Abstract
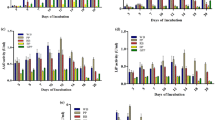
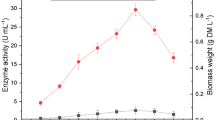
Six agro-industrial wastes were evaluated as a support for ligninolytic enzyme production by the white-rot fungus Lentinus polychrous Lév. under solid-state fermentation. Enzyme production was markedly different according to the substrate used. Rice bran (RB) yielded the highest laccase activity of 1,449 U/L (after 21 days of culture) with specific activity of 4.4 U/g substrate. Rice bran supplemented with rice husk (RH) (2:1 by wt) showed high laccase activity of 1,425 U/L with specific activity of 10.0 U/g substrate (after 17 days of culture). The crude enzyme of the RH-RB culture also contained manganese peroxidase (MnP) and manganese-independent peroxidase (MIP) activities in relative proportions of 1.9:1.4:1 of laccase:MnP:MIP, respectively. Zymogram studies showed the same isoenzyme pattern with these ligninolytic enzymes. The high enzyme production level and low substrate cost of SSF-L. polychrous Lév. suggest that it has potential for industrial applications. Our studies showed that the crude enzyme from this culture exhibited in vitro decolorization of Indigo Carmine. The highest efficiency of dye decolorization was observed under alkaline conditions (pH 9.0) at an initial dye concentration of 10 mg/L. The rather high pH conditions and high efficiency in Indigo Carmine decolorization make the enzyme further interest for the applications in treatment of waste water from the textile industry, which contains synthetic dyes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Solomon, E. I., U. M. Sundaram, and T. E. Machonkin (1996) Multicopper oxidases and oxygenases. Chem. Rev. 96: 2563–2605.
Majcherczyk, A., C. Johannes, and A. Hüttermann (1999) Oxidation of aromatic alcohols by laccase from Trametes versicolor mediated by the 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulphonic acid) cation radical and dication. Appl. Microbiol. Biotechnol. 51: 267–276.
Camarero, S., D. Ibarra, A. T. Martínez, J. Romero, A. Gutiérrez, and J. C. DelRío (2007) Paper pulp delignification using laccase and natural mediators. Enz. Microb. Technol. 40: 1264–1271.
Gutierrez, A., J. Rencoret, D. Ibarra, S. Molina, S. Camarero, J. Romero, J. C. DelRio, and A. T. Martinez (2007) Removal of lipophilic extractives from paper pulp by laccase and lignin-derived phenols as natural mediators. Environ. Sci. Technol. 41: 4124–4129.
Dodor, D. E., H. M. Hwang, and S. I. N. Ekunwe (2004) Oxidation of anthracene and benzo(a)pyrene by immobilized laccase from Trametes versicolor. Enz. Microb. Tech. 35: 210–217.
Camarero, S., D. Ibarra, M. J. Martínez, and A. T. Martínez (2005) Lignin-derived compounds as efficient laccase mediators for decolourization of different types of recalcitrant dyes. Appl. Environ. Microbiol. 71: 1775–1784.
Wu, Y., Y. Teng, Z. Li, X. Liao, and Y. Luo (2008) Potential role of polycyclic aromatic hydrocarbons (PAHs) oxidation by fungal laccase in the remediation of an aged contaminated soil. Soil Biol. Biochem. 40: 789–796.
Ncanana, S. and S. Burton (2007) Oxidation of 8-hydroxyquinoline catalyzed by laccase from Trametes pubescens yields an antioxidant aromatic polymer. J. Mol. Catal. B: Enzym. 44: 66–71.
Santhiago, M. and I. C. Vieira (2007) L-Cysteine determination in pharmaceutical formulations using a biosensor based on laccase from Aspergillus oryzae. Sens. Actuators B: Chem. 128: 279–285.
Wang, J., H. X. Wang, and T. B. Ng (2007) A peptide with HIV-1 reverse transcriptase inhibitory activity from the medicinal mushroom Russula paludosa. Peptides 28: 560–565.
Moldes, D., M. Lorenzo, and M. A. Sanroman (2004) Different proportions of laccase isoenzymes produced by submerged cultures of Trametes versicolor grown on lignocellulosic wastes. Biotechnol. Lett. 26: 327–330.
Pandey, A., P. Selvakumar, C. R. Soccol, and P. Nigam (1999) Solid-state fermentation for the production of industrial enzymes. Curr. Sci. 77: 149–162.
