Abstract
Synchronous multiple gastric cancer (SMGC) was a special type of gastric cancer with relatively low incidence. This article was designed to demonstrate that the total tumor volume (TTV) should be treated as an important prognostic factor in SMGC patients with curative gastrectomy. This study retrospective analyzed 140 SMGC patients who received curative gastrectomy between December 2004 and December 2014 in our hospital. Clinicopathological features, preoperative evaluation, surgical treatment, and outcome parameters were reviewed and analyzed. This study applied univariate and multivariate analyses to identify the most significant prognostic factors. In the univariate analysis, the TTV, pTTVNM, pN stage, pT of main tumor were all significant prognostic factors in SMGC patients (all P < 0.05). In the multivariate analysis, pN stage, TTV and pTTVNM were confirmed to be independent prognostic factors (all P < 0.05). In the comparison of survival analysis, the pTTVNM stage system (P < 0.05) was superior to the pTNM stage system (P > 0.05) in SMGC patients. In conclusion, the TTV should be considered as an independent prognostic factor in overall survival in SMGC patients who received curative gastrectomy. The pTTVNM stage should be recommended as a suitable staging system for SMGC patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) is the second leading cancer in the world, and the prognosis of advanced GC remains poor [1]. Synchronous multiple gastric cancer (SMGC) was a special type of gastric cancer. In previous studies, the incidence of SMGCs was reported to be 5–10% [2]. The criteria for SMGCs were defined as follows: (1) each tumor must be histopathologic confirmed by pathology reports; (2) tumors are separated from each other in the stomach; (3) no tumor is result from extension or metastasis of another tumor [3].
In recent years, the American Joint Committee on Cancer (AJCC)/ Union of International Cancer Control (UICC) tumor-node-metastasis (TNM) staging system was widely used in gastric cancer. However, in patients with SMGCs, some studies suggested that the traditional TNM classification for GC may not suitable [4]. In several types of malignant tumors, such as hepatocellular carcinoma, tumor volume (TV) was proved to be an important prognostic factor [5]. However, the relationship between TV and overall survival in SMGC patients was still unknown. This study was designed to elucidate the prognostic significance of TV in SMGC patients after curative surgery.
Materials and Methods
Patients
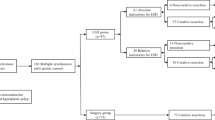
A total of 2360 patients with gastric cancer who underwent gastrectomy at Shandong University Qilu Hospital during December 2004 and December 2014 were enrolled into this study. The inclusion criteria were as follows: (1) patients who had more than one primary gastric tumor, with each tumor been identified as gastric adenocarcinoma by histopathological examination; (2) complete clinicopathological records including follow-up data; (3) received curative resection with D2 lymphadenectomy. The exclusion criteria were: (1) patients with distant metastasis before or during surgery; (2) patients with incomplete clinicopathological information; (3) patients with Borrmann type IV GC (diffuse infiltration type).
In a resected specimen, tumor with the most advanced depth of invasion was defined as the main tumor, while the other was defined as accessory tumor [6]. The tumor volume (TV) equaled to π*(tumor diameter/2)2*tumor invasion depth. The tumor diameter was the maximum diameter of the tumor. The tumor invasion depth was the maximum invasion depth of tumor. The tumor diameter and invasion depth were recorded in pathology reports after surgery. The total tumor volume (TTV) equaled to the sum of each TV in SMGC patients.
Follow-up
All the patients were followed-up once per year or until death by telephone, e-mail or outpatient examination. The CT or MRI scan, endoscopy examination and laboratory tests were performed at every outpatient visit. The median follow-up time was 76 (range: 50–112) months. The last follow-up date was November 25, 2019. The overall survival (OS) was calculated from the date of surgery until the final date of follow-up or death.
