immunotherapy. Oncoimmunology 3:e968434
Arcamone FM (2009) Fifty years of chemical research at Farmitalia. Chemistry (Easton) 15:7774–7791
Banerjee I, De M, Dey G, Bharti R, Chattopadhyay S, Ali N, Chakrabarti P, Reis RL, Kundu SC, Mandal M (2019) A peptide-modified solid lipid nanoparticle formulation of paclitaxel modulates immunity and outperforms dacarbazine in a murine melanoma model. Biomater Sci 7:1161–1178
Bezu L, Sauvat A, Humeau J, Gomes-Da-Silva LC, Iribarren K, Forveille S, Garcia P, Zhao L, Liu P, Zitvogel L, Senovilla L, Kepp O, Kroemer G (2018) eIF2alpha phosphorylation is pathognomonic for immunogenic cell death. Cell Death Differ 25:1375–1393
Byun JY, Kim MJ, Eum DY, Yoon CH, Seo WD, Park KH, Hyun JW, Lee YS, Lee JS, Yoon MY, Lee SJ (2009) Reactive oxygen species-dependent activation of Bax and poly(ADP-ribose) polymerase-1 is required for mitochondrial cell death induced by triterpenoid pristimerin in human cervical cancer cells. Mol Pharmacol 76:734–744
Camilio KA, Wang MY, Mauseth B, Waagene S, Kvalheim G, Rekdal O, Sveinbjornsson B, Maelandsmo GM (2019) Combining the oncolytic peptide LTX-315 with doxorubicin demonstrates therapeutic potential in a triple-negative breast cancer model. Breast Cancer Res 21:9
Casares N, Pequignot MO, Tesniere A, Ghiringhelli F, Roux S, Chaput N, Schmitt E, Hamai A, Hervas-Stubbs S, Obeid M, Coutant F, Metivier D, Pichard E, Aucouturier P, Pierron G, Garrido C, Zitvogel L, Kroemer G (2005) Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J Exp Med 202:1691–1701
Castoldi F, Vacchelli E, Zitvogel L, Maiuri MC, Pietrocola F, Kroemer G (2019) Systemic autophagy in the therapeutic response to anthracycline-based chemotherapy. Oncoimmunology 8:e1498285
Cerella C, Teiten MH, Radogna F, Dicato M, Diederich M (2014) From nature to bedside: pro-survival and cell death mechanisms as therapeutic targets in cancer treatment. Biotechnol Adv 32:1111–1122
Chen X, Yang L, Zhang N, Turpin JA, Buckheit RW, Osterling C, Oppenheim JJ, Howard OM (2003) Shikonin, a component of chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1. Antimicrob Agents Chemother 47:2810–2816
Chen HM, Wang PH, Chen SS, Wen CC, Chen YH, Yang WC, Yang NS (2012) Shikonin induces immunogenic cell death in tumor cells and enhances dendritic cell-based cancer vaccine. Cancer Immunol Immunother 61:1989–2002
Cirone M, Garufi A, Di Renzo L, Granato M, Faggioni A, D’orazi G (2013) Zinc supplementation is required for the cytotoxic and immunogenic effects of chemotherapy in chemoresistant p53-functionally deficient cells. Oncoimmunology 2:e26198
Colangelo T, Polcaro G, Ziccardi P, Muccillo L, Galgani M, Pucci B, Milone MR, Budillon A, Santopaolo M, Mazzoccoli G, Matarese G, Sabatino L, Colantuoni V (2016) The miR-27a-calreticulin axis affects drug-induced immunogenic cell death in human colorectal cancer cells. Cell Death Dis 7:e2108
Cragg GM, Pezzuto JM (2016) Natural products as a vital source for the discovery of cancer chemotherapeutic and chemopreventive agents. Med Princ Pract 25(Suppl 2):41–59
Davids LM, Kleemann B, Kacerovska D, Pizinger K, Kidson SH (2008) Hypericin phototoxicity induces different modes of cell death in melanoma and human skin cells. J Photochem Photobiol B 91:67–76
D’eliseo D, Manzi L, Velotti F (2013) Capsaicin as an inducer of damage-associated molecular patterns (DAMPs) of immunogenic cell death (ICD) in human bladder cancer cells. Cell Stress Chaperones 18:801–808
D’eliseo D, Di Renzo L, Santoni A, F Velotti (2017) Docosahexaenoic acid (DHA) promotes immunogenic apoptosis in human multiple myeloma cells, induces autophagy and inhibits STAT3 in both tumor and dendritic cells. Genes Cancer 8:426–437
