Abstract
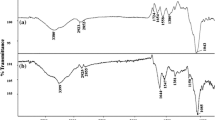
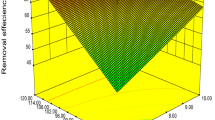
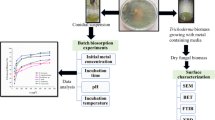
This study investigates the equilibrium, kinetics and thermodynamics of Nickel(II) biosorption from aqueous solution by the fungal mat of Trametes versicolor (rainbow) biomass. The optimum biosorption conditions like pH, contact time, biomass dosage, initial metal ion concentration and temperaturewere determined in the batch method. The biosorbent was characterized by FTIR, SEM and BET surface area analysis. The experimental data were analyzed in terms of pseudo-first-order, pseudo-secondorder and intraparticle diffusion kinetic models, further it was observed that the biosorption process of Ni(II) ions closely followed pseudo-second-order kinetics. The equilibrium data of Ni(II) ions at 303, 313, and 323 K were fitted to the Langmuir and Freundlich isotherm models. Langmuir isotherm provided a better fit to the equilibrium data andthe maximum monolayer biosorption capacity of the T. versicolor(rainbow) biomass for Ni(II) was 212.5 mg/g at pH 4.0. The calculated thermodynamic parameters, ΔG○, ΔH○, and ΔS○, demonstrated that the biosorption of Ni(II) ions onto the T. versicolor (rainbow) biomass was feasible, spontaneous and endothermic at 303 ∼ 323 K. The performance of the proposed fungal biosorbent was also compared with that of many other reported sorbents for Nickel(II) removal and it was observed that the proposed biosorbent is effective in terms of its high sorption capacity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Xuan, Z., Y. Tang, X. Li, Y. Liu, and F. Luo (2006) Study on equilibrium, kinetics and isotherm of biosorption of lead ions on to pretreated chemically modified orange peel. Biochem. Eng. J. 31: 160–164.
Zafar, M. N., R. Nadeem, and M. A. Hanif (2007) Biosorption of nickel from protonated rice bran. J. Hazard. Mater. 143: 478–485.
Padmavathy, V. (2008) Biosorption of nickel ions by baker’s yeast: Kinetic, thermodynamic and desorption studies. Bioresour. Technol. 99: 3100–3109.
Dahiya, S., R. M. Tripathi, and A. G. Hegde (2008) Biosorption of heavy metals and radionuclide from aqueous solutions by pretreated Arca shell biomass. J. Hazard. Mater. 150: 376–386.
Sud, D., G. Mahajan, and M. P. Kaur (2008) Agricultural waste material as potential adsorbent for sequestering heavy metal from aqueous solution-a review. Bioresour. Technol. 99: 6017–6027.
Akar, S. T., T. Akar, Z. Kaynak, B. Anilan, A. Cabuk, O. Tabak, T. A. Demir, and T. Gedikbey (2009) Removal of copper(II) ions from synthetic solution and real wastewater by the combined action of dried Trametes versicolor cells and montmorillonite. Hydrometallurgy 97: 98–104.
Gupta, V. K., A. Rastogi, V. K. Saini, and N. Jain (2006) Biosorption of copper(II) from aqueous solutions by Spirogyra species. J. Colloid Interface Sci. 296: 59–63.
Lodeiro, P., B. Cordero, Z. Grille, R. Herrero, and M. E. Sastre de Vicente (2004) Physicochemical studies of cadmium(II) biosorption by the invasive alga in Europe, Sargassum muticum. Biotechnol. Bioeng. 88: 237–247.
Wang, J. L. and C. Chen (2006) Biosorption of heavy metals by Saccharomyces cerevisiae: A review. Biotechnol. Adv. 24: 427–451.
Cayllahua, J. E. B., R. J. de Carvalho, and M. L. Torem (2009) Evaluation of equilibrium, kinetic and thernodynamic parameters for biosorption of nickel(II) ions onto bacteria strain, Rhodococcus opacus. Miner. Eng. 22: 1318–1325.
