Abstract
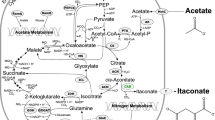
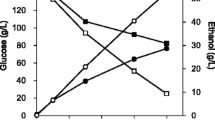
For growth and energy, Corynebacterium glutamicum has the ability to assimilate numerous carbon sources in the form of either single or combined substrates. During the growth of C. glutamicum on substrate mixtures, it has shown the ability to co-metabolize the majority of these carbon sources and displays monophasic growth, unlike other microorganisms such as Escherichia coli and Bacillus subtilis, which exhibit either diauxic or biphasic growth. Here, a recombinant strain of C. glutamicum ATCC 13032 was selected for the use in the production of itaconic acid (IA), which is a promising biochemical building block that could be an alternative material for polymer synthesis. For this purpose, an engineered C. glutamicum ATCC 13032 pCH-cadAopt was constructed by introducing the plasmid pCH-cadAopt, which expressed a cis-aconitate dehydrogenase gene (cadA) that originated from Aspergillus terreus. The production of IA was evaluated using a combined mixture of maltose and sodium acetate. The monophasic growth of C. glutamicum in the presence of maltose and sodium acetate was observed and showed final IA titer of 12.63 g/L, and a molar yield of 0.38 mol/mol after 240 h of cultivation. The present study suggests the possibility of utilizing a mixture of carbon sources by C. glutamicum to improve IA production.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gerstmeir, R., V. F. Wendisch, S. Schnicke, H. Ruan, M. Farwick, D. Reinscheid, and B. J. Eikmanns (2003) Acetate metabolism and its regulation in Corynebacterium glutamicum. J Biotechnol. 104: 99–122.
Monod, J. (1949) The growth of bacterial cultures. Annu Rev Microbiol. 3: 371–394.
Brückner, R. and F. Titgemeyer (2002) Carbon catabolite repression in bacteria: choice of the carbon source and autoregulatory limitation of sugar utilization. FEMS Microbiol. Lett. 209: 141–148.
Stülke, J. and W. Hillen (2000) Regulation of carbon catabolism in Bacillus species. Annu. Rev. Microbiol. 54: 849–880.
Harder, W. and L. Dijkhuizen (1982) Strategies of mixed substrate utilization in microorganisms. Philos. Trans. R. Soc. Lond. B Biol. Sci. 297: 459–480.
Kovárová-Kovar, K. and T. Egli (1998) Growth kinetics of suspended microbial cells: from single-substrate-controlled growth to mixed-substrate kinetics. Microbiol. Mol. Biol. Rev. 62: 646–666.
Lee, J.-Y., Y.-A. Na, E. Kim, H.-S. Lee, and P. Kim (2016) The actinobacterium Corynebacterium glutamicum, an industrial workhorse. J. Microbiol. Biotechnol. 26: 807–822. (Erratum published 2016, J. Microbiol. Biotechnol. 26: 1341)
Zahoor, A., S. N. Lindner, and V. F. Wendisch (2012). Metabolic engineering of Corynebacterium glutamicum aimed at alternative carbon sources and new products. Comput. Struct. Biotechnol. 3: e201210004.
Krause, F. S., A. Henrich, B. Blombach, R. Krämer, B. J. Eikmanns, and G. M. Seibold (2010). Increased glucose utilization in Corynebacterium glutamicum by use of maltose, and its application for the improvement of L-valine productivity. Appl. Environ. Microbiol. 76: 370–374.
Cocaign, M., C. Monnet, and N. D. Lindley (1993) Batch kinetics of Corynebacterium glutamicum during growth on various carbon substrates: use of substrate mixtures to localise metabolic bottlenecks. Appl. Microbiol. Biotechnol. 40: 526–530.
Dominguez, H., C. Nezondet, N. D. Lindley, and M. Cocaign (1993) Modified carbon flux during oxygen limited growth of Corynebacterium glutamicum and the consequences for amino acid overproduction. Biotechnol. Lett. 15: 449–454.
Wendisch, V. F., A. A. de Graaf, H. Sahm, and B. J. Eikmanns (2000) Quantitative determination of metabolic fluxes during coutilization of two carbon sources: comparative analyses with Corynebacterium glutamicum during growth on acetate and/or glucose. J. Bacteriol. 182: 3088–3096.
Claes, W. A., A. Pühler, and J. Kalinowski (2002) Identification of two prpDBC gene clusters in Corynebacterium glutamicum and their involvement in propionate degradation via the 2-methylcitrate cycle. J. Bacteriol. 184: 2728–2739.
