Abstract
Objective
To investigate the mechanisms of excitotoxic effects of glutamate on human neuroblastoma SH-SY5Y cells.
Methods
SH-SY5Y cell viability was measured by MTT assay. Other damaged profile was detected by lactate dehydrogenase (LDH) release and by 4′, 6-diamidino-2-phenylindole (DAPI) staining. The cytosolic calcium concentration was tested by calcium influx assay. The glutamate-induced oxidative stress was analyzed by cytosolic glutathione assay, superoxide dismutase (SOD) assay and extracellular malondialdehyde (MDA) assay.
Results
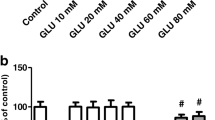
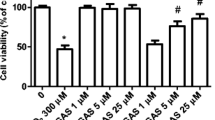
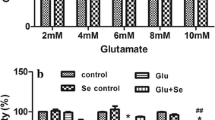
Glutamate treatment caused damage in SHSY5Y cells, including the decrease of cell viability, the increase of LDH release and the alterations of morphological structures. Furthermore, the concentration of cytoplasmic calcium in SH-SY5Y cells was not changed within 20 min following glutamate treatment, while cytosolic calcium concentration significantly increased within 24 h after glutamate treatment, which could not be inhibited by MK801, an antagonist of NMDA receptors, or by LY341495, an antagonist of metabotropic glutamate receptors. On the other hand, oxidative damage was observed in SH-SY5Y cells treated with glutamate, including decreases in glutathione content and SOD activity, and elevation of MDA level, all of which could be alleviated by an antioxidant Tanshinone IIA (Tan IIA, a major active ingredient from a Chinese plant Salvia Miltiorrhiza Bge).
Conclusion
Glutamate exerts toxicity in human neuroblastoma SH-SY5Y cells possibly through oxidative damage, not through calcium homeostasis destruction mediated by NMDA receptors.
摘要
目的
探讨谷氨酸导致人神经母细胞瘤细胞(SH-SY5Y cells)兴奋性毒损伤的机制。
方法
MTT法检测SH-SY5Y细胞存活率; 测定乳酸脱氢酶释放量观察细胞损伤程度; DAPI染色法观察细胞凋亡形态学特点; 钙流法检测胞浆钙离子浓度变化; 以胞内谷胱甘肽、 超氧化物歧化酶活性和胞外丙二醛含量检测谷氨酸引发SH-SY5Y细胞的氧化应激状态。
结果
谷氨酸导致SH-SY5Y细胞受损, 包括存活率下降、 乳酸脱氢酶释放量增多及形态结构发生改变; 谷氨酸处理20 min 后, 胞浆钙离子浓度无显著改变, 而处理24 h 后, 胞浆钙离子大量增加, 且MK801 (NMDA受体拮抗剂)及LY341495 (代谢型谷氨酸受体拮抗剂)均不能抑制钙离子内流的增多; 谷氨酸可导致SH-SY5Y氧化损伤, 包括胞内谷胱甘肽含量减少、 超氧化物歧化酶活性降低、 胞外脂质过氧化产物丙二醛水平升高等, 而丹参酮IIA (一种抗氧化剂)可减轻这些氧化损伤。
结论
谷氨酸导致SH-SY5Y细胞兴奋性毒损伤可能是通过氧化损伤产生的, 而不依赖于NMDA 受体介导的钙稳态的破坏。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ludolph AC, Münch C. Neurotoxic mechanisms of degeneration in motor neuron diseases. Drug Metab Rev 1999, 31: 619–634.
Shaw PJ. Calcium, glutamate, and amyotrophic lateral sclerosis: more evidence but no certainties. Ann Neurol 1999, 46: 803–805.
Spencer PS. Food toxins, AMPA receptors, and motor neuron diseases. Drug Metab Rev 1999, 31: 561–587.
Rothman SM, Olney JW. Glutamate and the pathophysiology of hypoxic-ischemic brain damage. Ann Neurol 1986, 19: 105–111.
Doble A. The role of excitotoxicity in neurodegenerative disease: implications for therapy. Pharmacol Ther 1999, 81: 163–221.
Hynd MR, Scott HL, Dodd PR. Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer’s disease. Neurochem Int 2004, 45: 583–595.
Blandini F, Greenamyre JT, Nappi G. The role of glutamate in the pathophysiology of Parkinson’s disease. Funct Neuro 1996, 11: 3–15.
Zeron MM, Chen NS, Moshaver A, Lee AT, Wellington CL, Hayden MR, et al. Mutant huntingtin enhances excitotoxic cell death. Mol Cell Neurosci 2001, 17: 41–53.
Ankacrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, et al. Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron 1995, 15: 961–973.
Behl C, Widmann M, Trapp T, Holsboer F. 17β-estradiol protects neurons from oxidative stress-induced cell death in vitro. Biochem Biophys Res Commun 1995, 216: 473–482.
Schubert D, Piasecki D. Oxidative glutamate toxicity can be a component of the excitotoxicity cascade. J Neurosci 2001, 21: 7455–7462.
Pereira CMF, Oliveira CR. Glutamate toxicity on a PC12 cell line involves glutathione depletion and oxidative stress. Free Radical Biol Med 1997, 23: 637–647.
Pereira C, Santos MS, Oliveira C. Metabolic inhibition increases glutamate susceptibility on a PC12 cell line. J Neurosci Res 1998, 51: 360–370.
Schubert D, Kimura H, Maher P. Growth factors and vitamin E modify neuronal glutamate toxicity. Proc Natl Acad Sci U S A 1992, 89: 8264–8267.