Amini, M., H. Younesi, and N. Bahramifar (2009) Biosorption of nickel(II) from aqueous solution by Aspergillus niger: Response surface methodology and isotherm study. Chemosphere 75: 1483–1491.
Fourest, E. and J. C. Roux (1992) Heavy metal biosorption by fungal mycelia byproducts: Mechanisms and influence of pH. Appl. Microbiol. Biotechnol. 37: 399–403.
Kshama, A. S. and V. K. Vaidya (2011) Kinetics and equilibrium studies on biosorption of nickel from aqueous solution by dead fungal biomass of Mucor hiemalis. Chem. Eng. J. 171: 1234–1245.
Yan, G. and T. Viraraghavan (2003) Heavy metal removal from aqueous solution by fungus Mucor rouxii. Water Res. 37: 4486–4496.
Munagapati, V. S., V. Yarramuthi, S. K. Nadavala, S. R. Alla, and K. Abburi (2010) Biosorption of Cu(II), Cd(II) and Pb(II) by Acacia leucocephala bark powder: Kinetics, equilibrium and thermodynamics. Chem. Eng. J. 157: 357–365.
Murphy, V., H. Hughes, and P. McLoughlin (2007) Cu(II) binding by dried biomass of red, green and brown macroalgae. Water Res. 41: 731–740.
Tuzen, M., A. Sari, D. Mendil, O. D. Uluozlu, M. Soylak, and M. Dogan (2009) Characterization of biosorption process of As(III) on green algae Ulothrix cylindricum. J. Hazard. Mater. 165: 566–572.
Ozer, A., D. Ozer, and H. I. Ekiz (2004) The equilibrium and kinetic modeling of the biosorption of copper(II) ions on Cladophora crispate. Adsorption 10: 317–326.
Deng, S. and Y. P. Ting (2005) Characterization of PEI-modified biomass and biosorption of Cu(II), Pb(II) and Ni(II). Water Res. 39: 2167–2177.
Aksu, Z. (2001) Equilibrium and kinetic modeling of cadmium( II) biosorption by C. vulgaris in a batch system: Effect of temperature. Sep. Purif. Technol. 21: 285–294.
Nuhoglu, Y. and E. Malkoc (2009) Thermodynamic and kinetic studies for environmentaly friendly Ni(II) biosorption using waste pomace of olive oil factory. Bioresour. Technol. 100: 2375–2380.
Malkoc, E. and Y. Nuhoglu (2005) Investigations of nickel(II) removal from aqueous solutions using tea factory waste. J. Hazard. Mater. 127: 120–128.
Sari, A., M. Tuzen, D. Citak, and M. Soylak (2007) Adsorption characteristics of Cu(II) and Pb(II) onto expanded perlite from aqueous solution. J. Hazard. Mater. 148: 387–394.
Ozer, A. and D. Ozer (2003) Comparative study of the biosorption of Pb(II), Ni(II) and Cr(VI) ions onto S. cerevisiae: Determination of biosorption heats. J. Hazard. Mater. 100: 219–229.
Lagergren, S. (1898) About the theory of so-called adsorption of soluble substances. K. Sven. Vetenskapsakad. Handl. 24: 1–39.
Ho, Y. S., G. Mckay, D. A. J. Wase, and C. F. Foster (2000) Study of the sorption of divalent metal ion on to peat. Adsorpt. Sci. Technol. 18: 639–650.
Weber, W. J. and J. C. Morris (1963) Kinetics of adsorption on carbon from solution. J. Sanit. Eng. Div. Am. Soc. Civ. Eng. 89: 31–59.
Venkata Mohan, S., S. V. Ramaniah, B. Rajkumar, and P. N. Sarma (2007) Biosorption of fluoride from aqueous phase onto algal Spirogyra Io1 and evaluation of adsorption kinetics. Bioresour. Technol. 98: 1006–1011.
Ozcan, A. S., B. Erdem, and B. Ozcan (2004) Adsorption of acid blue 193 from aqueous solutions onto Na-bentonite and DTMAbentonite. J. Colloid Interface Sci. 280: 44–54.