Statistical Analysis
The Life Tables method analyzed OS rates according to the new subgroup of TTV. The univariate and multivariate analyses were applied to identify the most significant classification correlated with prognosis. To determine the appropriate cutoffs for the TTV, the cut-point survival analysis was adopted. The Kaplan-Meier method was used to calculate overall survival curves based on the length of time between primary surgical treatment and final follow-up or death. The log-rank test was used to assess statistical differences between curves. Cox proportional hazards regression model was used to identify independent prognostic factors. P < 0.05 was considered statistically significant. To decide the most appropriate cut-offs of TTV based on these SMGCs patients, we recomputed the likelihood associated with all possible pairs of TTV cut-offs ranging from 1 to 50 (cm3) at intervals of 1. The statistical analysis was performed using SAS 9.4 (SAS Institute Inc., Kerry, USA).
Results
Clinicopathological Outcomes
One hundred and forty SMGCs patients were included in this study based on these criteria. Among these patients, 86 (61.4%) patients were male and 54 (38.6%) were female. The median age of these patients was 61.2 years (range, 41–91 years). All 140 SMGC patients had more than one primary tumor in the stomach, with a total of 287 tumors. Among these tumors, 86 (30.0%), 71 (24.7%), and 130 (45.3%) were located in the lower, middle, and upper thirds of the stomach, respectively. Poor differentiation and signet ring cell histology (182 tumors, 63.4%) was more than high and middle differentiation histology (105 tumors, 36.6%). The average total tumor volume (TTV) of each patient was 7.1 (range, 0.4–25.5) cm3.
All patients received curative gastrectomy with D2 lymphadenectomy. A total of 58 (41.4%) patients underwent subtotal gastrectomy, while 82 (58.6%) patients underwent total gastrectomy. The median number of lymph nodes resected per patient was 21 (range, 11–54) by histopathological examination after surgery. A total of 101 patients with tumor stage of II/ III received adjuvant chemotherapy after surgery. In these patients, 53 patients received XELOX (capecitabine plus oxaliplatin), 38 patients received SOX (S-1 plus oxaliplatin) and 10 patients received other regiments (S-1/ mFOLFOX/ DCF/ ECF). The patient characteristics were summarized in Table 1.
Univariate and Multivariate Analyses of Prognostic Factors
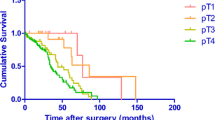
The results of cut-point survival analysis demonstrated that the appropriate cut-offs of TTV between resulting subgroups were 1.0, 7.0 and 12.0 cm3. According to this stratified standard, the 140 patients were classified into 4 groups: 57 (40.7%) patients were TTV1, 37 (26.4%) patients were TTV2, 30 (21.4%) patients were TTV3, 16 (11.5%) patients were TTV4. In the univariate analysis, these 4 groups which classified according to the TTV classification were significantly correlated with prognosis (P < 0.001). To further study the prognostic factors of SMGC, we introduced the TTV to the 8th AJCC/UICC pTNM staging system by replace the pT stage of main tumor with TTV. Other variables, including the pT stage of main tumor (P = 0.006), pN stage (P < 0.010) and pTTVNM (P < 0.001) were all significant predictors of survival. We did observe the improvement of median survival in patients who received adjuvant chemotherapy (54 months vs. 40 months). Patients who received chemotherapy of XELOX regiment showed better survival than patients who did not receive adjuvant chemotherapy (P < 0.05). However, there was no statistical significance between patients who received SOX or other regiments (S-1/ mFOLFOX/ DCF/ ECF) and patients who did not receive adjuvant chemotherapy (P > 0.05). Survival curves of SMGC patients according to TTV, pT, pN and pTTVNM were shown in Figs. 1a, b and c and 2a.
Survival curve according to TTV classification, pT stage and pN stage after surgery. a. There were statistically significant differences between patients with TTV2, TTV3 and TTV4 (P = 0.015, P = 0.021, P < 0.001, respectively) b. There were statistically significant differences between patients with pT2, pT3 and pT4 (P = 0.386, P = 0.020, P = 0.006, respectively). c. Patients with pN1, pN2 and pN3 were significantly associated with different survival outcomes (P = 0.351, P = 0.018, P < 0.001, respectively)
Comparison of survival analysis between the 8th AJCC/ UICC pTNM classification and our suggested pTTVNM classification in SMGC patients. a. Patients classified according to the pTTVNM classification were significantly correlated with prognosis (P = 0.002, P < 0.001, respectively). b. Patients classified according to the 8th AJCC/UICC pTNM classification were not correlated with prognosis (P > 0.05, P = 0.060, respectively)
In the multivariate survival analysis, the TTV classification, pN stage and pTTVNM stage were confirmed to be independent prognostic factors (All P < 0.05). However, the pT stage was not independent prognostic factor (P > 0.05). The detailed data of univariate and multivariate analyses were shown in Table 2.
