Abstract
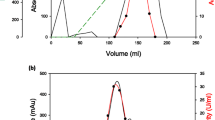
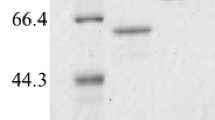
Amylomaltase and transglucosidase were combined to produce long-chain isomaltooligosaccharides (IMOs). IMOs are effective prebiotics that stimulate the growth of healthy bacteria in human intestines and thus promote better overall health. In this study, the p17bAMY amylomaltase was expressed from its gene, which had been directly isolated from soil samples, while transglucosidase was purchased and purified by a gel-filtration column. Crude amylomaltase was purified by heat treatment, Q-, and phenyl-sepharose column. The purified amylomaltase had a molecular weight of 57 kDa. Specificity on the substrates of the amylomaltase was also studied and it was found that this enzyme was able to catalyze transglucosylation activity using substrates G2 to G7. However, G3 was the most preferred substrate for the enzyme. Here, K m-G3 and k cat/K m were 23 mM and 1.72 × 108 mM/min, respectively. Amylomaltase and transglucosidase were tested both alone and in combination on a G3 substrate to study the efficient process for the IMOs production. The obtained products from the enzymatic reactions were monitored using the TLC analytical method and a densitometer. The amylomaltase led to products containing linear maltooligosaccharides, while the transglucosidase produced short-chain IMOs. Interestingly, when amylomaltase and transglucosidase were used in combination, long-chain IMOs with sizes larger than IMO4 were observed under the determined condition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Takaha, T. and S. M. Smith (1999) The functions of 4-alpha-glucanotransferases and their use for the production of cyclic glucans. Biotechnol. Genet. Eng. 16: 257–280.
Pugsley, A. P. and C. Dubreuil (1998) Molecular characterization of malQ, the structural gene for the Escherichia coli enzyme amylomaltase. Mol. Microbiol. 2: 473–479.
Goda, S., O. Eissa, M. Akhtar, and N. Minton (1997) Molecular analysis of a Clostridium butyricum NCIMB 7423 gene encoding 4-alpha-glucanotransferase and characterization of the recombinant enzyme produced in Escherichia coli. Microbiol. 143: 3287–3294.
Jeon, B., H. Taguchi, H. Sakai, T. Ohshima, T. Wakagi, and H. Matsuzawa (1997) 4-alpha-glucanotransferase from the hyperthermophilic archaeon Thermococcus litoralis-enzyme purification and characterization, and gene cloning, sequencing and expression in Escherichia coli. Eur. J. Biochem. 248: 171–178.
Terada, Y., K. Fujii, T. Takaha, and S. Okada (1999) Thermus aquaticus ATCC 33923 amylomaltase gene cloning and expression and enzyme characterization: Production of cycloamylose. Appl. Environ. Microb. 65: 910–915.
Bhuiyan, S. H., M. Kitaoka, and K. Hayashi (2003) A cycloamylose-forming hyperthermostable 4-α-glucanotransferase of Aquifex aeolicus expressed in Escherichia coli. J. Mol. Catal. B: Enzym. 22: 45–53.
Kaper, T., B. Talik, T. J. Ettema, H. Bos, M. J. van der maarel, and L. Dijkhuizen (2005) Amylomaltase of Pyrobaculum aerophilum IM2 produces thermoreversible starch gels. Appl. Environ. Microb. 71: 5098–5106.
Bang, B. Y., H. J. Kim, H. Y. Kim, M. Y. Baik, S. C. Ahn, C. H. Kim, and C. S. Park (2006) Cloning and overexpression of 4-α-glucanotransferase from Thermus brockianus (TBGT) in E. coli. J. Microbiol. Biotechnol. 16: 1809–1813.
Lee, B. H., D. K. Oh, and S. H. Yoo (2009) Characterization of 4-alpha-glucanotransferase from Synechocystis sp. PCC 6803 and its application to various corn starches. N. Biotechol. 26: 29–36.
Srisimarat, W., A. Powviriyakul, J. Kaulpiboon, K. Krusong, W. Zimmermann, and P. Pongsawasdi (2010) A novel amylomaltase from Corynebacterium glutamicum and analysis of the large-ring cyclodextrin products. J. Incl. Phenom. Macro. 70: 369–375.
