Abstract
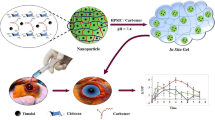
Two different chitosan (CS) nanocarriers namely nanoparticles and nanoemulsion were developed to prolong Indomethacin (IM) precorneal residence time and to improve its ocular bioavailability the main limitations in its management of post-operative inflammation and intraocular irritation after cataract extraction. CS-nanoparticles were developed by modified ionic gelation of CS with tripolyphosphate while nanoemulsion was prepared by spontaneous emulsification technique. Transmission electron microscopy revealed regular well-identified spherical shape. The nanoparticles had a mean size of 280 nm, a zeta potential of + 17 mV and high loading efficiency of 84.8 % while the mean size of nanoemulsion was affected by the nature of the surfactant used and varies between 220–690 nm. In vitro release studies, performed under sink conditions, revealed small initial burst release during the first hour followed by slow gradual drug release of 76 and 86% from nanoparticles and nanoemulsion respectively during a 24 h peroid. In vivo studies and histopathological examination revealed that eyes of rabbits treated with nanoemulsion showed clearer healing of corneal chemical ulcer with moderate effective inhibition of polymorph nuclear leuckocytic infiltration (PMNLs) compared with nanoparticles preparation. Moreover, following topical instillation of CS-nanoemulsion to rabbits, it was possible to achieve therapeutic concentration of IM in the cornea through out the duration of the study and fairly high IM level in inner ocular structure, aqueous humor. These levels were significantly higher than those obtained following instillation of IM solution. Therefore, CS nanocarriers developed in this study were able to contact intimately with the cornea providing slow gradual IM release with long-term drug level thereby increasing delivery to both external and internal ocular tissues.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agnihotri, S. A., Mallikarjuna, N. N., and Aminabhavi, T. M., Recent advances on chitosan-based micro-and nanoparticles in drug delivery. J. Control. Rel., 100, 5–28 (2004).
Ammoury, N., Fessi, H., Devissagguet, J. P., Puisieux, F., and Benita, S., In vitro release kinetic pattern of indomethacin from poly (D,L-lactide) nanocapsules. J. Pharm. Sci., 79, 763–767 (1990).
Artusson, P., Lindmark, T., Davis, S. S., and Illum, L., Effect of chitosan on the permeability of monolayers of intestinal epithelial cells (caco-2). Pharm. Res., 11, 1358–1361 (1994).
Bochot, A., Fattal, E., Grossiord, J. L., Puisieux, F., and Couvereur, P., Characterization of a new ocular delivery system based on a dispersion of liposomes in a thermosensitive gel. Int. J. Pharm., 162, 119–127 (1998).
Calvo, P., Alonso, M., Vila-Jato, J. L., and Robinson, J. R., Improved ocular bioavailability of indomethacin by ocular drug carriers. J. Pharm. Phaemacol., 48, 1147–1152 (1996a).
Calvo, P., Remuýán-López, C., Vila-Jato, J. L., and Alonso, M., Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J. Appl. Polym. Sci., 63, 125–132 (1997b).
Calvo, P., Vila-Jato, J. L., and Alonso, M., Comparative in vitro evaluation of several colloidal systems, nanoparticles, nanocapsules and nanoemulsions, as ocular drug carriers. J. Pharm. Sci., 85, 530–536 (1996b).
Calvo, P., Vila-Jato, J. L., and Alonso, M., Evaluation of cationic polymer-coated nanocapsules as ocular drug carriers. Int. J. Pharm., 153, 41–50 (1997a).
Davis, J. T. and Rideal, E. K., Interfacial Phenomena. Academic Press, New York (1963).
De Campos, A. M., Diebold, Y., Carvaiho, E. L. S., Sanchez, A., and Alonso, M. J., Chitosan nanoparticles as new ocular drug delivery system: in vitro stability, in vivo fate, and cellular toxicity. Pharm. Res., 21, 803–810 (2004).
De Campos, A. M., Sánchez, A., Gref, R., Calvo, P., and Alonso, M. J., The effect of PEG versus a chitosan coating on the interaction of drug colloidal carriers with the ocular mucosa. Eur. J. Pharm. Sci., 20, 73–81 (2003).
De Campos, A. M., Sánchez, A., Maria, J., and Alonso, M. J., Chitosan nanoparticles: a new vehicle for the improvement of the delivery of drugs to the ocular surface. Application to cyclosporine A. Int. J. Pharm., 224, 159–168 (2001).
Ding, S., Recent developments in ophthalmic drug delivery. Pharm. Sci. Technol. Today., 1, 328–335 (1998).
Dodane, V., Khan, M. A., and Merwin, J. R., Effect of chitosan on epithelium permeability and structure. Int. J. Pharm., 182, 21–32 (1999).
Dornish, M., Hagan, A., Hansson, E., Pecheur, C., Verdier, F., and Skaugrud, Q., Safety of protasan: ultrapure chitosan salts for biomedical and pharmaceutical use. Seventh International Conference on Chitin and Chitosan (1997).
Felt, O., Furrer, P., Mayer, J.M., Plazonnet, B., Buri, P., and Gurny, R., Topical use of chitosan in ophthalmology: tolerance assessment and evaluation of precorneal retention. Int. J. Pharm., 180, 185–193 (1999).
Ford, J., Woolfe, J., and Florence, A. T., Nanospheres of cyclosporine A: Poor oral absorption in dogs. Int. J. Pharm., 183, 3–6 (1999).
Grit, M., de Smidt, J. H., Struijke, A., and Crommelin, J. A., Hydrolysis of phosphatidylcholine in aqueous liposome dispersions. Int. J. Pharm., 50, 1–6 (1989).
