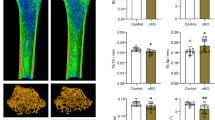
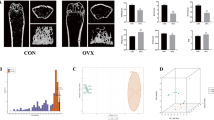
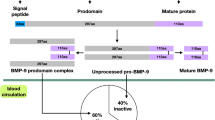
Abstract
Ces3 is a lipolytic enzyme predominantly present in liver and adipocytes, with recent reports of its presence in skeletal muscles, as well. A cross-linking study to understand the various interacting proteins involved in bone-adipose axis could provide novel targets for drug development. We explored the functional role of Ces3 in osteoblasts and mesenchymal stem cells differentiating into osteoblast lineage using in vitro models. We also investigated the physiological functions of Ces3 by stable gene knockdown of Ces3 and exogenous Ces3 induction, and examined the interacting proteins by Co-IP and in-silico analysis. Data from our study suggests that Ces3 is highly expressed in osteoblasts and promotes proliferation of the cells by increasing the expressions of osteogenic marker proteins and genes. For the first time, our mechanistic studies revealed that Ces3 interacts with BMP11 protein for regulation of osteoblast differentiation and activates the ALK2 and BMP type II receptors via Smad 1/5/9 signaling pathways. In addition, we identified the various partner proteins linked to Ces3 and BMP11 which are also involved in the metabolic network of osteoblasts. In silico analysis revealed a direct and strong interaction between Ces3 and BMP11 which influences the growth and regulation of osteoblasts. Current data unveiled a hitherto unknown mechanism of Ces3 and BMP11 in the bone-adipose axis, shedding light on Ces3 as a pharmacotherapeutic target to treat metabolic disorders.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Acvr1 :
-
gene encoding activin receptor-like kinase-2
- Acvr1b :
-
gene encoding activin receptor type-1B
- Acvr2a :
-
gene encoding activin receptor type-2A
- Acvr2b :
-
gene encoding activin receptor type-2B
- ALK2 :
-
activin receptor-like kinase-2
- ALP :
-
alkaline phosphatase
- Bglap :
-
gene encoding osteocalcin
- BMP11/Bmp11 :
-
bone morphogenetic protein 11 / encoding gene
- Bmpr2 :
-
gene encoding bone morphogenetic protein receptor type 2
- BMPRII :
-
bone morphogenetic protein receptor type 2
- Ces3/Ces3 :
-
carboxylesterase 3 / encoding gene
- COXIV :
-
cytochrome oxidase IV
- GDF11 :
-
growth differentiation factor-11
- Osx :
-
Osterix
- Runx2/Runx2 :
-
Runt-related transcription factor 2 / encoding gene
- Sp7 :
-
gene encoding osterix
- Tgfβ3 :
-
gene encoding transforming growth factor beta-3
References
Gómez-Ambrosi, J., A. Rodríguez, V. Catalán, and G. Frühbeck (2008) The bone-adipose axis in obesity and weight loss. Obes. Surg. 18: 1134–1143.
Kushwaha, P., M. J. Wolfgang, and R. C. Riddle (2018) Fatty acid metabolism by the osteoblast. Bone. 115: 8–14.
Gong, K., B. Qu, D. Liao, D. Liu, C. Wang, J. Zhou, and X. Pan (2016) MiR-132 regulates osteogenic differentiation via down-regulating sirtuin1 in a peroxisome proliferator-activated receptor β/d-dependent manner. Biochem. Biophys. Res. Commun. 478: 260–267.
Blair, H. C., Q. C. Larrouture, Y. Li, H. Lin, D. Beer-Stoltz, L. Liu, R. S. Tuan, L. J. Robinson, P. H. Schlesinger, and D. J. Nelson (2017) Osteoblast differentiation and bone matrix formation in vivo and in vitro. Tissue Eng. Part B Rev. 3: 268–280.
Savopoulos, C., C. Dokos, G. Kaiafa, and A. Hatzitolios (2011) Adipogenesis and osteoblastogenesis: trans-differentiation in the pathophysiology of bone disorders. Hippokratia. 15: 18–21.
