Abstract
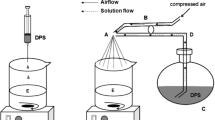
The aim of this study was the preparation of microparticles containing rifampicin using a biodegradable polymer poly(3-hydroxybutyrate-co-3-hydroxyvalerate) for oral administration produced by a bacteria. The poly(3-hydroxybutyrate-co-3-hydroxyvalerate) microparticles with and without rifampicin were prepared by the emulsification and solvent evaporation method, in which chloroform and polyvinyl alcohol are used as the solvent and emulsifier, respectively. Microparticles were obtained within a size range of 20-60 μm by changing the initial poly(3-hydroxybutyrate-co-3-hydroxyvalerate), polyvinyl alcohol and rifampicin concentrations. An encapsulation efficiency value of 14% was obtained. The optimized total yield of 60% of the poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/ rifampicin was obtained. A load of 0.035 mg/1 mg of PHBV was reached. Almost 90% of the drug loaded in the microparticles was released after 24 h. The size, encapsulation efficiency and ribampicin release of the microparticles varied as a function of the initial poly(3-hydroxybutyrate-co-3-hydroxyvalerate), polyvinyl alcohol and rifampicin concentrations. It was demonstrated that the microencapsulated rifampicin, although was not totally available in the medium, exhibited a similar inhibition value as free rifampicin at 24 h of incubation with S. aureus. Cytotoxicity assays demonstrated a reduction of the toxicity when rifampicin was microencapsulated in poly(3-hydroxybutyrate-co-3-hydroxyvalerate) while maintaining its antibacterial activity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Anisimova, Y. V., Gelperina, S. I., Peloquim, C. A., and Heifets, L. B., Nanoparticles as antituberculosis drugs carriers: effect on activity against Mycobacterium tuberculosis in human monocyte-derived macrophages., J. Nanoparticle. Res. 2, 165–171 (2000).
Bianchi, V. and Fortunati, E., Cellular effects of an anionic surfactant detected in V79 fibroblasts by different cytotoxicity tests. Toxicol In Vitro, 4, 9–16 (1990).
Borefreund, E. and Puerner, J. A., A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR 90). J. Tissue Cult. Methods, 9, 7–9 (1984).
Borin, A., Ferrão, M. F., Mello, C., Cordi. L., Pataca, L. C. M., Durán, N., and Poppi, R. J., Quantification of Lactobacillus in fermented milk by multivariate image analysis with least-squares support-vector machines. Anal. Bioanal. Chem., 387, 1105–1112 (2007).
Campbel, E. A., Korzheva, N., Mustaev, A., Murakami, K., Nair, S., Goldfarb, A., and Darst, S. A., Structural mechanism for rifampicin inhibition of bacterial RNA polymerase. Cell, 104, 901–912 (2001).
Coimbra, P. A., De Sousa, H. C., and Gil, M. H., Preparation and characterization of flurbiprofen-loaded poly(3-hydroxybutyrate-co-3-hydroxyvalerate) microspheres. J. Microencapsul., 25, 170–178 (2008).
Celikkaya, E., Denkbas, E. B., and Piskin, E., Rifampicin carrying poly (D,L-lactide)/poly(ethylene glycol) microspheres: loading and release. Artif. Organs, 20, 743–751 (1996).
Chen, J. and Davis, S. S., The release of diazepam from poly(hydroxybutyrate-co-hydroxyvalerate) microspheres. J Microencapsul, 19, 191–201 (2002).
Cingi, M. R., De Angelis, I., Fortunati, E., Reggiani, D., Bianchi, V., Tiozzo, R., and Zucco, F., Choice and standardization of test protocols in cytotoxicology: a multicentre approach. Toxicol In Vitro, 5, 119–125 (1991).