Diederich M, Cerella C (2016) Non-canonical programmed cell death mechanisms triggered by natural compounds. Semin Cancer Biol 40–41:4–34
Diederich M, Muller F, Cerella C (2017) Cardiac glycosides: from molecular targets to immunogenic cell death. Biochem Pharmacol 125:1–11
Fan Y, Kuai R, Xu Y, Ochyl LJ, Irvine DJ, Moon JJ (2017) Immunogenic cell death amplified by co-localized adjuvant delivery for cancer immunotherapy. Nano Lett 17:7387–7393
Fredly H, Ersvaer E, Gjertsen BT, Bruserud O (2011) Immunogenic apoptosis in human acute myeloid leukemia (AML): primary human AML cells expose calreticulin and release heat shock protein (HSP) 70 and HSP90 during apoptosis. Oncol Rep 25:1549–1556
Galluzzi L, Buque A, Kepp O, Zitvogel L, Kroemer G (2017) Immunogenic cell death in cancer and infectious disease. Nat Rev Immunol 17:97–111
Garg AD, Krysko DV, Vandenabeele P, Agostinis P (2012) Hypericin-based photodynamic therapy induces surface exposure of damage-associated molecular patterns like HSP70 and calreticulin. Cancer Immunol Immunother 61:215–221
Garg AD, Dudek AM, Agostinis P (2013a) Cancer immunogenicity, danger signals, and DAMPs: what, when, and how? BioFactors 39:355–367
Garg AD, Dudek AM, Ferreira GB, Verfaillie T, Vandenabeele P, Krysko DV, Mathieu C, Agostinis P (2013b) ROS-induced autophagy in cancer cells assists in evasion from determinants of immunogenic cell death. Autophagy 9:1292–1307
Garg AD, Elsen S, Krysko DV, Vandenabeele P, De Witte P, Agostinis P (2015) Resistance to anticancer vaccination effect is controlled by a cancer cell-autonomous phenotype that disrupts immunogenic phagocytic removal. Oncotarget 6:26841–26860
Garg AD, Vandenberk L, Koks C, Verschuere T, Boon L, Van Gool SW, Agostinis P (2016) Dendritic cell vaccines based on immunogenic cell death elicit danger signals and T cell-driven rejection of high-grade glioma. Sci Transl Med 8:328ra327
Garofalo M, Villa A, Rizzi N, Kuryk L, Rinner B, Cerullo V, Yliperttula M, Mazzaferro V, Ciana P (2019) Extracellular vesicles enhance the targeted delivery of immunogenic oncolytic adenovirus and paclitaxel in immunocompetent mice. J Control Release 294:165–175
Giglio P, Gagliardi M, Tumino N, Antunes F, Smaili S, Cotella D, Santoro C, Bernardini R, Mattei M, Piacentini M, Corazzari M (2018) PKR and GCN2 stress kinases promote an ER stress-independent eIF2alpha phosphorylation responsible for calreticulin exposure in melanoma cells. Oncoimmunology 7:e1466765
Graham W, Roberts JB (1953) Intravenous colchicine in the management of gouty arthritis. Ann Rheum Dis 12:16–19
Hirata Y, Uemura D (1986) Halichondrins—antitumor polyether macrolides from a marine sponge. Pure Appl Chem 58(5):701
Huang FY, Lei J, Sun Y, Yan F, Chen B, Zhang L, Lu Z, Cao R, Lin YY, Wang CC, Tan GH (2018) Induction of enhanced immunogenic cell death through ultrasound-controlled release of doxorubicin by liposome-microbubble complexes. Oncoimmunology 7:e1446720
Inoue S, Setoyama Y, Odaka A (2014) Doxorubicin treatment induces tumor cell death followed by immunomodulation in a murine neuroblastoma model. Exp Ther Med 7:703–708
Ji S, Lee JY, Schror J, Mazumder A, Jang DM, Chateauvieux S, Schnekenburger M, Hong CR, Christov C, Kang HJ, Lee Y, Han BW, Kim KW, Shin HY, Dicato M, Cerella C, Konig GM, Orlikova B, Diederich M (2018) The dialkyl resorcinol stemphol disrupts calcium homeostasis to trigger programmed immunogenic necrosis in cancer. Cancer Lett 416:109–123