Muruganandan, S., A. M. Ionescu, and C. J. Sinal (2020) At the crossroads of the adipocyte and osteoclast differentiation programs: future therapeutic perspectives. Int. J. Mol. Sci. 21: 2277.
Berendsen, A. D. and B. R. Olsen (2014) Osteoblast-adipocyte lineage plasticity in tissue development, maintenance and pathology. Cell Mol. Life Sci. 71: 493–49.
Javed, A. (2001) Runt homology domain transcription factors (Runx, Cbfa, and AML) mediate repression of the bone sialoprotein promoter: evidence for promoter context-dependent activity of Cbfa proteins. Mol. Cell Biol. 21: 2891–2905.
Zhang, C. (2010) Transcriptional regulation of bone formation by the osteoblast-specific transcription factor Osx. J. Orthop. Surg. Res. 5: 37.
Wang, R. N., J. Green, Z. Wang, Y. Deng, M. Qiao, M. Peabody, Q. Zhang, J. Ye, Z. Yan, S. Denduluri, O. Idowu, M. Li, C. Shen, A. Hu, R. C. Haydon, R. Kang, J. Mok, M. J. Lee, H. L. Luu, and L. L. Shi (2014) Bone morphogenetic protein (BMP) signaling in development and human diseases. Genes Dis. 1: 87–105.
Katagiri, T. and T. Watabe (2016) Bone morphogenetic proteins. Cold Spring Harb. Perspect. Biol. 8: a021899.
Zhang, Y. H., L. H. Pan, Y. Pang, J. X. Yang, M. J. Lv, F. Liu, X. F. Qu, X. X. Chen, H. J. Gong, D. Liu, and Y. Wei (2018) GDF11/BMP11 as a novel tumor marker for liver cancer. Exp. Ther. Med. 15: 3495–3500.
Sharma, D., S. Kosalge, and S. R. Dixit (2017) Influence of GDF-11 in aging process: an review. PharmaTutor. 5: 18–29.
Añón-Hidalgo, J., V. Catalán, A. Rodríguez, B. Ramírez, C. Silva, J. C. Galofré, J. Salvador, G. Frühbeck, and J. Gómez-Ambrosi (2019) Circulating GDF11 levels are decreased with age but are unchanged with obesity and type 2 diabetes. Aging. 11: 1733–1744.
Pham, H. G., S. Mukherjee, M. J. Choi, and J. W. Yun (2021) Bmp11 regulates thermogenesis in white and brown adipocytes. Cell Biochem. Funct. 39: 496–510.
Raichur, S., R. L. Fitzsimmons, S. A. Myers, M. A. Pearen, P. Lau, N. Erikson, S. M. Wang, and G. E. O. Muscat (2010) Identification and validation of the pathways and functions regulated by the orphan nuclear receptor, ROR alpha1, in skeletal muscle. Nucleic Acids Res. 38: 4296–4312.
Lian, J., R. Nelson, and R. Lehner (2018) Carboxylesterases in lipid metabolism: from mouse to human. Protein Cell. 9: 178–195.
Li, D. (2019) The impact of carboxylesterases in drug metabolism and pharmacokinetics. Curr. Drug Metab. 20: 91–102.
Wang, X., N. Rida, J. Shi, A. H. Wu, B. E. Bleske, and H. J. Zhu (2017) A comprehensive functional assessment of carboxylesterase 1 nonsynonymous polymorphisms. Drug Metab. Dispos. 45: 1149–1155.
Laizure, S. C., V. Herring, Z. Hu, K. Witbrodt, and R. B. Parker (2013) The role of human carboxylesterases in drug metabolism: have we overlooked their importance? Pharmacotherapy. 33: 210–222.
Zhao, B., J. Bie, J. Wang, S. A. Marqueen, and S. Ghosh (2012) Identification of a novel intracellular cholesteryl ester hydrolase (carboxylesterase 3) in human macrophages: compensatory increase in its expression after carboxylesterase 1 silencing. Am. J. Physiol. Cell Physiol. 303: C427–C435.
