Abstract
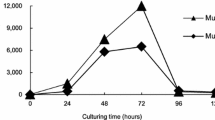
To investigate the effects of bacteria contaminated in microalgal cultivation, several bacteria were isolated from four photobioreactors for Chlorella sp. KR-1 culture. A total of twenty-one bacterial strains isolated from the reactors and identified by 16S rRNA gene sequencing. Six bacteria, which were found from more than two reactors of the four photobioreactors, were introduced into co-culturing experiments with Chlorella sp. KR-1. Then, the bacterial influences on the productivity of microalgal biomass and lipids were assessed in the photoautotrophic- and mixotrophic microalgal cultivation by comparing them with axenic culture of Chlorella sp. KR-1. The results showed that both biomass and lipid production were significantly enhanced under mixotrophic conditions compared to photoautotropic conditions. However, an excess ratio (more than 10%) of bacterial cells to microalgal cells at the initial stage of mixotrophic cultivation has limited the growth of Chlorella sp. KR-1 because of the relatively fast growth of bacteria, especially under mixotrophic conditions. Moreover, it was proven that the strong biofilm formability of Sphingomonas sp. MB6 is the responsible strain to cause the biomass aggregation observed during the early stage of co-culture. The high abundance of Sphingomonas sp. MB6 during early cultivation period shown by qPCR results was also well corresponded with the period shown a strong biofilm formation, which suggested the applicability of qPCR to monitor a specific bacterial group in a microalgal culture.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ma, X., W. Zhou, Z. Fu, Y. Cheng, M. Min, Y. Liu, Y. Zhang, P. Chen, and R. Ruan (2014) Effect of wastewater-borne bacteria on algal growth and nutrients removal in wastewater-based algae cultivation system. Bioresour. Technol. 167: 8–13.
Amin, S. (2009) Review on biofuel oil and gas production processes from microalgae. Energy Convers. Manag. 50: 1834–1840.
Acién, F. G., J. M. Fernández, J. J. Magán, and E. Molina (2012) Production cost of a real microalgae production plant and strategies to reduce it. Biotechnol. Adv. 30: 1344–1353.
Kumar, A., S. Ergas, X. Yuan, A. Sahu, Q. Zhang, J. Dewulf, F. X. Malcata, and H. van Langenhove (2010) Enhanced CO(2) fixation and biofuel production via microalgae: Recent developments and future directions. Trends Biotechnol. 28: 371–380.
Cho, S., N. Lee, S. Park, J. Yu, T. T. Luong, Y. -K Oh, and T. Lee (2013) Microalgae cultivation for bioenergy production using wastewaters from a municipal WWTP as nutritional sources. Bioresour. Technol. 131: 515–520.
Safonova, E. and W. Reisser (2005) Growth promoting and inhibiting effects of extracellular substances of soil microalgae and cyanobacteria on Escherichia coli and Micrococcus luteus. Phycol. Res. 53: 189–193.
Lee, A. K., D. M. Lewis, and P. J. Ashman (2009) Microbial flocculation, a potentially low-cost harvesting technique for marine microalgae for the production of biodiesel. J. Appl. Phycol. 21: 559–567.
Zhou, D., Y. Li, Y. Yang, Y. Wang, C. Zhang, and D. Wang (2015) Granulation, control of bacterial contamination, and enhanced lipid accumulation by driving nutrient starvation in coupled wastewater treatment and Chlorella regularis cultivation. Appl. Microbiol. Biotechnol. 99: 1531–1541.
Muñoz, R. and B. Guieysse (2006) Algal-bacterial processes for the treatment of hazardous contaminants: A review. Water Res. 40: 2799–2815.
Gonzalez, L. E. and Y. Bashan (2000) Increased Growth of the Microalga Chlorella vulgaris when coimmobilized and cocultured in alginate beads with the plant-growth-promoting bacterium Azospirillum brasilense. Appl. Environ. Microbiol. 66: 1527–1531.
Kim, B. -H., R. Ramanan, D. -H. Cho, H. -M. Oh, and H. -S. Kim (2014) Role of Rhizobium, a plant growth promoting bacterium, in enhancing algal biomass through mutualistic interaction. Biomass and Bioenergy 69: 95–105.
Demuez, M., C. González-Fernández, and M. Ballesteros (2015) Algicidal microorganisms and secreted algicides: New tools to induce microalgal cell disruption. Biotechnol. Adv. 33: 1615–1625.