Jin T, Wu H, Wang Y, Peng H (2016) Capsaicin induces immunogenic cell death in human osteosarcoma cells. Exp Ther Med 12:765–770
Johansson MP, Maaheimo H, Ekholm FS (2017) New insight on the structural features of the cytotoxic auristatins MMAE and MMAF revealed by combined NMR spectroscopy and quantum chemical modelling. Sci Rep 7:15920
Kubin A, Wierrani F, Burner U, Alth G, Grunberger W (2005) Hypericin–the facts about a controversial agent. Curr Pharm Des 11:233–253
Kumar SS (2017) Maytenus ovatus (schweinf.) An African medicinal plant yielding potential anti-cancer drugs. Biomed J Sci Tech Res. https://doi.org/10.26717/BJSTR.2017.01.000571
Kupchan SM, Komoda Y, Court WA, Thomas GJ, Smith RM, Karim A, Gilmore CJ, Haltiwanger RC, Bryan RF (1972) Maytansine, a novel antileukemic ansa macrolide from Maytenus ovatus. J Am Chem Soc 94:1354–1356
Ladoire S, Penault-Llorca F, Senovilla L, Dalban C, Enot D, Locher C, Prada N, Poirier-Colame V, Chaba K, Arnould L, Ghiringhelli F, Fumoleau P, Spielmann M, Delaloge S, Poillot ML, Arveux P, Goubar A, Andre F, Zitvogel L, Kroemer G (2015) Combined evaluation of LC3B puncta and HMGB1 expression predicts residual risk of relapse after adjuvant chemotherapy in breast cancer. Autophagy 11:1878–1890
Lee JY, Talhi O, Jang D, Cerella C, Gaigneaux A, Kim KW, Lee JW, Dicato M, Bachari K, Han BW, Silva AMS, Orlikova B, Diederich M (2018) Cytostatic hydroxycoumarin OT52 induces ER/Golgi stress and STAT3 inhibition triggering non-canonical cell death and synergy with BH3 mimetics in lung cancer. Cancer Lett 416:94–108
Li TF, Li K, Zhang Q, Wang C, Yue Y, Chen Z, Yuan SJ, Liu X, Wen Y, Han M, Komatsu N, Xu YH, Zhao L, Chen X (2018) Dendritic cell-mediated delivery of doxorubicin-polyglycerol-nanodiamond composites elicits enhanced anti-cancer immune response in glioblastoma. Biomaterials 181:35–52
Lin TJ, Liang WM, Hsiao PW, SP M, Wei WC, Lin HT, Yin SY, Yang NS (2015) Rapamycin promotes mouse 4T1 tumor metastasis that can be reversed by a dendritic cell-based vaccine. PLoS ONE 10:e0138335
Lin SY, Hsieh SY, Fan YT, Wei WC, Hsiao PW, Tsai DH, Wu TS, Yang NS (2018) Necroptosis promotes autophagy-dependent upregulation of DAMP and results in immunosurveillance. Autophagy 14:778–795
Liu P, Zhao L, Loos F, Iribarren K, Lachkar S, Zhou H, Gomes-Da-Silva LC, Chen G, Bezu L, Boncompain G, Perez F, Zitvogel L, Kepp O, Kroemer G (2017) Identification of pharmacological agents that induce HMGB1 release. Sci Rep 7:14915
Liu Q, Chen F, Hou L, Shen L, Zhang X, Wang D, Huang L (2018a) Nanocarrier-mediated chemo-immunotherapy arrested cancer progression and induced tumor dormancy in desmoplastic melanoma. ACS Nano 12:7812–7825
Liu Q, Zhu H, Tiruthani K, Shen L, Chen F, Gao K, Zhang X, Hou L, Wang D, Liu R, Huang L (2018b) Nanoparticle-mediated trapping of Wnt family member 5A in tumor microenvironments enhances immunotherapy for B-Raf proto-oncogene mutant melanoma. ACS Nano 12:1250–1261
Loi S, Pommey S, Haibe-Kains B, Beavis PA, Darcy PK, Smyth MJ, Stagg J (2013) CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc Natl Acad Sci USA 110:11091–11096
Lu J, Liu X, Liao YP, Wang X, Ahmed A, Jiang W, Ji Y, Meng H, Nel AE (2018) Breast cancer chemo-immunotherapy through liposomal delivery of an immunogenic cell death stimulus plus interference in the IDO-1 pathway. ACS Nano 12:11041–11061
Mastria EM, Cai LY, Kan MJ, Li X, Schaal JL, Fiering S, Gunn MD, Dewhirst MW, Nair SK, Chilkoti A (2018) Nanoparticle formulation improves doxorubicin efficacy by enhancing host antitumor immunity. J Control Release 269:364–373
Matzinger P (1994) Tolerance, danger, and the extended family. Annu Rev Immunol 12:991–1045