Comparison of Survival Analysis Between the TNM Stage System and TTVNM Stage System
Moreover, we directly compared the differences in prognostic prediction between the 8th edition pTNM classification and the pTTVNM classification. Detailed survival differences between the traditional pTNM staging system and our suggested pTTVNM staging system were showed in Table 3. The results indicated that the pTTVNM categorization system significantly predicted survival in patients with SMGCs (P < 0.05) (Fig. 2a). However, the 8th pTNM staging system could not (P > 0.05) (Fig. 2b).
Discussion
Synchronous multiple gastric carcinomas (SMGCs) were defined as two or more cancerous tumors in one stomach [7]. So far, the etiology of SMGC is still not clear. The SMGC can occur in any part of the stomach, but most occur in the antrum or cardia of gastric wall. With the improvement of endoscopic and imaging diagnostic techniques, the detection rate of SMGC is increasing. The incidence of SMGC reported in the literature is about 0.4% -2.4%, accounting for about 1.2% − 10.7% of all gastric cancer [8]. This study shows that SMGC accounts for about 5.9% (140/ 2360) of all gastric cancer patients. Some studies suggested that SMGC were more common in early gastric cancer. They compared multiple primary early gastric cancer with single early gastric cancer, found that there was no significant difference in prognosis [9]. However, with the increasing application of endoscopic submucosal dissection (ESD) in early gastric cancer in recent years, surgically resected SMGC gradually transformed into advanced gastric cancer [10]. In our study, we included 80.7% (113/140) cases of advanced gastric cancer (T stage: 2–4 stages of primary tumor) in surgical cases.
The main treatment for SMGC is surgical resection. The extent of resection depends on the location of each primary lesion and the number of primary tumors [11]. Some early studies recommended total gastrectomy for SMGC to avoid residual lesions, however, with the improvement of preoperative examination, prophylactic total gastrectomy is unnecessary [12]. However, patients with gastric cancer should still be aware of possible missed lesions. Detailed preoperative gastroscopy is an effective mean to detect SMGC. Other imaging examinations, such as nuclear magnetic resonance (MRI), contrast-enhanced computed tomography (CT) and upper gastrointestinal angiography can also help to detect missed lesions [13]. The combination of multiple examinations can effectively improve the diagnostic rate of SMGC. Due to the non-specific distribution of SMGC in the stomach, the whole stomach should be carefully checked during surgery to avoid missed lesions. Postoperative regular endoscopic fiberscope is an effective mean to detect tumor recurrence.
For decades, tumor invasion depth (pT) has been considered as one of the most important pathological factors. However, our study suggested that the pT stage may be not suitable in patients with SMGC (P > 0.05). The tumor volume (TV) was found to be an important prognostic factor in several carcinomas, such as hepatic carcinoma [14], malignant melanoma [15] and non-small-cell lung cancer [16]. Previous study found that tumor volume could predict survival in patients with renal cell carcinoma [17]. In a retrospective study, Jiang et al. reported that tumor volume was a prognostic factor in patients with GCs [18]. In our study, the calculate of tumor volume was based on tumor diameter and tumor invasion depth. It gives a new index to improve the prognostic assessment in SMGC patients. Patients with SMGC have two or more lesions in the stomach. Take this point into consideration, we first introduce a new index: total tumor volume (TTV). The TTV equals the sum of TV of each gastric tumor in a SMGC patient. In the univariate analysis, the 4 groups classified according to the TTV classification were significantly correlated with prognosis (P < 0.05). In multivariate survival analysis, the TTV classification was an independent prognostic factor (P < 0.05), however, the pT stage was not (P > 0.05). These results showed that the TTV classification was superior to the pT classification in SMGC patients.