Couto, S. R. and M. A. Sanromán (2005) Application of solid-state fermentation to ligninolytic enzyme production. Biochem. Eng. J. 22: 211–219.
Meza, J. C., A. Lomascolo, L. Casalot, J. C. Sigoillot, and R. Auria (2005) Laccase production by Pycnoporus cinnabarinus grown on sugar-cane bagasse: influence of ethanol vapours as inducer. Proc. Biochem. 40: 3365–3371.
Niladevi, K. N., R. K. Sukumaran, and P. Prema (2007) Utilization of rice straw for laccse production by Streptomyces psammoticus in solid-state fermentation. J. Ind. Microbiol. Biotechnol. 34: 665–674.
Silva, E. M., A. Machuca, and A. M. F. Milagres (2005) Evaluating the growth and enzyme production from Lentinula edodes strains. Proc. Biochem. 40: 161–164.
Wang, S.-L., Y.-H. Yen, I.-L. Shih, A. C. Chang, W.-T. Chang, W.-C. Wu, and Y.-D. Chai (2003) Production of xylanases from rice bran by Streptomyces actuosus A-151. Enz. Microb. Technol. 33: 917–925.
Singhania, R. R., A. K. Patel, C. R. Soccol, and A. Pandey (2009) Recent advances in solid-state fermentation. Biochem. Eng. J. 44: 13–18.
Wesenberg, D., I. Kyriakides, and S. N. Agathos (2003) White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol. Adv. 22: 161–187.
Gregori, A., M. Švagelj, B. Pahor, M. Berovič, and F. Pohleven (2008) The use of spent brewery grains for Pleurotus ostreatus cultivation and enzyme production. New Biotechnology 25: 157–161.
Shrivastava, R., V. Christian, and B. R. M. Vyas (2005) Enzymatic decolorization of sulfonphthalein dyes. Enz. Microb. Technol. 36: 333–337.
Singh, D. and S. Chen (2008) The white-rot fungus Phanerochaete chrysosporium: conditions for the production of lignin-degrading enzymes. Appl. Microbiol. Biotechnol. 81: 399–417.
Asses, N., L. Ayed, H. Bouallagui, I. Ben Rejeb, M. Gargouri, and M. Hamdi (2009) Use of Geotrichum candidum for olive mill wastewater treatment in submerged and static culture. Biores. Technol. 100: 2182–2188.
Ruksawong, P. and T. W. Flegel (2001) Thai mushrooms and other fungi. p. 198. National Center for Genetic Engineering and Biotechnology, Bangkok, Thailand.
Klinhom, W. and U. Klinhom (2005) Fifty seven medicinal mushrooms of northeastern, Thailand. pp. 136–137. Thai Holistic Health Foundation, Bangkok, Thailand.
Klinhom, U., W. Klinhom, and W. Kanchanamayoon (2003) Mushrooms and traditional knowledge in northeastern Thailand. Proceedings of 2nd International Conference on Medicinal Mushroom and the International Conference on Biodiversity and Bioactive Compounds. July 17–19. Pattaya, Thailand.
Pukahuta, C., P. Suwanarit, E. Shinagawa, H. Hoshida, and Y. Nishizawa (2004) Combination of laccase, xylanase, and cellulase in lignocellulose degradation by white rot fungi, Lentinus polychrous Lév. and L. squarrosulus Mont. Kasetsart J. (Nat. Sci.) 38: 65–73.
Khammuang S. and R. Sarnthima (2007) Laccase from spent mushroom compost of Lentinus polychrous Lev. and its potential for remazol brilliant blue R decolorization. Biotechnology 6: 408–413.
Shin, K. S. and Y. J. Lee (2000) Purification and characterization of a new member of the laccase family from the white-rot basidiomycete Coriolus hirutus. Arch. Biochem. Biophys. 384: 109–115.
Leonowicz, A. and K. Grzywnowicz (1981) Quantitative estimation of laccase forms in some white-rot fungi using syringaldazine as a substrate. Enz. Microbiol. Technol. 3: 55–58.
Collins, P. J., M. J. J. Kotterman, J. A. Field, and A. D. W. Dobson (1996) Oxidation of anthracene and benzo(a) pyrene by laccases from Trametes versiocolor. Appl. Environ. Microbiol. 62: 4563–4567.
Archibald, F. S. (1992) A new assay for lignin-type peroxidases employing the dye Azure B. Appl. Environ. Microbiol. 58: 3110–3116.