Froissard P, Duval D. Cytotoxic effects of glutamic acid on PC12 cells. Neurochem Int 1994, 24: 485–493.
Roshandel NR, Razavi LL, Far BT, Mahmoudian M. Mebudipine and dibudipine protect PC12 cells against oxygen-glucose deprivation and glutamate-induced cell death. Pathophysiology 2008, 15: 227–231.
Chen WD, Li YL, Li L. Influence of potassium chloride or glutamate on calcium ion permeability of human neuroblastoma SH-SY5Y cell line. Chin J Cell Biol 2000, 22: 206–209.
Nikolova S, Lee YS, Lee YS, Kim JA. Rac1-NADPH oxidaseregulated generation of reactive oxygen species mediates glutamate-induced apoptosis in SH-SY5Y human neuroblastoma cells. Free Radic Res 2005, 39: 1295–1304.
Gao M, Zhang WC, Liu QS, Hu JJ, Liu GG, Du GH. Pinocembrin prevents glutamate-induced apoptosis in SH-SY5Y neuronal cells via decrease of bax/bcl-2 ratio. Eur J Pharmacol 2008, 591: 73–79.
Olney JW. Inciting excitotoxic cytocide among central neurons. Adv Exp Med Biol 1986, 203: 631–645.
Choi DW. Calcium: still center-stage in hypoxic-ischemic neuronal death. Trends Neurosci 1995, 18: 58–60.
Leon R, Wu H, Jin Y, Wei JN, Buddhala C, Prentice H, et al. Protective function of Taurine in glutamate-induced apoptosis in cultured neurons. J Neurosci Res 2009, 87: 1185–1194.
Tan S, Sagara Y, Liu Y, Maher P, Schubert D. The regulation of peroxide production during programmed cell death. J Cell Biol 1998, 141: 1423–1432.
Tan S, Wood M, Maher P. Oxidative stress in nerve cells induces a form of cell death with characteristics of both apoptosis and necrosis. J Neurochem 1998, 71: 95–105.
Leclerc CL, Chi CL, Awobuluyi M, Sucher NJ. Expression of N-methyl-D-aspartate receptor subunit mRNAs in the rat pheochromocytoma cell line PC12. Neurosci Lett 1995, 201: 103–106.
Casado M, L-Gaujardo A, Mellstrom B, Naranjo JR, Lerma J. Functional N-methyl-D-aspartate receptors in clonal pheochromocytoma cells. J Physiol 1996, 490: 391–404.
Sucher NJ, Brose N, Deitcher DL, Awobluluyi M, Gasie GP, Bading H, et al. Expression of endogenous NMDAR1 transcripts without receptor protein suggests posttranscriptional control in PC12 cells. J Biol Chem 1993, 268: 22299–22304.
Naarala J, Tervo P, Loikkanen J, Savolainen K. Blocking of carbachol-induced calcium mobilization by glutamate receptor antagonists. Neurosci Res Commun 2001, 30: 1–6.
Akundi RS, Hüll M, Clement HW, Fiebich BL. 1-Trichloromethy1,2,3,4-tetrahydro-β-carboline (TaClo) induces apoptosis in human neuroblastoma cell lines. Ann NY Acad Sci 2003, 1010: 304–306.
Adamec E, Beermann ML, Nixon RA. Calpain I activation in rat hippocampal neurons in culture is NMDA receptor selective and not essential for excitotoxic cell death. Mol Brain Res 1998, 54: 35–48.
Río ED, Mclaughlin M, Downes CP, Nicholls DG. Differential coupling of G-protein-linked receptors to Ca2+ mobilization through inositol(1,4,5)trisphosphate or ryanodine receptors in cerebellar granule cells in primary culture. Eur J Neurosci 1999, 11: 3015–3022.
Fagni L, Ango F, Prezeau L, Worley PF, Pin JP, Bockaert J. Control of constitutive activity of metabotropic glutamate receptors by Homer proteins. Int Congr Ser 2003, 1249: 245–251.
Sato H, Tamba M, Ishii T, Bannai S. Cloning and expression of a plasma membrane cystine/glutamate exchange transporter composed of two distinct proteins. J Biol Chem 1999, 274: 11455–11458.
Murphy TH, Miyamoto M, Sastre A, Schnaar RL, Coyle JT. Glutamate toxicity in a neuronal cell line involves inhibition of cystine transport leading to oxidative stress. Neuron 1989, 2: 1547–1558.
Lin R, Wang WR, Liu JT, Yang GD, Han CJ. Protective effect of tanshinone IIA on human umbilical vein endothelial cell injured by hydrogen peroxide and its mechanism. J Ethnopharmacol 2006, 108: 217–222.
Li YI, Elmer G, LeBoeuf RC. Tanshinone IIA reduces macrophage death induced by hydrogen peroxide by upregulating glutathione peroxidase. Life Sci 2008, 83: 557–562.
Murata T, Omata N, Fujibayashi Y, Waki A, Sadato N, Yoshimoto M, et al. Neurotoxicity after hypoxia/during ischemia due to glutamate with/without free radicals as revealed by dynamic changes in glucose metabolism. Brain Res 2000, 865: 259–263.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Sun, ZW., Zhang, L., Zhu, SJ. et al. Excitotoxicity effects of glutamate on human neuroblastoma SH-SY5Y cells via oxidative damage. Neurosci. Bull. 26, 8–16 (2010). https://doi.org/10.1007/s12264-010-0813-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-010-0813-7