Abstract
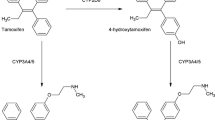
The aim of this study was to investigate the effect of naringin on the bioavailability and pharmacokinetics of tamoxifen and of its metabolite, 4-hydroxytamoxifen in rats. The pharmacokinetic parameters of tamoxifen and 4-hydroxytamoxifen were determined by HPLC after pretreating with naringin (1.5, 7.5, and 15 mg/kg) 30 min before orally administering tamoxifen (10 mg/kg). Compared with the control group (treated with tamoxifen alone), naringin pretreated animals showed significantly (p<0.01) increased areas under the plasma concentration-time curves (AUC) and peak tamoxifen concentrations (Cmax). The absolute bioavailabilities (AB%) of tamoxifen in naringin pretreated animals were enhanced versus control (from 32.8% to 47.1%), and the relative bioavailabilities (RB%) of tamoxifen in the naringin pretreated groups were 2.02–2.88 times higher than that in the control. No significant changes in the terminal half-life (t1/2) or Tmax of tamoxifen were observed in the naringin pretreated groups. The AUCs of 4-hydroxytamoxifen after pretreating naringin were also significantly elevated (p<0.05) versus the control. But metabolite ratios (MR; AUC of 4-hydroxytamoxifen to tamoxifen) were significantly lower. These results suggest that the enhanced bioavailability of tamoxifen in the presence of naringin might be due to the inhibition of CYP3A4 by naringin. If the results of this study are further confirmed by clinical trials, tamoxifen dosages should be adjusted to avoid potential drug interaction when tamoxifen is used clinically in combination with naringin-containing dietary supplements.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bailey, D. G.., Arnold, J. M. O., and Spence, J. D., Grapefruit juice-drug interactions. Br. J. Clin. Pharmacol., 46, 101–110 (1998).
Buchanan, C. M., Buchanan, N. L., Edgar, K. J., Little, J. L., Malcolm, M. O., Ruble, K. M., Wacher, V. J., and Wempe, M. F., Pharmacokinetics of tamoxifen after intravenous and oral dosing of tamoxifen-hydroxybutenyl-beta-cyclodextrin formulations. J. Pharm. Sci., in press (2006).
Buckley, M. T. and Goa, K. L., Tamoxifen: a reappraisal of its pharmacodynamic and pharmacokinetic properties, and therapeutic use. Clin. Pharmacokin., 37, 451–490 (1989).
Crewe, H. K., Ellis, S. W., Lennard, M. S., and Tucker, G. T., Variable contribution of cytochromes P450 2D6, 2C9 and 3A4 to the 4-hydroxylation of tamoxifen by human liver microsomes. Biochem. Pharmacol., 53, 171–178 (1997).
Daniel, P. C., Gaskell, J., Bishop, H., Campbell, C., and Nicholson, R., Determination of tamoxifen and biologically active metabolites in human breast tumour and plasma. Eur. J. Cancer Clin. Oncol., 17, 1183–1189 (1981).
Dixon, R. A. and Steele, C., Flavonoids and isoflavonoids-gold mine for metabolic engineering. Trends Plant Sci., 4, 394–400 (1999).
Eagling, V. A., Profit, L., and Back, D. J., Inhibition of the CYP3A4-mediated metabolism and P-glycoprotein-mediated transport of the HIV-1 protease inhibitor saquinavir by grapefruit juice components. Br. J. Clin. Pharmacol., 48, 543–552 (1999).
Fornander, T., Hellstrom, A. C., and Moberger, B., Descriptive clinicopathologic study of 17 patients with endometrial cancer during or after adjuvant tamoxifen in early breast cancer. J. Nat. Cancer Inst., 85, 1850–1855 (1993).
Fried, K. M. and Wainer, I. W., Direct determination of tamoxifen and its four major metabolites in plasma using coupled column high-performance liquid chromatography. J. Chromatogr. B. Biomed. Appl., 655, 261–268 (1994).
Gant, T. W., O’Connor, C. K., Corbitt, R., Thorgeirsson, U., and Thorgeirsson, S. S., In vivo induction of liver P-glycoprotein expression by xenobiotics in monkeys. Toxicol. Appl. Pharmacol., 133, 269–276 (1995).
Gottesman, M. M. and Pastan, I., Biochemistry of multidrug resistance mediated by the multidrug transporter. Annu. Rev. Biochem., 62, 385–427 (1993).