Meenakshi, S. and N. Viswanathan (2007) Identification of selective ion-exchange resin for fluoride sorption. J. Colloid Interface Sci. 308: 438–450.
Langmuir, I. (1916) The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 38: 2221–2295.
Hanif, M. A., R. Nadeem, H. N. Bhatti, N. R. Ahmad, and T. M. Ansari (2007) Ni(II) biosorption by Cassia fistula (Golden Shower) biomass. J. Hazard. Mater. 139: 345–355.
Reddy, D. H. K., D. K. V. Ramana, K. Seshaiah, and A. V. R. Reddy (2011) Biosorption of Ni(II) from aqueous phase by Moringa oleifera bark, a low cost biosorbent. Desalination 268: 150–157.
Gupta, V. K., A. Rastogi, and A. Nayak (2010) Biosorption of nickel onto treated alga (Oedogonium hatei): Application of isotherm and kinetic models. J. Colloid Interface Sci. 342: 533–539.
Gabr, R. M., S. H. A. Hassan, and A. A. M. Shoreit (2008) Biosorption of lead and nickel by living and non-living cells of Pseudomonas aeruginosa ASU 6a. Int. Biodeter. Biodegr. 62: 195–203.
Sheng, P. X., Y. P. Ting, J. P. Chen, and L. Hong (2004) Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: Characterization of biosorptive capacity and investigation of mechanisms. J. Colloid Interface Sci. 275: 131–141.
Bermudez, Y. G., I. L. R. Rico, O. G. Bermudez, and E. Guibal (2011) Nickel biosorption using Gracilaria caudata and Sargassum muticum. Chem. Eng. J. 166: 122–131.
Malkoc, E. (2006) Ni(II) removal from aqueous solutions using cone biomass of Thuja orientalis. J. Hazard. Mater. 137: 899–908.
Ozer, A., G. Gurbuz, A. Calimli, and B. K. Korbahti (2008) Investigation of nickel(II) biosorption on Enteromorpha prolifera: Optimization using response surface analysis. J. Hazard. Mater. 152: 778–788.
Quintelas, C., Z. Rocha, B. Silva, B. Fonseca, H. Figueiredo, and T. Tavares (2009) Removal of Cd(II), Cr(VI), Fe(III) and Ni(II) from aqueous solutions by an E. Coli biofilm supported on Kaolin. Chem. Eng. J. 149: 319–324.
Ozturk, A., T. Artan, and A. Ayar (2004) Biosorption of nickel(II) and copper(II) ions from aqueous solution by streptomyces coelicolor A3(2). Colloids Surf. B. 34: 105–111.
Ozturk, A. (2007) Removal of nickel from aqueous solution by the bacterium Bacillus thuringiensis. J. Hazard. Mater. 147: 518–523.
Cetinkaya, G. D., Z. Aksu, A. Ozturk, and T. Kutsal (1999) A comparative study on heavy metal biosorption characteristics of some algae. Proc. Biochem. 34: 885–892.
Iqbal, M. and A. Saeed (2007) Production of an immobilized hybrid biosorbent for the sorption of Ni(II) from aqueous solution. Proc. Biochem. 42: 148–157.
Freundlich, H. M. F. (1906) Uber die adsorption in losungen. Z. Phys. Chem. 57: 385–470.
Viswanathan, N., C. S. Sundaram, and S. Meenakshi (2009) Sorption behaviour of fluoride on carboxylated cross-linked chitosan beads. Colloids Surf. B. 68: 48–54.
Srivastava, V. C., M. M. Swamy, I. D. Mall, B. Prasad, and I. M. Mishra (2006) Adsorptive removal of phenol by bagasse fly ash and activated carbon: Equilibrium, kinetics and thermodynamics. Colloids Surf. A. 272: 89–104.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subbaiah, M.V., Yun, Y.S. Biosorption of Nickel(II) from aqueous solution by the fungal mat of Trametes versicolor (rainbow) biomass: equilibrium, kinetics, and thermodynamic studies. Biotechnol Bioproc E 18, 280–288 (2013). https://doi.org/10.1007/s12257-012-0401-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-012-0401-y