Abstract
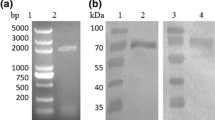
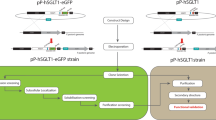
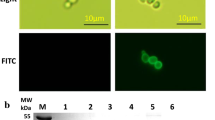
We developed (GLP-1A2G)2-HSA (GGH) analogs that are resistant to degradation and also show high serum glucose-reducing activity in vivo. Five analogs with N-terminal extensions were designed based on the protein GGH. Next, we constructed recombinant plasmids capable of expressing the five analogs in methylotrophic yeast Pichia pastoris GS115. Expression reached 150 mg/L in a small-scale incubation. Fusion proteins were successfully purified from the supernatant using ultrafiltration concentration, affinity absorption chromatography, hydrophobic chromatography, ion exchange chromatography and gel filtration. A single band was observed on SDS-PAGE and the purity was 97%. Activity test results suggested that both A-GGH and G-GGH showed better activity in vitro and that their cAMP levels were significantly increased by 10-fold compared to GGH without N-terminal extension. Additionally, A-GGH efficiently enhanced the glucoselowering effect, which was maintained after the administration for 24 h. A-GGH is a potential drug for treating type 2 diabetes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dou, W. F., J. Y. Lei, L. F. Zhang, Z. H. Xu, Y. Chen, and J. Jin (2008) Expression, purification, and characterization of recombinant human serum albumin fusion protein with two human glucagon-like peptide-1 mutants in Pichia pastoris. Protein Expr. Purif. 61: 45–49.
Burcelin, R. and S. Dejager (2010) GLP-1: What is known, new and controversial in 2010. Diabetes Metab. 36: 503–509.
Li, Y., X. Zheng, L. Tang, W. Xu, and M. Gong (2011) GLP-1 analogs containing disulfide bond exhibited prolonged half-life in vivo than GLP-1. Peptides 32: 1303–1312.
Huang, Y. S., Z. Chen, Y. Q. Chen, G. C. Ma, J. F. Shan, W. Liu, and L. F. Zhou (2008) Preparation and characterization of a novel exendin-4 human serum albumin fusion protein expressed in Pichia pastoris. J. Pept. Sci. 14: 588–595.
Zhou, J., Z. H. Cai, L. Li, C. Kou, and Y. F. Gao (2009) Preparation and PEGylation of exendin-4 peptide secreted from yeast Pichia pastoris. Eur. J. Pharm. Biopharm. 72: 412–417.
Zhou, J., J. Chu, Y. H. Wang, H. Wang, Y. P. Zhuang, and S. L. Zhang (2008) Purification and bioactivity of exendin-4, a peptide analogue of GLP-1, expressed in Pichia pastoris. Biotechnol. Lett. 30: 651–656.
O’Harte, F. P., M. H. Mooney, A. Lawlor, and P. R. Flatt (2000) N-terminally modified glucagon-like peptide-1(7-36) amide exhibits resistance to enzymatic degradation while maintaining its antihyperglycaemic activity in vivo. Biochim. Biophys. Acta 1474: 13–22.
John, H., E. Maronde, W. G. Forssmann, M. Meyer, and K. Adermann (2008) N-terminal acetylation protects glucagon-like peptide GLP-1-(7-34)-amide from DPP-IV-mediated degradation retaining cAMP- and insulin-releasing capacity. Eur. J. Med. Res. 13: 73–78.
Lei, J. Y., L. F. Zhang, and J. Jin (2006) Secretory expression of the fusion protein IFNβ-HSA in Pichia pastoris. China Biotechnol. 26: 13–18.
Kang, H. A., E. S. Choi, W. K. Hong, J. Y. Kim, S. M. Ko, J. H. Sohn, and S. K. Rhee (2000) Proteolytic stability of recombinant human serum albumin secreted in the yeast Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 53: 575–582.
Chung, H. S., J. Y. Oh, S. B. Yoo, S. M. Lee, and H. S. Cho (2011) The N-terminal alanine-extended GLP-1/IgG-Fc fusion protein confers resistance to DPP-IV and reduces serum glucose level in db/db mice. Regul. Pept. 170: 1–3.
Hargrove, D. M., E. S. Kendall, J. M. Reynolds, A. N. Lwin, J. P. Herich, P. A. Smith, B. R. Gedulin, S. D. Flanagan, C. M. Jodka, J. A. Hoyt, K. M. McCowen, D. G. Parkes, and C. M. Anderson (2007) Biological activity of AC3174, a peptide analog of exendin-4. Regul. Pept. 141: 113–119.
Song, G. M., Y. Huan, S. J. Sun, Y. T. Chen, Q. Liu, and Z. F. Shen (2010) Biological activity of EXf, a peptide analogue of exendin-4. Eur. J. Pharmacol. 628: 261–267.
Baggio, L. L., Q. Huang, T. J. Brown, and D. J. Drucker (2004) A recombinant human glucagon-like peptide (GLP)-1-albumin protein (albugon) mimics peptidergic activation of GLP-1 receptordependent pathways coupled with satiety, gastrointestinal motility, and glucose homeostasis. Diabetes. 53: 2492–2500.
Chen, J., G. Bai, Y. Cao, Z. Gao, Q. Zhang, Y. Zhu, and W. Yang (2007) One-step purification of a fusion protein of glucagon-like peptide-1 and human serum albumin expressed in pichia pastoris by an immunomagnetic separation technique. Biosci. Biotechnol. Biochem. 71: 2655–2662.
Zhuo, Z. H., X. Z. Wang, and M. X. Ding (2000) Cell Biology. pp. 158–164. Higher education press, Beijing, China.
Ueda, T., T. Ito, K. Tomita, H. Togame, M. Fumoto, K. Asakura, T. Oshima, S. Nishimura, and K. Hanasaki (2010) Identification of glycosylated exendin-4 analogue with prolonged blood glucose-lowering activity through glycosylation scanning substitution. Bioorg. Med. Chem. Lett. 20: 4631–4634.
Li, C., Y. Huan, N. Shen, L. Ji, S. Sun, S. Liu, Q. Liu, L. Gao, F. Tan, Y. Wang, and Z. Shen (2010) A novel GLP-1 analog, BPI3006, with potent DPP IV resistance and good glucoregulatory effect. Biochem. Biophys. Res. Commun. 400: 563–568.
Green, B. D., M. H. Mooney, V. A. Gault, N. Irwin, C. J. Bailey, P. Harriott, B. Greer, P. R. Flatt, and F. P. O’Harte (2004) Lys9 for Glu9 substitution in glucagon-like peptide-1(7-36)amide confers dipeptidylpeptidase IV resistance with cellular and metabolic actions similar to those of established antagonists glucagon-like peptide-1(9-36)amide and exendin (9-39). Metabolism. 53: 252–259.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dou, W., Feng, J., Zhang, X. et al. Expression, purification, and bioactivity of (GLP-1A2G)2-HSA analogs in Pichia pastoris GS115. Biotechnol Bioproc E 18, 1076–1082 (2013). https://doi.org/10.1007/s12257-013-0356-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-013-0356-7