Kawaguchi, H., K. Yoshihara, K. Y. Hara, T. Hasunuma, C. Ogino, and A. Kondo (2018) Metabolome analysis-based design and engineering of a metabolic pathway in Corynebacterium glutamicum to match rates of simultaneous utilization of D-glucose and L-arabinose. Microb. Cell Fact. 17: 76.
Kawaguchi, H., A. A. Vertès, S. Okino, M. Inui, and H. Yukawa (2006) Engineering of a xylose metabolic pathway in Corynebacterium glutamicum. Appl. Environ. Microbiol. 72: 3418–3428.
Klement, T. and J. Büchs (2013) Itaconic acid—a biotechnological process in change. Bioresour. Technol. 135: 422–431.
El-Imam, A. A. and C. Du (2014) Fermentative itaconic acid production. J. Biodivers Biopros. Dev. 1: 119.
Magalhães, A. I., Jr, J. C. de Carvalho, J. D. C. Medina, and C. R. Soccol (2017) Downstream process development in biotechnological itaconic acid manufacturing. Appl. Microbiol. Biotechnol. 101: 1–12.
Krull, S., A. Hevekerl, A. Kuenz, and U. Prüße (2017) Process development of itaconic acid production by a natural wild type strain of Aspergillus terreus to reach industrially relevant final titers. Appl. Microbiol. Biotechnol. 101: 4063–4072.
Saha B. C. (2017) Emerging biotechnologies for production of itaconic acid and its applications as a platform chemical. J. Ind. Microbiol. Biotechnol. 44: 303–315.
Schlembach, I., H. Hosseinpour Tehrani, L. M. Blank, J. Büchs, N. Wierckx, L. Regestein, and M. A. Rosenbaum (2020) Consolidated bioprocessing of cellulose to itaconic acid by a co-culture of Trichoderma reesei and Ustilago maydis. Biotechnol. Biofuels 13: 207.
Demir, H. T., E. Bezirci, J. Becker, H. H. Tehrani, E. Nikerel, N. Wierck, and M. Türker (2021) High level production of itaconic acid at low pH by Ustilago maydis with fed-batch fermentation. Bioprocess Biosyst. Eng. 44: 749–758.
Hosseinpour Tehrani, H., J. Becker, I. Bator, K. Saur, S. Meyer, A. C. Rodrigues Lóia, L. M. Blank, and N. Wierckx (2019) Integrated strain- and process design enable production of 220 g L−1 itaconic acid with Ustilago maydis. Biotechnol. Biofuels 12: 263.
Becker, J., H. H. Tehrani, P. Ernst, L. M. Blank, and N. Wierckx (2020) An optimized Ustilago maydis for itaconic acid production at maximal theoretical yield. J. Fungi (Basel) 7: 20.
Harder, B.-J., K. Bettenbrock, and S. Klamt (2016) Model-based metabolic engineering enables high yield itaconic acid production by Escherichia coli. Metab. Eng. 38: 29–37.
Harder, B.-J., K. Bettenbrock, and S. Klamt (2018) Temperature-dependent dynamic control of the TCA cycle increases volumetric productivity of itaconic acid production by Escherichia coli. Biotechnol Bioeng. 115: 156–164.
Okamoto, S., T. Chin, K. Hiratsuka, Y. Aso, Y. Tanaka, T. Takahashi, and H. Ohara (2014) Production of itaconic acid using metabolically engineered Escherichia coli. J. Gen. Appl. Microbiol. 60: 191–197.
Merkel, M., D. Kiefer, M. Schmollack, B. Blombach, L. Lilge, M. Henkel, and R. Hausmann (2022) Acetate-based production of itaconic acid with Corynebacterium glutamicum using an integrated pH-coupled feeding control. Bioresour. Technol. 351: 126994.
Otten, A., M. Brocker, and M. Bott (2015) Metabolic engineering of Corynebacterium glutamicum for the production of itaconate. Metab. Eng. 30: 156–165.
Sambrook, J., E. F. Fritsch, and T. Maniatis (1989) Molecular Cloning: A Laboratory Manual. 2nd ed. Cold Spring Harbor Laboratory Press.
Vertès, A. A., M. Inui, M. Kobayashi, Y. Kurusu, and H. Yukawa (1993) Presence of mrr- and mcr-like restriction systems in coryneform bacteria. Res. Microbiol. 144: 181–185.
Tateno, T., H. Fukuda, and A. Kondo (2007) Direct production of L-lysine from raw corn starch by Corynebacterium glutamicum secreting Streptococcus bovis alpha-amylase using cspB promoter and signal sequence. Appl. Microbiol. Biotechnol. 77: 533–541.