To further investigate the prognostic significant of TTV, we introduced a new classification system: the total tumor volume-node-metastasis (TTVNM) system. To further confirm the superiority of the pTTVNM classification to the pTNM stage system in SMGC patients, we directly compared these two classifications in this study. We found that the pTTVNM classification (P < 0.05) was an appropriate prognostic classification for predicting the overall survival of SMGC patients, rather than the 8th edition pTNM classification (P > 0.05). We assumed the reason was that TV was a direct indicator of tumor burden [19]. Larger TV generally indicated quicker proliferation of tumor cells, more lymph nodes metastasis, and more possible distant metastasis [20]. These results suggested that the pTTVNM staging was superior to the pTNM staging for prognostic assessment in SMGC patients.
Our study showed that the traditional pTNM staging system exhibits some limitations in SMGC patients. In our opinion, although TTV was not a direct pathological factor in the postoperative pathology report, but we could easily calculate it by tumor diameter and tumor invasion depth. This index could reflect tumor burden better than pT in SMGC patients. Furthermore, we invented a new classification system in SMGC patients, which could predict the prognosis more accurately.
Conclusions
For patients with SMGC who underwent curative gastrectomy, TTV was an independent prognostic factor. Moreover, TTV-based classification was superior to pT classification for predicting prognosis in patients with SMGC. Incorporation of TTV into the pTTVNM staging system could compensate for the limitations of traditional pTNM stage system in SMGC patients [21]. The TTV should be recommended as an important clinicopathologic factor to improve the accuracy of prognostic prediction in patients with SMGC.
Abbreviations
- SMGC:
-
Synchronous multiple gastric cancer
- TTV:
-
Total tumor volume
- GC:
-
Gastric cancer
- AJCC:
-
American Joint Committee on Cancer
- UICC:
-
Union of International Cancer Control
- TNM:
-
Tumor-node-metastasis
- TV:
-
Tumor volume
References
Isobe T, Hashimoto K, Kizaki J, Murakami N, Aoyagi K, Koufuji K, Akagi Y, Shirouzu K (2013) Characteristics and prognosis of synchronous multiple early gastric cancer. World J Gastroenterol 19(41):7154–7159
Nozaki I, Hato S, Kobatake T, Ohta K, Kubo Y, Nishimura R, Kurita A (2014) Incidence of metachronous gastric cancer in the remnant stomach after synchronous multiple cancer surgery. Gastric Cancer 17(1):61–66
Borie F, Plaisant N, Millat B, Hay JM, Fagniez PL, De Saxce B, French Associations for Surgical Research (2003) Treatment and prognosis of early multiple gastric cancer. Eur J Surg Oncol 29(6):511–514
Deng J, Liang H, Sun D, Wang D, Pan Y (2010) Suitability of 7th UICC N Stage for Predicting the Overall Survival of Gastric Cancer Patients After Curative Resection in China. Ann Surg Oncol 17(5):1259–1266
Pak K, Cheon GJ, Nam HY, Kim SJ, Kang KW, Chung JK, Kim EE, Lee DS (2014) Prognostic value of metabolic tumor volume and total lesion glycolysis in head and neck cancer: a systematic review and meta-analysis. J Nucl Med 55(6):884–890
Eom BW, Lee JH, Choi IJ, Kook MC, Nam BH, Ryu KW, Kim YW (2012) Pretreatment risk factors for multiple gastric cancer and missed lesions. J Surg Oncol 105(8):813–817
Fujita T, Gotohda N, Takahashi S, Nakagohri T, Konishi M, Kinoshita T (2009) Clinical and histopathological features of remnant gastric cancers, after gastrectomy for synchronous multiple gastric cancers. J Surg Oncol 100(6):466–471