Wang, X., L. Gong, S. Liang, X. Han, C. Zhu, and Y. Li (2005) Algicidal activity of rhamnolipid biosurfactants produced by Pseudomonas aeruginosa. Harmful Algae 4: 433–443.
Najdenski, H. M., L. G. Gigova, I. I. Iliev, P. S. Pilarski, J. Lukavsky, I. V.Tsvetkova, M. S. Ninova, and V. K. Kussovski (2013) Antibacterial and antifungal activities of selected microalgae and cyanobacteria. Int. J. Food. Sci. Technol. 48: 1533–1540.
Azov, Y. (1982) Effect of pH on inorganic carbon uptake in algal cultures inorganic carbon uptake in algal cultures. Appl. Environ. Microbiol. 43: 1300–1306.
More, T. T., J. S. S. Yadav, S. Yan, R. D. Tyagi, and R. Y. Surampalli (2014) Extracellular polymeric substances of bacteria and their potential environmental applications. J. Environ. Manage. 144: 1–25.
Mitra, D., J. van Leeuwen, and B. Lamsal (2012) Heterotrophic/mixotrophic cultivation of oleaginous Chlorella vulgaris on industrial co-products. Algal Res. 1: 40–48.
Wan, M., P. Liu, J. Xia, J. N. Rosenberg, G. A. Oyler, M. J. Betenbaugh, Z. Nie, and G. Qiu (2011) The effect of mixotrophy on microalgal growth, lipid content, and expression levels of three pathway genes in Chlorella sorokiniana. Appl. Microbiol. Biotechnol. 91: 835–844.
Praveenkumar, R., B. Kim, E. Choi, K. Lee, J. -Y. Park, J. -S. Lee, Y. -C. Lee, and Y. -K. Oh (2014) Improved biomass and lipid production in a mixotrophic culture of Chlorella sp. KR-1 with addition of coal-fired flue-gas. Bioresour. Technol. 171: 500–505.
Brennan, L. and P. Owende (2010) Biofuels from microalgae -A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 14: 557–577.
Tsushima, I., T. Kindaichi, and S. Okabe (2007) Quantification of anaerobic ammonium-oxidizing bacteria in enrichment cultures by real-time PCR. Water Res. 41: 785–794.
Fierer, N., J. A. Jackson, R. Vilgalys, and R. B. Jackson (2005) Assessment of soil microbial community structure by use of taxon-specific quantitative PCR assays. Appl. Environ. Microbiol. 71: 4117–4120.
Smith, C. J. and A. M. Osborn (2009) Advantages and limitations of quantitative PCR (Q-PCR)-based approaches in microbial ecology. FEMS Microbiol. Ecol. 67: 6–20.
Lakaniemi, A.-M., V. M. Intihar, O. H. Tuovinen, and J. A. Puhakka (2012) Growth of Chlorella vulgaris and associated bacteria in photobioreactors. Microb. Biotechnol. 5: 449-449.
Cho, S., D. Lee, T. T. Luong, S. Park, Y. K. Oh, and T. Lee (2011) Effects of carbon and nitrogen sources on fatty acid contents and composition in the green microalga, Chlorella sp. 227. J. Microbiol. Biotechnol. 21: 1073–1080.
Cho, S., T. T. Luong, D. Lee, Y. -K. Oh, and T. Lee (2011) Reuse of effluent water from a municipal wastewater treatment plant in microalgae cultivation for biofuel production. Bioresour. Technol. 102: 8639–8645.
Lee, J. -S., D. -K. Kim, J. -P. Lee, S. -C. Park, J. -H. Koh, H. -S. Cho, and S. -W. Kim (2002) Effects of SO2 and NO on growth of Chlorella sp. KR-1. Bioresour. Technol. 82: 1–4.
Na, J. -G., H. S. Lee, Y. -K. Oh, J. -Y. Park, C. H. Ko, S. -H. Lee, K. B. Yi, S. H. Chung, and S. G. Jeon (2011) Rapid estimation of triacylglycerol content of Chlorella sp. by thermogravimetric analysis. Biotechnol Lett. 33: 957–960.
Griffiths, M. J., C. Garcin, R. P. van Hille, and S. T. L. Harrison (2011) Interference by pigment in the estimation of microalgal biomass concentration by optical density. J. Microbiol. Methods 85: 119–123.