Coccoli, V., Luciani, A., Orsi, S., Guarino, V., Causa, F., and Netti, P. A., Engineering of poly(e-caprolactone) microcarriers to modulate protein encapsulation capability and release kinetic. J. Mater. Sci. Mater. Med. 19, 1703–1711 (2008).
Corrêa, D. H. A., Melo, P. S., Carvalho, C. A. A., de Azevedo, MBM, Durán, N., and Haun, M., Dehydrocrotonin and its β-cyclodextrin complex: Cytotoxicity in V79 fibroblasts and rat cultured hepatocytes. Eur. J. Pharmacol., 510, 17–24 (2005).
Dang, M. H., Birchler, F., and Wirtermantel, E., Toxicity screening of biodegradable polymers 2 Evaluation of cell culture test with medium extract. J. Environ. Polym. Degrad., 5, 49–56 (1997).
Denizot, F. and Lang, R., Rapid colorimetric assay for cell growth and survival modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Methods, 89, 271–277 (1986).
Durán, N., De Oliveira, A., and De Azevedo, M. M. M., In vitro studies on the release of isoniazid incorporated in poly (epsilon-caprolactone). J. Chemother., 18, 473–479 (2006).
Durán, N., Marcato, P. D., Buffo, C. M. S., De Azevedo, M. M. M., and Esposito, E. Poly(epsilon-caprolactone)/propolis extract: microencapsulation and antibacterial activity evaluation. Pharmazie, 62, 287–290 (2007).
Esmaeili, F., Hosseini-Nasr, M-H., Rad-Malekshahi, M., Samadi, N., Atyabi, F., and Dinarvand, R. Preparation and antibacterial activity evaluation of rifampicin-loaded poly (lactideco-glycolide) nanoparticles. Nanomedicine: NBM., 3, 161–167 (2007).
Fumori, I. and Makino, K. Preparation and properties of monodispersed rifampicin-loaded poly(lactide-co-glycolide) microspheres. Colloid Surf. B. Biointerfaces, 39, 17–21 (2004).
Gogolewski, S., Jovanivich, M., Perren, M., Dillon, J. G., and Hughes, M. K., The effect of melt-processing on the degradation of selected polyhydroacids, polylactide, polyhydroxybutyrate and poly(hydroxybutyrate-co-hydroxyvalerate). J. Biomed. Mater. Res., 27, 1135–1148 (1993).
Govender, T., Stolnik, S., Garnett, M. C., Illum, S., and Davis, S. S., PLGA nanoparticles prepared by nanoprecipitation: drug loading and release studies of a water soluble drug. J. Control. Release, 57, 171–185 (1999).
Harma, R., Saxena, D., Dwivedi, A. K., and Misra, A., Inhalable microparticles containing drug combinations to target alveolar macrophages for treatment of pulmonary tuberculosis. Pharm. Res., 18, 1405–1410 (2001).
Kamath, M. P., Shenoy, B. D., Tiwari, S. B., Karki, R., Udupa, N., and Kotian, M., Prolonged release biodegradable vesicular carriers for rifampicin-formulation and kinetics of release. Indian J. Exp. Biol., 38, 113–138 (2000).
Kassab, A. C., Xu, K., Denkbas, E. B., Dou, Y., Zhao, S., and Piskin, E., Rifampicin carrying polyhydroxybutyrate microspheres as a potential chemoembolizant agent. J. Biomater. Sci. Polym., 8, 947–961 (1997).
Labana, S., Pandey, R., Sharma, S., and Khuller, G. K., Chemotherapeutic activity against murine tuberculosis of once weekly administered drugs (isoniazid and rifampicin) encapsulated in liposomes. Int. J. Antimicrob. Agents, 20, 301–304 (2002).
Marcato, P. D. and Duran, N., New aspects of nanopharmaceutical delivery systems. J. Nanosci. Nanotechnol., 8, 2216–2229 (2008).