Tanaka, N., S. Takahashi, Z. Z. Fang, T. Matsubara, K. W. Krausz, A. Qu, and F. J. Gonzalez (2014) Role of white adipose lipolysis in the development of NASH induced by methionine- and choline-deficient diet. Biochim. Biophys. Acta. 1841: 1596–1607.
Soni, K. G., R. Lehners, P. Metalnikov, P. O’Donnell, M. Semache, W. Goas, K. Ashman, A. V. Pshezhetsky, and G. A. Mitchell (2004) Carboxylesterase 3 (EC 3.1.1.1) is a major adipocyte lipase. J. Biol. Chem. 279: 40683–40689.
Okazaki, H., M. Igarashi, M. Nishi, M. Tajima, M. Sekiya, S. Okazaki, N. Yahagi, K. Ohashi, K. Tsukamoto, M. Amemiya-Kudo, T. Matsuzaka, H. Shimano, N. Yamada, J. Aoki, R. Morikawa, Y. Takanezawa, H. Arai, R. Nagai, T. Kadowaki, J. I. Osuga, and S. Ishibashi (2006) Identification of a novel member of the carboxylesterase family that hydrolyzes triacylglycerol: a potential role in adipocyte lipolysis. Diabetes. 55: 2091–2097.
Ruby, M. A., J. Massart, D. M. Hunerdosse, M. Schönke, J. C. Correia, S. M. Louie, J. L. Ruas, E. Näslund, D. K. Nomura, and J. R. Zierath (2017) Human carboxylesterase 2 reverses obesity-induced diacylglycerol accumulation and glucose intolerance. Cell Rep. 18: 636–646.
Ross, M. K., T. M. Streit, and K. L. Herring (2010) Carboxylesterases: dual roles in lipid and pesticide metabolism. J. Pestic Sci. 35: 257–264.
Quiroga, A. D., L. Li, M. Trotzmuller, R. Nelson, S. D. Proctor, H. Kofeler, and R. Lehner (2012) Deficiency of carboxylesterase 1/esterase-x results in obesity, hepatic steatosis, and hyperlipidemia. Hepatology. 56: 2188–2198.
Dominguez, E., A. Galmozzi, J. W. Chang, K. L. Hsu, J. Pawlak, W. Li, C. Godio, J. Thomas, D. Partida, S. Niessen, P. E. O’Brien, A. P. Russell, M. J. Watt, D. K. Nomura, B. F. Cravatt, and E. Saez (2014) Integrated phenotypic and activity-based profiling links Ces3 to obesity and diabetes. Nat. Chem. Biol. 10: 113–121.
Lian, J., E. Wei, S. P. Wang, A. D. Quiroga, L. Li, A. D. Pardo, J. van der Veen, S. Sipione, G. A. Mitchell, and R. Lehner (2012) Liver specific inactivation of carboxylesterase 3/triacylglycerol hydrolase decreases blood lipids without causing severe steatosis in mice. Hepatology. 56: 2154–2162.
Yang, L., X. Li, H. Tang, Z. Gao, K. Zhang, and K. Sun (2019) A unique role of carboxylesterase 3 (Ces3) in β-adrenergic signaling-stimulated thermogenesis. Diabetes. 68: 1178–1196.
Chang, J., S. Oikawa, H. Iwahashi, E. Kitagawa, I. Takeuchi, M. Yuda, C. Aoki, Y. Yamada, G. Ichihara, M. Kato, and S. Ichihara (2014) Expression of proteins associated with adipocyte lipolysis was significantly changed in the adipose tissues of the obese spontaneously hypertensive/NDmcr-cp rat. Diabetol. Metab. Syndr. 6: 8.
Magnum, L. C., X. Hou, A. Borazjani, J. H. Lee, M. K. Ross, and J. A. Crow (2018) Silencing carboxylesterase 1 in human THP-1 macrophages perturbs genes regulated by PPARγ/RXR and RAR/RXR: down-regulation of CYP27A1-LXRα signaling. Biochem. J. 475: 621–642.