Ho, P. C., Saville, D. J., Coville, P. C., and Wanwimolruk, S., Content of CYP3A4 inhibitors, naringin, naringenin and bergapten in grapefruit and grapefruit juice products. Pharmaceut. Acta. Helv., 74, 379–385 (2000).
Ho, P. C., Saville, D. J., and Wanwimolruk, S., Inhibition of human CYP3A4 activity by grapefruit flavonoids, furanocoumarins and related compounds. J. Pharm. Pharm. Sci., 4, 217–227 (2001).
Jacolot, F., Simon, I., Dreano, I., Beaune, P., Riche, C., and Berthou, F., Identification of the cytochrome P450 IIIA family as the enzymes involved in the N-demethylation of tamoxifen in human liver microsomes. Biochem. Pharmacol., 41, 1911–1919 (1991).
Jaiyesimi, I. A., Buzdar, A. U., Decker, D. A., and Hortobagyi, G. N., Use of tamoxifen for breast cancer: twenty-eight years later. J. Clin. Oncol., 13, 513–529 (1995).
Jordan, V. C., Collins, M. M., Rowsby, L., and Prestwich, G., A monohydroxylated metabolite of tamoxifen with potent antioestrogenic activity. J. Endocrinol., 75, 305–316 (1977).
Kim, D. H., Jung, E. A., Sohng, I. S., Han, J. A., Kim, T. H., and Han, M. J., Intestinal bacterial metabolism of flavonoids and its relation to some biological activities, Arch. Pharm. Res., 21, 17–23 (1998).
Mani, C., Gelboin, H. V., Park, S. S., Pearce, R., Parkinson, A., and Kupfer, D., Metabolism of the antimammary cancer antiestrogenic agent tamoxifen, I. Cytochrome P450-catalysed N-demethylation and 4-hydroxylation. Drug Metab. Dispos., 21, 645–656 (1993).
Meier, C. R. and Jick, H., Tamoxifen and risk of idiopathic venous thromboembolism. Br. J. Clin. Pharmacol., 45, 608–612 (1998).
Nijveldt, R. J., van Nood, E., van Hoorn, D. E. C., Boelens, P. G., van Norren, K., and van Leeuwen, P. A. M., Flavonoids: a review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr., 74, 418–425 (2001).
Powles, T. J., The case for clinical trials of tamoxifen for prevention of breast cancer. Lancet, 340, 1145–1147 (1992).
Rao, U. S., Fine, R. L., and Scarborough, G. A., Antiestrogens and steroid hormones: substrates of the human P-glycoprotein. Biochem. Pharmacol., 48, 287–292 (1994).
Shin, S. C., Choi, J. S., and Li, X., Enhanced bioavailability of tamoxifen after oral administration of tamoxifen with quercetin in rats. Int. J. Pharm., 313, 144–149 (2006).
Stone, R., NIH fends off critics of tamoxifen study. Science, 258, 734 (1992).
Takanaga, H., Ohnishi, A., Matsuo, H., and Sawada, Y., Inhibition of vinblastine efflux mediated by P-glycoprotein by grapefruit juice components in caco-2 cells. Biol. Pharm. Bull., 21, 1062–1066 (1998).
Thiebaut, F., Tsuruo, T., Hamada, H., Cottesman, M. M., Pastan, I., and Willingham, M. C., Cellular localization of the multidrug-resistance gene product P-glycoprotein in normal human tissues. Proc. Natl. Acad. Sci. U.S.A., 84, 7735–7738 (1987).
Wacher, V. H., Silverman, J. A., Zhang, Y., and Benet, L. Z., Role of P-glycoprotein and cytochrome P450 3A in limiting oral absorption of peptides and peptidomimetics. J. Pharm. Sci., 87, 1322–1330 (1998).
Watkins, P. B., The barrier function of CYP3A4 and P-glycoprotein in the small bowel. Adv. Drug Deliv. Rev., 27, 161–170 (1996).
Yeum, C. H. and Choi, J. S., Effect of naringin pretreatment on bioavailability of verapamil in rabbits. Arch. Pharm. Res., 29, 102–107 (2006).
Zhang, H., Wong, C. W., Coville, P. G., and Wanwimolruk, S., Effect of the grapefruit flavonoid naringin on pharmacokinetics of quinine in rats. Drug Metabol. Drug Interact., 17, 351–363 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, JS., Kang, K.W. Enhanced tamoxifen bioavailability after oral administration of tamoxifen in rats pretreated with naringin. Arch. Pharm. Res. 31, 1631–1636 (2008). https://doi.org/10.1007/s12272-001-2161-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-2161-7