Mazumder A, Cerella C, Diederich M (2018a) Natural scaffolds in anticancer therapy and precision medicine. Biotechnol Adv 36:1563–1585
Mazumder A, Lee JY, Talhi O, Cerella C, Chateauvieux S, Gaigneaux A, Hong CR, Kang HJ, Lee Y, Kim KW, Kim DW, Shin HY, Dicato M, Bachari K, Silva AMS, Orlikova-Boyer B, Diederich M (2018b) Hydroxycoumarin OT-55 kills CML cells alone or in synergy with imatinib or Synribo: involvement of ER stress and DAMP release. Cancer Lett 438:197–218
Menger L, Vacchelli E, Adjemian S, Martins I, Ma Y, Shen S, Yamazaki T, Sukkurwala AQ, Michaud M, Mignot G, Schlemmer F, Sulpice E, Locher C, Gidrol X, Ghiringhelli F, Modjtahedi N, Galluzzi L, Andre F, Zitvogel L, Kepp O, Kroemer G (2012) Cardiac glycosides exert anticancer effects by inducing immunogenic cell death. Sci Transl Med 4:143ra199
Messmer MN, Snyder AG, Oberst A (2019) Comparing the effects of different cell death programs in tumor progression and immunotherapy. Cell Death Differ 26:115–129
Michaud M, Martins I, Sukkurwala AQ, Adjemian S, Ma Y, Pellegatti P, Shen S, Kepp O, Scoazec M, Mignot G, Rello-Varona S, Tailler M, Menger L, Vacchelli E, Galluzzi L, Ghiringhelli F, Di Virgilio F, Zitvogel L, Kroemer G (2011) Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 334:1573–1577
Michaud M, Xie X, Bravo-San Pedro JM, Zitvogel L, White E, Kroemer G (2014) An autophagy-dependent anticancer immune response determines the efficacy of melanoma chemotherapy. Oncoimmunology 3:e944047
Molinari R, D’eliseo D, Manzi L, Zolla L, Velotti F, Merendino N (2011) The n3-polyunsaturated fatty acid docosahexaenoic acid induces immunogenic cell death in human cancer cell lines via pre-apoptotic calreticulin exposure. Cancer Immunol Immunother 60:1503–1507
Monk BJ, Brady MF, Aghajanian C, Lankes HA, Rizack T, Leach J, Fowler JM, Higgins R, Hanjani P, Morgan M, Edwards R, Bradley W, Kolevska T, Foukas P, Swisher EM, Anderson KS, Gottardo R, Bryan JK, Newkirk M, Manjarrez KL, Mannel RS, Hershberg RM, Coukos G (2017) A phase 2, randomized, double-blind, placebo- controlled study of chemo-immunotherapy combination using motolimod with pegylated liposomal doxorubicin in recurrent or persistent ovarian cancer: a Gynecologic Oncology Group partners study. Ann Oncol 28:996–1004
Morceau F, Chateauvieux S, Orsini M, Trecul A, Dicato M, Diederich M (2015) Natural compounds and pharmaceuticals reprogram leukemia cell differentiation pathways. Biotechnol Adv 33:785–797
Orlikova B, Diederich M (2012) Power from the garden: plant compounds as inhibitors of the hallmarks of cancer. Curr Med Chem 19:2061–2087
Orlikova B, Legrand N, Panning J, Dicato M, Diederich M (2014) Anti-inflammatory and anticancer drugs from nature. Cancer Treat Res 159:123–143
Papa MZ, Yang JC, Vetto JT, Shiloni E, Eisenthal A, Rosenberg SA (1988) Combined effects of chemotherapy and interleukin 2 in the therapy of mice with advanced pulmonary tumors. Cancer Res 48:122–129
Pellicciotta I, Yang CP, Goldberg GL, Shahabi S (2011) Epothilone B enhances Class I HLA and HLA-A2 surface molecule expression in ovarian cancer cells. Gynecol Oncol 122:625–631
Qin J, Kunda NM, Qiao G, Tulla K, Prabhakar BS, Maker AV (2019) Vaccination with mitoxantrone-treated primary colon cancer cells enhances tumor-infiltrating lymphocytes and clinical responses in colorectal liver metastases. J Surg Res 233:57–64
Radogna F, Diederich M (2018) Stress-induced cellular responses in immunogenic cell death: implications for cancer immunotherapy. Biochem Pharmacol 153:12–23