Ngo, T. T. and H. M. Lenhoff (1980) A sensitive and versatile chromogenic assay for peroxidase and peroxidasecoupled reactions. Anal. Biochem. 105: 389–397.
Sarnthima, R. and S. Khammuang (2008) Evaluation of dyes decolourization by the crude enzyme from Pleurotus sajor-caju grown on sorghum seed media. Pakistan J. Biol. Sci. 11: 62–67.
Bradford, M. M. (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal. Biochem. 72: 248–254.
Rossi, I. H., A. C. Monteiro, J. O. Machado, and J. C. Barbosa (2003) Supplementation of sugarcane bagasse with rice bran and sugarcane molasses for shiitake (Lentinula edodes) spawn production. Braz. J. Microbiol. 34: 61–65.
Tong, P., Y. Hong, Y. Xiao, M. Zhang, X. Tu, and T. Cui (2007) High production of laccase by a new basidiomycete, Trametes sp. Biotechnol. Lett. 29: 295–301.
Winquist, E., U. Moilanen, A. Mettälä and A. Hatakka (2008) Production of lignin modifying enzymes on industrial waste material by solid-state cultivation of fungi. Biochem. Eng. J. 42: 128–132.
Akavia, E., A. Beharav, S. P. Wasser, and E. Nevo (2009) Disposal of agro-industrial by-products by organic cultivation of the culinary and medicinal mushroom Hypsizygus marmoreus. Waste Management 29: 1622–1627.
Elisashvili, V., M. Penninckx, E. Kachlishvili, N. Tsiklauri, E. Metreveli, T. Kharziani, and G. Kvesitadze (2008) Lentinus edodes and Pleurotus species lignocellulolytic enzymes activity in submerged and solid-state fermentation of lignocellulosic wastes of different com-position. Biores. Technology. 99: 457–462.
Baldrian, P. and L. Gabriel (2003) Lignocellulose degradation by Pleurotus ostreatus in the presence of cadmium. FEMS Microbiol. Lett. 220: 235–240.
Reddy, G. V., P. R. Babu, P. Komaraiah, K. R. R. M. Roy, and I. L. Kothari (2003) Utilization of banana waste for the production of lignolytic and cellulolytic enzymes by solid substrate fermentation using two Pleurotus species (P. ostreatus and P. sajor-caju). Proc. Biochem. 38: 1457–1462.
Singh, G., N. Capalash, R. Goel, and P. Sharma (2007) A pH-stable laccase from alkali-tolerant γ-proteobacterium JB: Purification, characterization, and indigo carmine degradation. Enz. Microb. Tech. 41: 794–799.
Basto, C., C. J. Silva, G. Gübitz, and A. Cavaco-Paulo (2007) Stability and decolourization ability of Trametes villosa laccase in liquid ultrasonic fields. Ultrason. Sonochem. 14: 355–362.
Couto, S. R., M. A. Sanromán, D. Hofer, and G. M. Gübitz (2004) Stainless steel sponge: A novel carrier for the immobilisation of the white-rot fungus Trametes hirsuta for decolourization of textile dyes. Biores. Technol. 95: 67–72.
Johannes C. and A. Majcherczyk (2000) Natural mediators in the oxidation of polycyclic aromatic hydrocarbons by laccase-mediator systems. Appl. Environ. Microbiol. 66: 524–528.
Mechichi, T., N. Mhiri, and S. Sayadi (2006) Remazol Brilliant Blue R decolourization by the laccase from Trametes trogii. Chemosphere. 64: 998–1005.
Couto, S. R., E. Rosales, and M. A. Sanroman (2006) Decolourization of synthetic dyes by Trametes hirsuta in expanded-bed reactors. Chemosphere 62: 1558–1563.
Jadhav, S. U., S. D. Kalme, and S. P. Govindwar (2008) Biodegradation of methyl red by Galactomyces geotrichum MTCC 1360. Int. Biodeter. Biodegr. 62: 135–142.
Nyanhongo, G. S., J. Gomes, G. M. Gübitz, R. Zvauya, J. Read, and W. Steiner (2002) Decolorization of textile dyes by laccase from a newly isolated strain of Trametes modesta. Water Res. 36: 1449–1456.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarnthima, R., Khammuang, S. & Svasti, J. Extracellular ligninolytic enzymes by Lentinus polychrous Lév. under solid-state fermentation of potential agro-industrial wastes and their effectiveness in decolorization of synthetic dyes. Biotechnol Bioproc E 14, 513–522 (2009). https://doi.org/10.1007/s12257-008-0262-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0262-6