Mukherjee, S., M. Choi, and J. W. Yun (2019) Novel regulatory roles of carboxylesterase 3 in lipid metabolism and browning in 3T3-L1 white adipocytes. Appl. Physiol. Nutr. Metab. 44: 1089–1098.
Dimauro, I., T. Pearson, D. Caporossi, and M. J. Jackson (2012) A simple protocol for the subcellular fractionation of skeletal muscle cells and tissue. BMC Res. Notes 5: 513.
Bridges, D., L. Chang, I. J. Lodhi, N. A. Clark, and A. R. Saltiel (2012) TC10 is regulated by caveolin in 3T3-L1 adipocytes. PLoS One. 7: e42451.
Duhovny, D., R. Nussinov, and H. J. Wolfson (2002) Efficient unbound docking of rigid molecules. Proceedings of the Second Workshop on Al- rithms in Bioinformatics. September 17–21. Rome, Italy.
Zhang, C., G. Vasmatzis, J. L. Cornette, and C. DeLisi (1997) Determination of atomic desolvation energies from the structures of crystallized proteins. J. Mol. Biol. 267: 707–726.
Joo, J. I., T. S. Oh, D. H. Kim, D. K. Choi, X. Wang, J. W. Choi, and J. W. Yun (2011) Differential expression of adipose tissue proteins between obesity-susceptible and -resistant rats fed a high-fat diet. Proteomics. 11: 1429–1448.
Mukherjee, S., K. R. Aseer, and J. W. Yun (2020) Roles of macrophage colony stimulating factor in white and brown adipocytes. Biotechnol. Bioprocess Eng. 25: 29–38.
Hanna, H., L. M. Mir, and F. M. Andre (2018) In vitro osteoblastic differentiation of mesenchymal stem cells generates cell layers with distinct properties. Stem Cell Res. Ther. 9: 203.
Jensen, E. D., R. Gopalakrishnan, and J. J. Westendorf (2010) Regulation of gene expression in osteoblasts. Biofactors. 36: 25–32.
Jo, I., J. M. Lee., H. Suh., and H. Kim (2007) Bone tissue engineering using marrow stromal cells. Biotechnol. Bioprocess Eng. 12: 48–53.
Challa, T. D., L. G. Straub, M. Balaz, E. Kiehlmann, O. Donze, G. Rudofsky, J. Ukropec, B. Ukropcova, and C. Wolfrum (2015) Regulation of de novo adipocyte differentiation through cross talk between adipocytes and preadipocytes. Diabetes. 64: 4075–4087.
Zhu, M., E. Kohan, J. Bradley, M. Hedrick, P. Benhaim, and P. Zuk (2009) The effect of age on osteogenic, adipogenic and proliferative potential of female adipose-derived stem cells. J. Tissue Eng. Regen. Med. 3: 290–301.
Lu, Q., M. L. Tu, C. J. Li, L. Zhang, T. J. Jiang, T. Liu, and X. H. Luo (2016) GDF11 inhibits bone formation by activating Smad2/ 3 in bone marrow mesenchymal stem cells. Calcif. Tissue Int. 99: 500–509.
Shen, G. S., H. B. Zhou, H. Zhang, B. Chen, Z. P. Liu, Y. Yuan, X. Z. Zhou, and Y. J. Xu (2018) The GDF11-FTO-PPARγ axis controls the shift of osteoporotic MSC fate to adipocyte and inhibits bone formation during osteoporosis. Biochim. Biophys. Acta. Mol. Basis Dis. 1864: 3644–3654.
Zhang, Y., J. Shao, Z. Wang, T. Yang, S. Liu, Y. Liu, X. Fan, and W. Ye (2015) Growth differentiation factor 11 is a protective factor for osteoblastogenesis by targeting PPARgamma. Gene. 557: 209–214.
Suh, J., N. K. Kim, S. H. Lee, J. H. Eom, Y. Lee, J. C. Park, K. M. Woo, J. H. Baek, J. E. Kim, H. M. Ryoo, S. J. Lee, and Y. S. Lee (2020) GDF11 promotes osteogenesis as opposed to MSTN, and follistatin, a MSTN/GDF11 inhibitor, increases muscle mass but weakens bone. Proc. Natl. Acad. Sci. USA. 117: 4910–4920.