Pandey, R., Sharma, A., Zahoor, A., Sharma, S., Khuller, G. K., and Prasad, B., Poly (DL-lactide-co-glycolide) nanoparticlebased inhalable sustained drug delivery system for experimental tuberculosis. J. Antimicrob. Chemother., 52, 981–986 (2003).
Pandey, R., Sharma, S., and Khuller, G. K., Oral solid lipid nanoparticle-based antitubercular chemotherapy. Tuberculosis, 85, 415–420 (2005).
Pandey, R., Sharma, S., and Khuller, G. K., Chemotherapeutic efficacy of nanoparticle encapsulated antitubercular drugs. Drug Delivery, 13, 287–294 (2006a).
Pandey, R., Sharma, S., and Khuller, G. K., Oral poly(lactideco-glycolide) nanoparticle based antituberculosis drug delivery: Toxicological and chemotherapeutic implications. Indian J. Exper. Biol., 44, 459–467 (2006b).
Petrelli, D., Repetto, A., D’Ercole, S., Rombini, S., Ripa, S., Prenna, M., and Vitali, L. A., Analysis of meticillin-susceptible and meticillinresistant biofilm-forming Staphylococcus aureus from catheter infections isolated in a large Italian hospital. J. Med. Microbiol., 57, 364–372 (2008).
Pich, A., Schiemenz, N., Corten, C., and Adler, H. J. P., Preparation of poly(3hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) particles in O/W emulsion. Polymer, 47, 1912–1920 (2006).
Sendil, A., Gursel, I., Wise, D. L., and Hasirci, V., Antibiotic release from biodegradable PHBV microparticles. J. Control. Release, 59, 207–217 (1999).
Schaffazick, S. R., Guterres, S. S. U., Freitas, L. D., and Pohlmann, A. R., Physicochemical characterization and stability of the polymeric nanoparticle systems for drug administration. Quim Nova, 26, 726–737 (2003).
Sharma, R., Muttil, P., Yadav, A. B., Rath, S. K., Bajpai, V. B., Mani, U., and Misra, A., Uptake of inhalable microparticles affects defense responses of macrophages infected with Mycobacterium tuberculosis H37Ra. J. Antimicrob. Chemother., 59, 499–506 (2007).
Takenaga, M., Ohta, Y., Tokura, Y., Hamaguchi, A., Igarashi, R., Disratthakit, A., and Doi, N., Lipid microsphere formulation containing rifampicin targets alveolar macrophages. Drug Delivery., 15, 169–175 (2008).
Tomoda, K. and Makino, K., Effects of lung surfactants on rifampicin release rate from monodisperse rifampicinloaded PLGA microspheres. Colloids Surf. B: Biointerfaces 55, 115–124 (2007).
Tomoda, K., Kojima, S., Kajimoto, M., Watanabe, D., Nakajima, T., and Makino, K., Effects of pulmonary surfactant system on rifampicin release from rifampicin-loaded PLGA microspheres. Colloids Surf. B: Biointerfaces, 45, 1–6 (2005).
Ul-Ain, Q., Sharma, S., Khuller, G. K., and Garg, S. K., Alginate-based oral drug delivery system for tuberculosis: Pharmacokinetics and therapeutic effects. J. Antimicrob. Chemother., 51, 931–938 (2003).
Zahoor, A., Sharma, S., and Khuller, G. K., Inhalable alginate nanoparticles as antitubercular drug carriers against experimental tuberculosis. Inter. J. Antimicrob. Agents, 26, 298–303 (2005)
Zahoor, A., Sharma, S., and Khuller, G. K., Chemotherapeutic evaluation of alginate nanoparticle-encapsulated azole antifungal and antitubercular drugs against murine tuberculosis. Nanomedicine: NBM., 3, 239–243 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Durán, N., Alvarenga, M.A., Da Silva, E.C. et al. Microencapsulation of antibiotic rifampicin in poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Arch. Pharm. Res. 31, 1509–1516 (2008). https://doi.org/10.1007/s12272-001-2137-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-2137-7