Abstract
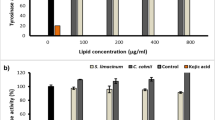
Tyrosinase is a key enzyme in the biosynthesis of melanin, and the use of inhibitors against tyrosinase can prevent hyperpigmentation by inhibiting enzymatic oxidation. However, the current use of tyrosine inhibitors is limited by their low activities and high toxicities. The aim of the present research was to develop novel whitening agents, or tyrosinase-targeted medicine, from a submerged culture of the fungus Ganoderma lucidum. Methyl lucidenate F was isolated from the ethanol-soluble-acidic components (ESACs) of G. lucidum, with the structure of ESACs elucidated via UV, LC-MS, and 13C-NMR spectral analysis. The tyrosinase inhibitory activity was measured using catechol as a substrate. Methyl lucidenate F displayed uncompetitive inhibition of the potato tyrosinase activity, for which Lineweaver-Burk plots revealed a maximum reaction rate (V max) of 0.4367/min, Michaelis constant (K m) of 6.765 mM and uncompetitive inhibition constant (K i) of 19.22 μM. Meanwhile, methyl lucidenate F (tetra cyclic triterpenoid) exhibited high tyrosinase inhibitory activity, with an IC50 of 32.23 μM. These results suggest that methyl lucidenate F may serve as a potential candidate for skin-whitening agents.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Espin, J. C. and H. J. Wichers (2001) Effect of captopril on mushroom tyrosinase activity in vitro. Biochim. Biophys. Acta. 1544: 289–300.
Murray, C., Y. D’Intino, R. MacCormick, B. Nassar, and N. Walsh (1999) Melanosis in association with metastatic malignant melanoma: Report of a case and a unifying concept of pathogenesis. Am. J. Dermatopathol. 21: 28–30.
Blaszczyk, W., M. L. Arning, K. P. Hoffmann, and J. T. Epplen (2005) A Tyrosinase missense mutation causes albinism in the Wistar rat. Pigment. Cell Res. 18: 144–145.
Asanuma, M., I. Miyazaki, and N. Ogawa (2003) Dopamine-or L-DOPA-induced neurotoxicity: The role of dopamine quinone formation and tyrosinase in a model of Parkinson’s disease. Neurotox. Res. 5: 165–176.
Ohad, N., M. Ramadan, and K. Soliman (2004) Chalcones as potent tyrosinase inhibitors: the effect of hydroxyl positions and numbers. Phytochem. 65: 1389–1395.
Lin, S. B., C. H. Li, S. S. Lee, and L. S. Kan (2003) Triterpeneenriched extracts from Ganoderma lucidum inhibit growth of hepatoma cells via suppressing protein kinase C, activating mitogen-activated protein kinases and G2-phase cell cycle arrest. Life Sci. 72: 2381–2390.
El-Mekkawy, S., M. R. Meselhy, N. Nakamura, Y. Tezuka, M. Hattori, N. Kakiuchi, K. Shimotohno, T. Kawahata, and T. Otake (1998) Anti-HIV-1 and anti-HIV-1-protease substances from Ganoderma lucidum. Phytochem. 49: 1651–1657.
Lin, Z. B. and H. N. Zhang (2004) Anti-tumor and immunoregulatory activities of Ganoderma lucidum and its possible mechanisms. Acta Pharmacol. Sin. 25: 1387–1395.
Hajjaj, H., C. Mace, M. Roberts, P. Niederberger, and L. B. Fay (2005) Effect of 26-oxygenosterols from Ganoderma lucidum and their activity as cholesterol synthesis inhibitors. Appl. Environ. Microbiol. 71: 3653–3658.
Hikino, H., M. Ishiyama, Y. Suzuki, and C. Konno (1989) Mechanisms of hypoglycemic activity of ganoderan B: A glycan of Ganoderma lucidum fruit bodies. Planta Med. 55: 423–428.
Tian, Y. P. and K. C. Zhang (2005) Purification and characterization of a novel proteinase A inhibitor from Ganoderma lucidum by submerged fermentation. Enz. Microb. Tech. 36: 357–361.
Schmidt, H. (1990) Quantitative determination of phenoloxidase (EC 1.14.18.1): Spectrophotometry measurement of the activity of mushroom and potato tyrosinase. Dermatol. Monatsschr. 176: 757–761.
Zhu, G. L., H. W. Zhong, and A. Q. Zhang (1990) Experiment of botanic physiology. pp. 37–39. Peking University Press, Beijing, China.
Baurin, N., E. Arnoult, T. Scior, Q. T. Do, and P. Bernard (2002) Preliminary screening of some tropical plants for anti-tyrosinase activity. J. Ethnopharmacol. 82: 155–158.
Boh, B., D. Hodžar, D. Dolnièar, M. Beroviè, and F. Pohleven (2000) Isolation and quantification of triterpenoid acids from Ganoderma applanatum of istrian origin. Food Technol. Biotechnol. 38: 11–18.
Lin, Z. B. (2001) The present-day study of Ganoderma lucidum. 2nd ed., pp. 157–187. Beijing Medicinal University Press, Beijing, China.
Um, S. J., M. S. Park, and S. H. Park (2003) Synthesis of new glycyrrhetinic acid (GA) derivatives and their effects on tyrosinase activity. Bioorg. Med. Chem. 11: 5345–5352.
Lu, S., Y. Hu, X. Liu, J. Li, J. Z. Diao, W. Zhao, B. Shu, Y. Ye, and K. W. Liu (2005) Study on the inhibition of kojic acid on potato tyrosinase. Chem. Res. Appl. 17: 729–732.
Khatib, S., O. Nerya, R. Musa, M. Shmuel, S. Tamir, and J. Vaya (2004) Chalcones as potent tyrosinase inhibitors: The importance of a 2,4-substituted resorcinol moiety. Bioorg. Med. Chem. 13: 433–441.
Nick, A., A. D. Wright, O. Sticher, and T. Rali (1994) Antibacterial triterpenoid acids from Dillenia papuana. J. Nat. Prod. 57: 1245–1250.
Reddy, V. L., V. Ravikanth, T. P. Rao, P. V. Diwan, and Y. Venkateswarlu (2001) A new triterpenoid from the fern Adiantum lunulatum and evaluation of antibacterial activity. Phytochem. 56: 173–175.
Gulcin, I., V. Mshvildadze, A. Gepdiremen, and R. Elias (2006) The antioxidant activity of a triterpenoid glycoside isolated from the berries of Hedera colchica: 3-O-(beta-D-glucopyranosyl)-hederagenin. Phytother. Res. 20: 130–134.
Sultanova, N., T. Makhmoor, A. Yasin, Z. A. Abilov, V. B. Omurkamzinova, R. Atta ur, and M. I. Choudhary (2004) Isotamarixen-a new antioxidant and prolyl endopeptidase-inhibiting triterpenoid from Tamarix hispida. Planta Med. 70: 65–67.
Author information
Authors and Affiliations
Corresponding author
Additional information
equal contribution to this research.
Rights and permissions
About this article
Cite this article
Zhang, L., Ding, Z., Xu, P. et al. Methyl lucidenate F isolated from the ethanol-soluble-acidic components of Ganoderma lucidum is a novel tyrosinase inhibitor. Biotechnol Bioproc E 16, 457–461 (2011). https://doi.org/10.1007/s12257-010-0345-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-010-0345-z