Zhang, Y., J. Cai, X. Shang, B. Wang, S. Liu, X. Chai, T. Tan, Y. Zhang, and T. Wen (2017) A new genome-scale metabolic model of Corynebacterium glutamicum and its application. Biotechnol. Biofuels 10: 169.
Wada, K., Y. Toya, S. Banno, K. Yoshikawa, F. Matsuda, and H. Shimizu (2017) 13C-metabolic flux analysis for mevalonate-producing strain of Escherichia coli. J. Biosc. Bioeng. 123: 177–182.
Becker, S. A., A. M. Feist, M. L. Mo, G. Hannum, B. Ø. Palsson, and M. J. Herrgard (2007) Quantitative prediction of cellular metabolism with constraint-based models: the COBRA Toolbox. Nat. Protoc. 2: 727–738.
Heirendt, L., S. Arreckx, T. Pfau, S. N. Mendoza, A. Richelle, A. Heinken, H. S. Haraldsdóttir, J. Wachowiak, S. M. Keating, V. Vlasov, S. Magnusdóttir, C. Y. Ng, G. Preciat, A. Žagare, S. H. J. Chan, M. K. Aurich, C. M. Clancy, J. Modamio, J. T. Sauls, A. Noronha, A. Bordbar, B. Cousins, D. C. El Assal, L. V. Valcarcel, I. Apaolaza, S. Ghaderi, M. Ahookhosh, M. Ben Guebila, A. Kostromins, N. Sompairac, H. M. Le, D. Ma, Y. Sun, L. Wang, J. T. Yurkovich, M. A. P. Oliveira, P. T. Vuong, L. P. El Assal, I. Kuperstein, A. Zinovyev, H. S. Hinton, W. A. Bryant, F. J. Aragón Artacho, F. J. Planes, E. Stalidzans, A. Maass, S. Vempala, M. Hucka, M. A. Saunders, C. D. Maranas, N. E. Lewis, T. Sauter, B. Ø. Palsson, I. Thiele, and R. M. T. Fleming (2019) Creation and analysis of biochemical constraint-based models using the COBRA Toolbox v.3.0. Nat. Protoc. 14: 639–702.
Wieschalka, S. and B. Litsanov (2021) Bio-Based Organic and Hydroxy Acid Production with Corynebacterium Glutamicum. pp. 2871–2899. Nova Science Publishers.
Li, A., N. van Luijk, M. ter Beek, M. Caspers, P. Punt, and M. van der Werf (2011) A clone-based transcriptomics approach for the identification of genes relevant for itaconic acid production in Aspergillus. Fungal Genet. Biol. 48: 602–611.
Yang, Z., X. Gao, H. Xie, F. Wang, Y. Ren, and D. Wei (2017) Enhanced itaconic acid production by self-assembly of two biosynthetic enzymes in Escherichia coli. Biotechnol. Bioeng. 114: 457–462.
Arnold, S., T. Tews, M. Kiefer, M. Henkel, and R. Hausmann (2019) Evaluation of small organic acids present in fast pyrolysis bio-oil from lignocellulose as feedstocks for bacterial bioconversion. GCB Bioenergy 11: 1159–1172.
Kiefer, D., M. Merkel, L. Lilge, R. Hausmann, and M. Henkel (2021) High cell density cultivation of Corynebacterium glutamicum on bio-based lignocellulosic acetate using pH-coupled online feeding control. Bioresour. Technol. 340: 125666.
Baronofsky, J. J., W. J. Schreurs, and E. R. Kashket (1984) Uncoupling by acetic acid limits growth of and acetogenesis by Clostridium thermoaceticum. Appl. Environ. Microbiol. 48: 1134–1139.
Acknowledgements
This work was partially supported by the New Energy and Industrial Technology Development Organization (NEDO), Japan, Grant Number JPNP18016, and by the Environmental Restoration and Conservation Agency, Japan, Grant Number JPMEERF20201005. The researcher (Taghreed Elkasaby) is funded by a full scholarship (the EJEP-HRDP Egypt-Japan Education Partnership; Human Resource Development Project) from the Ministry of Higher Education of the Arab Republic of Egypt.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Elkasaby, T., Hanh, D.D., Kawaguchi, H. et al. Co-utilization of Maltose and Sodium Acetate via Engineered Corynebacterium glutamicum for Improved Itaconic Acid Production. Biotechnol Bioproc E 28, 790–803 (2023). https://doi.org/10.1007/s12257-023-0091-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-023-0091-7