Hill, J. M., O’Callaghan, R. J., Hobden, J. A., Kaufman, E., Corneal collagen shields for ocular drug delivery. In: Mita, A.K. (Eds.), Ophthalmic Drug Delivery Systems. Marcel Dekker, New York, pp. 261–275 (1993).
Illum, L., Chitosan and its uses as a pharmaceutical excipient. Pharm. Res., 15, 1326–1331(1998).
Indu, P. K. and Smitha, R., Penetration enhancers and ocular bioadhesive: Two new avenues for ophathalmic drug delivery. Drug Devel. Ind. Pharm., 28, 353–369 (2002).
Jumaa, M. and Muller, B. W., Physicochemical properties of chitosanlipid emulsions and their stability during the autoclaving process. 183, 175–184 (1999).
Jumaa, M. and Muller, B. W., Development of a novel parenteral formulation for tetrazepam using a lipid emulsion. Drug Dev. Ind. Pharm., 27, 1115–1121 (2001).
Kaur, I. P. and kanwar, M., Ocular preparations: The formulation approach. Drug Devel. Ind. Pharm., 28, 473–493 (2002).
Klang, S. H., Siganos, C. S., Benita, S., and Frucht-Pery, J., Evaluation of a positively charged submicron emulsion of piroxicam on the rabbit corneum healing process following alkali burn. J. Control. Rel., 57, 19–27 (1999).
Koch, M. A., Dodane, V., Khan, M. A., and Merwin, J. R., Chitosan induced effects on epithelial morphology as seen by confocal scanning microscopy, Scanning, 20, 262–263 (1998).
Lehr, C. M., Bowstra, J. A., Schacht, E. H., and Juginger, H. E., In vitro evaluation of mucodhesive properties of chitosan and some others natural polymers. Int. J. Pharm., 78, 43–48 (1992).
Losa, C., Calvo, P., Castro, E., Vila-Jato, J. L., and Alonso, M. J., Improvement of ocular penetration of amikacin sulphate by association to poly(butylcyanoacrylate) nanoparticles. J. Pharm. Pharmacol., 43, 548–552 (1991).
Losa, C., Marchal-Heussler, L., Orallo, F., Vila-Jato, J. L., and Alonso, M. J., Design of new formulations for topical ocular administration: polymeric nanocapsules containing metipranolol. Pharm. Res., 10, 80–87 (1993).
Miyake, K., Sugiyama, S., Norimatsu, I., and Ozawa, T., Prevention of cystoid macular edema after lens extraction by topical indomethacin (III) radioimmunoassay measurement of prostaglandins in aqueous humor during and after lens extraction procedures. Graefe’s Arch. Clin. Exp. Ophthalmol., 209, 83–88 (1978).
Mucha, M., Rheological characteristics of semi-diluted chitosan solutions. Macromol. Chem. Physical., 198, 471–484 (1997).
Muchtar, S., Abdulrazik, M., Pery, J. F., and Benita, S., Ex-vivo permeation study of indomethacin from a submicron emulsion through albino cornea. J. Control Rel., 44, 55–64 (1997).
Paul, W. and Sharma, C., Chitosan, a drug carrier for the 21st century, STP. Pharma. Sci., 10, 5–22 (2000).
Pleyer, U., Lutz, S., Jusko, W., Nguyen, K., Narawane, M., Rückert, D., Mondino, B. J., and Lee, V. H., Ocular absorption of topically applied FK506 from liposomal and oil formulations in rabbit eye. Invest. Ophthalmol. Vis. Sci., 34, 2737–2742 (1993).
Ritger, P. L. and Peppas, N. A., A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J. Controlled Rel., 5, 37–42 (1987).
Schipper, N. G. M., Olsson, S., Hoostraate, A. J., Deboer, A. G., Varum, K. M., and Artursson, P., Chitosan as absorption for poorly absorbable drug 2: mechanism of absorption enhancement. Pharm. Res., 14, 923–929 (1997).
Skellern, G. G. and Salole, E. G., A high-speed liquid chromatographic analysis of indomethacin in plasma. J. Chromatography, 114, 483–485 (1975).
Srinivasan, B. D. and Kulkarni, P. S., Polymorphonuclear leukocyte response. Inhibition following corneal epithelial denudation by steroidal and nonsteroidal anti-inflammatory agents. Arch Ophthalmol., 99, 1085–1089 (1981).
Vandenberg, G. W., Drolet, C., Scott, S. L., and Noue, J. D., Factors affecting protein release from alginate-chitosan coacervate microcapsules during production and gastrointestinal simulation. J. Control. Rel., 77, 297–307 (2001).
VanSantvliet, L. and Ludwig, A., The influence of penetration enhancers on the volume instilled of eye drops. Eur. J. Pharm. Biopharm., 45, 189–198 (1998).
Wu, Y., Yang, W., Wang, C., HU, J., and Fu, S., Chitosan nanoparticles as a novel delivery system for ammonium glycyrrhizinate. Int. J. Pharm., 295, 235–245 (2005).
Xu, Y. and Du, Y., Effect of molecular structure of chitosan on protein delivery properties of chitosan nanoparticles. Int. J. Pharm., 250, 215–226 (2003).
Zhang, W., Prausnitz, M. R., and Edwards, A., Modal of transient drug diffusion across cornea. J. Control. Rel., 99, 241–258 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Badawi, A.A., El-Laithy, H.M., El Qidra, R.K. et al. Chitosan based nanocarriers for indomethacin ocular delivery. Arch. Pharm. Res. 31, 1040–1049 (2008). https://doi.org/10.1007/s12272-001-1266-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-1266-6