Zhou, G. -J., G. -G. Ying, S. Liu, L. -J. Zhou, Z. -F. Chen, and F. -Q. Peng (2014) Simultaneous removal of inorganic and organic compounds in wastewater by freshwater green microalgae. Environ. Sci. Proc. Impacts 16: 2018–2027.
Lee, Y. and H. Shen (2004) 3 Basic Culturing Techniques. p. 40. In: A. Richmond (ed.). Handbook of Microalgal Culture -Biotechnology and Applied Phycology. 1st ed., Blackwell, Oxford, UK.
Rodrigues, L. B., L. R. Dos Santos, V. Z. Tagliari, N. N. Rizzo, G. Trenhago, A. P. de Oliveira, F. Goetz, and V. P. do Nascimento (2010) Quantification of biofilm production on polystyrene by Listeria, Escherichia coli and Staphylococcus aureus isolated from a poultry slaughterhouse. Braz. J. Microbiol. 41: 1082–1085.
O’Toole, G. A. (2011) Microtiter dish biofilm formation assay. J. Vis. Exp. doi: 10.3791/2437
Zhu, F., R. Massana, F. Not, D. Marie, and D. Vaulot (2005) Mapping of picoeucaryotes in marine ecosystems with quantitative PCR of the 18S rRNA gene. FEMS Microbiol. Ecol. 52: 79–92.
Cheirsilp, B. and S. Torpee (2012) Enhanced growth and lipid production of microalgae under mixotrophic culture condition: effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 110: 510–516.
Liang, Y., N. Sarkany, and Y. Cui (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol. Lett. 31: 1043–1049.
Haass, D. and W. Tanner (1974) Regulation of hexose transport in Chlorella vulgaris: Characteristics of induction and turnover. Plant Physiol. 53: 14–20.
Cho, D. -H., R. Ramanan, J. Heo, J. Lee, B. -H. Kim, H. -M. Oh, and H. -S. Kim (2014) Enhancing microalgal biomass productivity by engineering a microalgal-bacterial community. Bioresour. Technol. 175C: 578–585.
Shrout, J. D., D. L. Chopp, C. L. Just, M. Hentzer, M. Givskov, and M. R. Parsek (2006) The impact of quorum sensing and swarming motility on Pseudomonas aeruginosa biofilm formation is nutritionally conditional. Mol. Microbiol. 62: 1264–1277.
Subashchandrabose, S. R., B. Ramakrishnan, M. Megharaj, K. Venkateswarlu, and R. Naidu (2011) Consortia of cyanobacteria/ microalgae and bacteria: Biotechnological potential. Biotechnol. Adv. 29: 896–907.
Watanabe, K., N. Takihana, H. Aoyagi, S. Hanada, Y. Watanabe, N. Ohmura, H. Saiki, and H. Tanaka (2005) Symbiotic association in Chlorella culture. FEMS Microbiol. Ecol. 51: 187–196.
Guo, Z. and Y. W. Tong (2013) The interactions between Chlorella vulgaris and algal symbiotic bacteria under photoautotrophic and photoheterotrophic conditions. J. Appl. Phycol. 26: 1483–1492.
Lee, K., S. Y. Lee, R. Praveenkumar, B. Kim, J. Y. Seo, S. G. Jeon, J. -G. Na, J. -Y. Park, D. -M. Kim, and Y. -K. Oh (2014) Repeated use of stable magnetic flocculant for efficient harvest of oleaginous Chlorella sp. Bioresour. Technol. 167: 284–290.
Balkwill, D. L., J. K. Fredrickson, and M. F. Romine (2006) Sphingomonas and related genera. pp. 605–629. In: E. F. DeLong, S. Lory, E. Stackebrandt, and F. Thompson (eds.). The prokaryotes. 2nd ed. Springer, Berlin Heidelberg, Germany.
Zhu, F., R. Massana, F. Not, D. Marie, and D. Vaulot (2005) Mapping of picoeucaryotes in marine ecosystems with quantitative PCR of the 18S rRNA gene. FEMS Microbiol. Ecol. 52: 79–92.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yun, M., Oh, YK., Praveenkumar, R. et al. Contaminated bacterial effects and qPCR application to monitor a specific bacterium in Chlorella sp. KR-1 culture. Biotechnol Bioproc E 22, 150–160 (2017). https://doi.org/10.1007/s12257-016-0584-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-016-0584-8