Radogna F, Cerella C, Gaigneaux A, Christov C, Dicato M, Diederich M (2016) Cell type-dependent ROS and mitophagy response leads to apoptosis or necroptosis in neuroblastoma. Oncogene 35:3839–3853
Radogna F, Dicato M, Diederich M (2019) Natural modulators of the hallmarks of immunogenic cell death. Biochem Pharmacol 162:55–70
Ramanathapuram LV, Hahn T, Dial SM, Akporiaye ET (2005) Chemo-immunotherapy of breast cancer using vesiculated alpha-tocopheryl succinate in combination with dendritic cell vaccination. Nutr Cancer 53:177–193
Riganti C, Castella B, Kopecka J, Campia I, Coscia M, Pescarmona G, Bosia A, Ghigo D, Massaia M (2013) Zoledronic acid restores doxorubicin chemosensitivity and immunogenic cell death in multidrug-resistant human cancer cells. PLoS ONE 8:e60975
Rios-Doria J, Durham N, Wetzel L, Rothstein R, Chesebrough J, Holoweckyj N, Zhao W, Leow CC, Hollingsworth R (2015) Doxil synergizes with cancer immunotherapies to enhance antitumor responses in syngeneic mouse models. Neoplasia 17:661–670
Romano E, Rufo N, Korf H, Mathieu C, Garg AD, Agostinis P (2018) BNIP3 modulates the interface between B16-F10 melanoma cells and immune cells. Oncotarget 9:17631–17644
Schneider NFZ, Cerella C, Simoes CMO, Diederich M (2017) Anticancer and immunogenic properties of cardiac glycosides. Molecules 22:1932
Schnekenburger M, Dicato M, Diederich M (2014) Plant-derived epigenetic modulators for cancer treatment and prevention. Biotechnol Adv 32:1123–1132
Schnekenburger M, Dicato M, Diederich MF (2019) Anticancer potential of naturally occurring immunoepigenetic modulators: a promising avenue? Cancer. https://doi.org/10.1002/cncr.32041
Schumacher M, Kelkel M, Dicato M, Diederich M (2011) Gold from the sea: marine compounds as inhibitors of the hallmarks of cancer. Biotechnol Adv 29:531–547
Senovilla L, Vitale I, Martins I, Tailler M, Pailleret C, Michaud M, Galluzzi L, Adjemian S, Kepp O, Niso-Santano M, Shen S, Marino G, Criollo A, Boileve A, Job B, Ladoire S, Ghiringhelli F, Sistigu A, Yamazaki T, Rello-Varona S, Locher C, Poirier-Colame V, Talbot M, Valent A, Berardinelli F, Antoccia A, Ciccosanti F, Fimia GM, Piacentini M, Fueyo A, Messina NL, Li M, Chan CJ, Sigl V, Pourcher G, Ruckenstuhl C, Carmona-Gutierrez D, Lazar V, Penninger JM, Madeo F, Lopez-Otin C, Smyth MJ, Zitvogel L, Castedo M, Kroemer G (2012) An immunosurveillance mechanism controls cancer cell ploidy. Science 337:1678–1684
Slingerland M, Cerella C, Guchelaar HJ, Diederich M, Gelderblom H (2013) Cardiac glycosides in cancer therapy: from preclinical investigations towards clinical trials. Invest New Drugs 31:1087–1094
Steinmetz MO, Prota AE (2018) Microtubule-targeting agents: strategies to hijack the cytoskeleton. Trends Cell Biol 28:776–792
Sukkurwala AQ, Adjemian S, Senovilla L, Michaud M, Spaggiari S, Vacchelli E, Baracco EE, Galluzzi L, Zitvogel L, Kepp O, Kroemer G (2014a) Screening of novel immunogenic cell death inducers within the NCI mechanistic diversity set. Oncoimmunology 3:e28473
Sukkurwala AQ, Martins I, Wang Y, Schlemmer F, Ruckenstuhl C, Durchschlag M, Michaud M, Senovilla L, Sistigu A, Ma Y, Vacchelli E, Sulpice E, Gidrol X, Zitvogel L, Madeo F, Galluzzi L, Kepp O, Kroemer G (2014b) Immunogenic calreticulin exposure occurs through a phylogenetically conserved stress pathway involving the chemokine CXCL8. Cell Death Differ 21:59–68
Tang W, Guo Z, Cao Z, Wang M, Li P, Meng X, Zhao X, Xie Z, Wang W, Zhou A, Lou C, Chen Y (2018) d-Sedoheptulose-7-phosphate is a common precursor for the heptoses of septacidin and hygromycin B. Proc Natl Acad Sci USA 115:2818–2823