Chen, G., C. Deng, and Y. P. Li (2012) TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int. J. Biol. Sci. 8: 272–288.
Wu, M., G. Chen, and Y. P. Li (2016) TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 4: 16009.
Lee, K. S., S. H. Hong, and S. C. Bae (2002) Both the Smad and p38 MAPK pathways play a crucial role in Runx2 expression following induction by transforming growth factor-β and bone morphogenetic protein. Oncogene. 21: 7156–7163.
Franceschi, R. T. and G. Xiao (2003) Regulation of the osteoblastspecific transcription factor, Runx2: responsiveness to multiple signal transduction pathways. J. Cell. Biochem. 88: 446–454.
Yamaguchi, A., K. Sakamoto, T. Minamizato, K. Katsube, and S. Nakanishi (2008) Regulation of osteoblast differentiation mediated by BMP, notch, and CCN3/NOV. Jpn. Dent. Sci. Rev. 44: 48–56.
Zhang, Y., Y. Wei, D. Liu, F. Liu, X. Li, L. Pan, Y. Pang, and D. Chen (2017) Role of growth differentiation factor 11 in development, physiology and disease. Oncotarget. 8: 81604–81616.
Liu, H., R. Zhang, D. Chen, B. O. Oyajobi, and M. Zhao (2012) Functional redundancy of type II BMP receptor and type IIB activin receptor in BMP2-induced osteoblast differentiation. J. Cell. Physiol. 227: 952–963.
Huntley, R., E. Jensen, R. Gopalakrishnan, and K. C. Mansky (2019) Bone morphogenetic proteins: their role in regulating osteoclast differentiation. Bone Rep. 10: 100207.
Leuderer, H. F., B. Shuting, and G. D. Longmore (2008) The LIM protein LIMD1 influences osteoblast differentiation and function. Exp. Cell Res. 314: 2884–2894.
Yang, W., H. Zheng, Y. Wang, F. Lian, Z. Hu, and S. Xue (2013) Nesprin-1 plays an important role in the proliferation and apoptosis of mesenchymal stem cells. Int. J. Mol. Med. 32: 805–812.
Fitzpatrick, L. A., E. Buzas, T. J. Gagne, A. Nagy, C. Horvath, V. Fererncz, A. Mester, B. Kari, M. Ruan, A. Falus, and J. Barsony (2003) Targeted deletion of histidine decarboxylase gene in mice increases bone formation and protects against ovariectomy-induced bone loss. Proc. Natl. Acad. Sci. USA. 100: 6027–6032.
Qui, X. W., X. L. Ma, X. Lin, F. Zhao, D. J. Li, Z. H. Chen, K. W. Zhang, R. Zhang, P. Wang, Y. Y. Xiao, Z. P. Miao, K. Dang, X. Y. Wu, and A. R. Qian (2020) Deficiency of Macf1 in osterix expressing cells decreases bone formation by Bmp2/Smad/Runx2 pathway. J. Cell Mol. Med. 24: 317–327.
Hu, L., P. Su, C. Yin, Y. Zhang, R. Li, K. Yan, Z. Chen, D. Li, G. Zhang, L. Wang, Z. Miao, A. Qian, and C. J. Xian (2018) Microtubule actin crosslinking factor 1 promotes osteoblast differentiation by promoting β-catenin/TCF1/Runx2 signaling axis. J. Cell. Physiol. 233: 1574–1584.
Huang, G. J., Z. He, and L. Ma (2012) ZFP932 suppresses cellular hedgehog response and Patched1 transcription. Vitam. Horm. 88: 309–332.
Imai, Y., M. Y. Youn, K. Ioune, I. Takada, A. Kouzmenko, and S. Kato (2013) Nuclear receptors in bone physiology and diseases. Physiol. Rev. 93: 481–523.