Lin, T. P. and J. Preiss (1987) Characterization of D-enzyme (4-α-glucanotransferase) in arabidopsis leaf. Plant Physiol. 86: 260–265.
Lee, H. S., J. H. Auh, H. G. Yoon, M. J. Kim, J. H. Park, S. S. Hong, M. H. Kang, T. J. Kim, T. W. Moon, J. W. Kim, and K. H. Park (2002) Cooperative action of alpha-glucanotransferase and maltogenic amylase for an improved process of isomaltooligosaccharide (IMO) production. J. Agr. Food Chem. 50: 2812–2817.
Gibson, G. R. and R. A. Rastal (2006) Prebiotics: Development & Application. pp. 42–46. John Wiley & Sons, Ltd., West Sussex, England.
Goulas, A. K., D. A. Fisher, G. K. Grimble, A. S. Grandison, and R. A. Rastall (2004) Synthesis of isomaltooligosaccharides and oligodextrans by the combined use of dextransucrase and dextranase. Enz. Microb. Technol. 35: 327–388.
Pan, Y. C. and W. C. Lee (2005) Production of high-purity isomalto-oligosaccharides syrup by the enzymatic conversion of transglucosidase and fermentation of yeast cells. Biotechnol. Bioeng. 89: 797–804.
Sawasdee, K. (2012) Expression and characterization of amylomaltase gene cloned directly from soil bacterial DNA. M. Sc. Thesis. Thammasat University, Bangkok, Thailand.
Bradford, M. M. (1976) A rapid and sensitive method for the qualitatively of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Weber, K. and M. Osborn (1975) Proteins and SDS: Molecular weight determination on polyacrylamide gels and related procedures. pp. 179–233. In: H. Neurath, R. L. Hill, and C. Border (eds.). The Proteins. Academic Press, NY, USA.
Robyt, J. F. and R. Mukerjea (1994) Separation and quantitative determination of nanogram quantities of maltodextrins and isomaltodextrins by thin-layer chromatography. Carbohyd. Res. 251: 187–202.
Vetere, A., A. Gamini, C. Campa, and S. Paoletti (2000) Regiospecific transglycolytic synthesis and structural characterization of 6-O-α-glucopyranosyl-glucopyranose (isomaltose). Biochem. Biophys. Res. Commum. 274: 99–104.
Yamamoto, T., T. Unno, Y. Watanabe, M. Yamamoto, M. Okuyama, H. Mori, S. Chiba, and A. Kimura (2004) Purification and characterization of Acremonium implicatum α-glucosidase having regioselectivity for α-1,3-glucosidic linkage. Biochim. Biophys. Acta 1700: 189–198.
Zhou, C., Y. Xue, Y. Zhang, Y. Zeng1, and Y. Ma (2009) Recombinant expression and characterization of Thermoanaerobactertengcongensis thermostable α-glucosidase with regioselectivity for high-yield isomalto oligosaccharides synthesis. J. Microbiol. Biotechnol. 19: 1547–1556.
Kita, A., H. Matsui, A. Somoto, A. Kimura, M. Takata, and S. Chiba (1991) Substrate specificity and subsite affinities of crystalline alpha-glucosidase from Aspergillus niger. Agric. Biol. Chem. 55: 2327–2335.
Lee, M. S., S. K. Cho, H. J. Eom, S. Y. Kim, T. J. Kim, and N. S. Han (2008) Optimized substrate concentrations for production of long-chain isomaltooligosaccharides using dextransucrase of Leuconostoc mesenteroides B-512F. J. Microbiol. Biotechnol. 18: 1141–1145.
Kaneko, T., T. Kohmoto, H. Kikuchi, M. Shiota, and H. Lino (1994) Effect of isomaltooligosaccharides with different degrees of polymerization on human fecal Bifidobacteria. Biosci. Biotechnol. Biochem. 58: 2288–2290.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rudeekulthamrong, P., Sawasdee, K. & Kaulpiboon, J. Production of long-chain isomaltooligosaccharides from maltotriose using the thermostable amylomaltase and transglucosidase enzymes. Biotechnol Bioproc E 18, 778–786 (2013). https://doi.org/10.1007/s12257-012-0777-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-012-0777-8