Tang X, Rao J, Yin S, Wei J, Xia C, Li M, Mei L, Zhang Z, He Q (2019) PD-L1 knockdown via hybrid micelle promotes paclitaxel induced cancer-immunity cycle for melanoma treatment. Eur J Pharm Sci 127:161–174
Thornburg CC, Britt JR, Evans JR, Akee RK, Whitt JA, Trinh SK, Harris MJ, Thompson JR, Ewing TL, Shipley SM, Grothaus PG, Newman DJ, Schneider JP, Grkovic T, O’keefe BR (2018) NCI program for natural product discovery: a publicly-accessible library of natural product fractions for high-throughput screening. ACS Chem Biol 13:2484–2497
Van Der Heijden R, Jacobs DI, Snoeijer W, Hallard D, Verpoorte R (2004) The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr Med Chem 11:607–628
Verfaillie T, Van Vliet A, Garg AD, Dewaele M, Rubio N, Gupta S, De Witte P, Samali A, Agostinis P (2013) Pro-apoptotic signaling induced by photo-oxidative ER stress is amplified by Noxa, not Bim. Biochem Biophys Res Commun 438:500–506
Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW (2013) Cancer genome landscapes. Science 339:1546–1558
Weaver BA (2014) How Taxol/paclitaxel kills cancer cells. Mol Biol Cell 25:2677–2681
Weber T, Dalen H, Andera L, Negre-Salvayre A, Auge N, Sticha M, Lloret A, Terman A, Witting PK, Higuchi M, Plasilova M, Zivny J, Gellert N, Weber C, Neuzil J (2003) Mitochondria play a central role in apoptosis induced by alpha-tocopheryl succinate, an agent with antineoplastic activity: comparison with receptor-mediated pro-apoptotic signaling. Biochemistry 42:4277–4291
Wen CC, Chen HM, Chen SS, Huang LT, Chang WT, Wei WC, Chou LC, Arulselvan P, Wu JB, Kuo SC, Yang NS (2011) Specific microtubule-depolymerizing agents augment efficacy of dendritic cell-based cancer vaccines. J Biomed Sci 18:44
Wu J, Waxman DJ (2018) Immunogenic chemotherapy: dose and schedule dependence and combination with immunotherapy. Cancer Lett 419:210–221
Xu Q, Chen C, Lin A, Xie Y (2017) Endoplasmic reticulum stress-mediated membrane expression of CRT/ERp57 induces immunogenic apoptosis in drug-resistant endometrial cancer cells. Oncotarget 8:58754–58764
Yang Y, Li XJ, Chen Z, Zhu XX, Wang J, Zhang LB, Qiang L, Ma YJ, Li ZY, Guo QL, You QD (2012) Wogonin induced calreticulin/annexin A1 exposure dictates the immunogenicity of cancer cells in a PERK/AKT dependent manner. PLoS ONE 7:e50811
Yin SY, Efferth T, Jian FY, Chen YH, Liu CI, Wang AH, Chen YR, Hsiao PW, Yang NS (2016) Immunogenicity of mammary tumor cells can be induced by shikonin via direct binding-interference with hnRNPA1. Oncotarget 7:43629–43653
Zitvogel L, Rusakiewicz S, Routy B, Ayyoub M, Kroemer G (2016) Immunological off-target effects of imatinib. Nat Rev Clin Oncol 13:431–446
Acknowledgements
The author thanks Dr. Flavia Radogna for proofreading and helpful comments. MD’s research at SNU is supported by National Research Foundation (NRF) [Grant Number 019R1A2C1009231] and by a grant from the MEST of Korea for Tumor Microenvironment Global Core Research Center (GCRC) [Grant Number 2011-0030001]. Support from Brain Korea (BK21) PLUS program and Creative-Pioneering Researchers Program at Seoul National University [Funding number: 370C-20160062] are acknowledged. MD also thanks the “Recherche Cancer et Sang” foundation, “Recherches Scientifiques Luxembourg” association, “Een Häerz fir kriibskrank Kanner” association, Action LIONS “Vaincre le Cancer” association and Télévie Luxembourg.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Diederich, M. Natural compound inducers of immunogenic cell death. Arch. Pharm. Res. 42, 629–645 (2019). https://doi.org/10.1007/s12272-019-01150-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-019-01150-z