Zu H, Wang F, Ma Y, Xue Y (2013) Stage-stratified analysis of prognostic significance of tumor size in patients with gastric cancer. PLoS One 8(1):e54502
Zhang J, Zhou Y, Jiang K, Shen Z, Ye Y, Wang S (2014) Evaluation of the seventh AJCC TNM staging system for gastric cancer: a meta-analysis of cohort studies. Tumour Biol 35(9):8525–8532
Zhao LY, Zhang WH, Chen XZ, Yang K, Chen XL, Liu K, Zhang B, Chen ZX, Chen JP, Zhou ZG, Hu JK (2015) Prognostic Significance of Tumor Size in 2405 Patients With Gastric Cancer: A Retrospective Cohort Study. Med (Baltim) 94(50):e2288
Li H, Chen XL, Li JR, Li ZL, Chen TW, Pu H, Yin LL, Xu GH, Li ZW, Reng J, Zhou P, Cheng ZZ, Cao Y (2016) Tumor volume of resectable gastric adenocarcinoma on multidetector computed tomography: association with N categories. Clinics 71(4):199–204
Yoon H, Lee DH (2014) New approaches to gastric cancer staging: beyond endoscopic ultrasound, computed tomography and positron emission tomography. World J Gastroenterol 20(38):13783–13790
Neri E, Guidi E, Pancrazi F, Castagna M, Castelluccio E, Balestri R, Buccianti P, Masi L, Falcone A, Manfredi B, Faggioni L, Bartolozzi C (2015) MRI tumor volume reduction rate vs tumor regression grade in the pre-operative re-staging of locally advanced rectal cancer after chemo- radiotherapy. Eur J Radiol 84(12):2438–2443
Li MX, Zhao H, Bi XY, Li ZY, Huang Z, Han Y, Zhou JG, Zhao JJ, Zhang YF, Wei WQ, Zhao DB, Cai JQ (2016) Total tumor volume predicts survival following liver resection in patients with hepatocellular carcinoma. Tumour Biol 37(7):9301–9310
Voss B, Wilop S, Jonas S, El-Komy MH, Schaller J, von Felbert V, Megahed M (2014) Tumor volume as a prognostic factor in resectable malignant melanoma. Dermatology 228(1):66–70
Yu Y, Guan H, Xing LG, Xiang YB (2015) Role of gross tumor volume in the prognosis of non-small cell lung cancer treated with 3D conformal radiotherapy: a meta-analysis. Clin Ther 37(10):2256–2266
Choi SM, Choi DK, Kim TH, Jeong BC, Seo SI, Jeon SS, Lee HM, Choi HY, Jeon HG (2015) A comparison of radiologic tumor volume and pathologic tumor volume in renal cell carcinoma (RCC). PLoS One 10(3):e0122019
Jiang N, Deng JY, Ding XW, Liu Y, Liang H (2015) Tumor volume as a prognostic factor was superior to the seventh edition of the pT classification in resectable gastric cancer. Eur J Surg Oncol 41(3):315–322
Lee JA, Yang D, Yoon WS, Park YJ, Kim CY, Moon HY, Lee SI (2014) Tumor volume reduction assessed by planning computed tomography in patients with rectal cancer during preoperative chemoradiation: impact of residual tumor volume on the prediction of pathologic tumor regression. Tumori 100(2):158–162
Lu J, Huang CM, Zheng CH, Li P, Xie JW, Wang JB, Lin JX (2013) Consideration of tumor size improves the accuracy of TNM predictions in patients with gastric cancer after curative gastrectomy. Surg Oncol 22(3):167–171
Dicken BJ, Saunders LD, Jhangri GS, de Gara C, Cass C, Andrews S, Hamilton SM (2004) Gastric cancer: establishing predictors of biologic behavior with use of population-based data. Ann Surg Oncol 11(6):629–635
Acknowledgments
This study was supported by grants from the Key Research and Development Foundation of Shandong Province [NO. 2017GSF218034] and the Science Foundation of Qilu Hospital of Shandong University [NO. 2017QLQN16]. The funding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tang, X., He, Q., Sun, G. et al. Total Tumor Volume Should be Considered as an Important Prognostic Factor for Synchronous Multiple Gastric Cancer Patients with Curative Gastrectomy. Pathol. Oncol. Res. 26, 2169–2175 (2020). https://doi.org/10.1007/s12253-020-00804-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-020-00804-4