Mao, M., M. Zhang, A. Ge, X. Ge, R. Gu, C. Zhang, Y. Fu, J. Gao, X. Wang, Y. Liu, and D. Zhu (2018) Granzyme B deficiency promotes osteoblastic differentiation and calcification of vascular smooth muscle cells in hypoxic pulmonary hypertension. Cell Death Dis. 9: 221.
Van Daalen, K. R., J. F. Reijneveld, and N. Bovenschen (2020) Modulation of inflammation by extracellular granzyme A. Front. Immunol. 11: 931.
Lee, B. S. (2018) Myosins in osteoclast formation and function. Biomolecules. 8: 157.
Munson, S., Y. Wang, W. Chang, and D. D. Bikle (2019) Myosin 1a regulates osteoblast differentiation independent of intestinal calcium transport. J. Endocr. Soc. 3: 1993–2011.
Shalev, M. and A. Elson (2019) The roles of protein tyrosine phosphatases in bone-resorbing osteoclasts. Biochim. Biophys. Acta Mol. Cell Res. 1866: 114–123.
Lezcano, V., T. Bellido, L. I. Plotkin, R. Boland, and S. Morelli (2014) Osteoblastic protein tyrosine phosphatases inhibition and connexin 43 phosphorylation by alendronate. Exp. Cell Res. 324: 30–39.
Garten, A., S. Petzold, A. Korner, S. I. Imai, and W. Kiess (2009) Nampt: Linking NAD biology, metabolism and cancer. Trends Endocrinol. Metab. 20: 130–138.
Shigunov, P., J. S. Silveira, C. Kuligovski, A. M. de Aguiar, C. K. Rebelatto, J. A. Moutinho, P. S. Brofman, M. A. Krieger, S. Goldenberg, D. Munroe, A. Correa, and B. Dallagiovanna (2012) PUMILIO-2 is involved in the positive regulation of cellular proliferation in human adipose-derived stem cells. Stem Cells Dev. 21: 217–227.
Zhang, Q., H. Matsui, H. Horiuchi, X. Liang, and K. Sasaki (2016) A-Raf and C-Raf differentially regulate mechanobiological response of osteoblasts to guide mechanical stress-induced differentiation. Biochem. Biophys. Res. Commun. 476: 438–444.
Knierim, E., H. Hirata, N. I. Wolf, S. Morales-Gonzalez, G. Schottmann, Y. Tanaka, S. Rudnik-Schöneborn, M. Orgeur, K. Zerres, S. Vogt, A. van Riesen, E. Gill, F. Seifert, A. Zwirner, J. Kirschner, H. H. Goebel, C. Hubner, S. Stricker, D. Meierhofer, W. Stenzel, and M. Schuelke (2016) Mutations in subunits of the activating signal cointegrator 1 complex are associated with prenatal spinal muscular atrophy and congenital bone fractures. Am. J. Hum. Genet. 98: 473–389.
Chen, Q., K. Liu, A. R. Robinson, C. L. Clauson, H. C. Blair, P. D. Robbins, L. J. Niedernhofer, and H. Ouyang (2013) DNA damage drives accelerated bone aging via an NF-kB-dependent mechanism. J. Bone Miner. Res. 28: 1214–1228.
Acknowledgements
This study was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT, No. 2019R1A2C2002163).
Author information
Authors and Affiliations
Contributions
Study design: SM and JWY. Data collection, analysis and interpretation: SM. Drafting manuscript: JPP. Revising and supervision of the manuscript: JWY. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The cellular in vitro models used in this study were commercially available. We did not use any human or animal samples and therefore did not require approval from the Ethics Committee.
Additional information
Conflict of Interest
The authors declare that they have no conflicts of interest associated with this study.
Data Availability
Data will be made available on reasonable request.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Mukherjee, S., Park, J.P. & Yun, J.W. Carboxylesterase3 (Ces3) Interacts with Bone Morphogenetic Protein 11 and Promotes Differentiation of Osteoblasts via Smad1/5/9 Pathway. Biotechnol Bioproc E 27, 1–16 (2022). https://doi.org/10.1007/s12257-021-0133-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-021-0133-y