Abstract
The aim of this study is to evaluate the clinicopathological significance of L-type amino acid transporter 1 (LAT1) expression in patients with advanced laryngeal squamous cell carcinoma (LSCC). A total of 73 patients with advanced LSCC were retrospectively reviewed. Tumor sections were stained by immunohistochemistry for LAT1, 4F2hc, system ASC amino acid transporter-2 (ASCT2), cell proliferation by Ki-67, microvessel density (MVD) determined by CD34 and p53. A positive LAT1, 4F2hc and ASCT2 expression (staining more than a quarter) in the primary sites were recognized in 85, 80 and 45 %, respectively, and a high LAT1, 4F2hc and ASCT2 expression (staining more than a half) yielded 48, 31 and 18 %, respectively. High expression of LAT1 was significantly associated with lymph node metastasis, 4F2hc, ASCT2, Ki-67 and p53. The expression of LAT1 was significantly correlated with ASCT2, 4F2hc, cell proliferation, and MVD. By univariate analysis, there was no statistically significant relationship between LAT1 expression and prognosis in advanced LSCC. LAT1, 4F2hc and ASCT2 were highly expressed in patients with advanced laryngeal cancer. Our study suggests that the expression of LAT1 plays a crucial role in the metastasis and tumor progression in advanced LSCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Head and neck cancer is a malignant tumor arising from a different site in the upper aerodigestive tract, and the most common sites include the larynx, the oropharynx, the hypopharynx, and the oral cavity [1]. Surgery plus sequential chemoradiotherapy is generally considered as an appropriate treatment for patients with locally advanced diseases [2]. Laryngeal squamous cell carcinoma (LSCC) is the second most common malignant tumor of the head and neck cancers, and the occurrence of LSCC is believed to be associated with tobacco use, alcohol consumption, sex, air pollution and occupational factors [1, 2]. But, LSCC is an aggressive malignant disease with dismal prognosis, and the patients with LSCC need a promising therapeutic strategy for improving their outcome. The prognostic factors to improve the prognosis after treatment are currently still controversial. Therefore, a reliable biomarker should be established for improving the therapeutic efficacy.
Amino acid transporters play an essential role in the growth, proliferation and survival of both normal cells and transformed cells [3]. Of these transporters, L-type amino acid transporter 1 (LAT1) and system ASC amino acid transporter-2 (ASCT2) are shown to be significantly linked to the pathogenesis of cancer cells [4, 5]. LAT1 is an L-type amino acid transporter that transports large neutral amino acids, such as leucine, isoleucine, valine, phenylalanine, tyrosine, tryptophan, methionine and histidine, and requires a covalent association with the heavy chain of 4 F2 cell surface antigen (4F2hc) for its functional expression in the plasma membrane [5, 6]. LAT1 has been closely related to cancerous or proliferative cells and is highly expressed in the proliferating tissues [4–6]. Several in vitro studies have implicated that a LAT1 inhibitor could be an effective therapeutic option for patients with human neoplasms [7–9]. ASCT2 is a Na+-dependent transporter responsible for the transport of neutral amino acids, including glutamine, leucine and isoleucine [10]. ASCT2 is a major glutamine transporter and is associated with tumor growth and proliferation in cancer cells [4, 11]. By several studies, both LAT1 and ASCT2 are shown to play crucial roles in the development and progression of various human neoplasms and are significant biomarkers for predicting worse outcome [7, 12–23]. But, the clinicopathological significance of amino acid transporters such as LAT1 and ASCT2 expression remains unknown in patients with LSCC. In the present study, we focused on the protein expression of LAT1 in the resected tissue specimens, and the expression of LAT1 and ASCT2 were correlated with cell proliferation and angiogenesis.
Material and Methods
Patients
We analyzed 80 consecutive patients with pathologically confirmed LSCC who underwent surgical resection at Gunma University Hospital and Gunma Prefectural Cancer Center between 2001 and 2012. Five patients had pathological stage I or II disease. The specimens of two patients were not available. Patients who had received primary chemotherapy and/or radiotherapy were excluded from the study. Therefore, a total of 73 patients with stage III/IV disease were analyzed. Formalin-fixed, paraffin-embedded tumor samples from 73 patients for the primary manifestation of LSCC were examined. All surgical specimens were reviewed and classified according to the WHO classification by the two experienced pathologists who were unaware of clinical or imaging findings. Pathologic tumour-node-metastasis (TNM) stages were established using the Classification of Malignant Tumours by the International Union against Cancer (UICC) and American Joint Committee on Cancer (AJCC) system. The study protocol was approved by the institutional review board.
Immunohistochemical Staining
LAT1 expression was determined by immunohistochemical staining with an anti-human LAT1 antibody (1.6 mg/mL, anti-human monoclonal mouse IgG1, KM3149, provided by Kyowa Hakko Co., Ltd.; dilution of 1:800). The specificity of the antibody was confirmed by Western blotting as shown in Supplemental Fig. S1. A specific single band was detected with the antibody in HEK293 cells stably expressing human LAT1 but not in cells stably expressing human LAT2 (Supplemental Fig. S1). An oligopeptide (RDSKGLAAAEPTAN) corresponding to amino acid residues 7–20 of a rabbit polyclonal antibody against ASCT2 (1:300 dilution) was synthesized. The N-terminal cysteine residue was used for conjugation with keyhole limpet hemocyanin. An anti-peptide antibody was produced as described elsewhere [20]. The anti-CD98 antibody used is an affinity-purified rabbit polyclonal antibody (Santa Cruz Biotechnology, Inc.; 1:100 dilution) raised against a peptide mapping to the carboxy terminus of human CD98. The detailed protocol for immunostaining has been published elsewhere [18–20]. The LAT1 and CD98 expression scores were defined by the extent of staining as follows: 1, ≤ 10 % of tumor area stained; 2, 11–25 % stained; 3, 26–50 % stained; 4, 51–75 % stained; and 5, ≥76 %. The tumors in which the stained tumor cells were scored as 3, 4, or 5 were defined as positive; tumors scoring 4 or 5 were defined as having high expression. The immunohistochemical staining for CD34, Ki-67, and p53 was performed according to the procedures described in the previous reports [18–20]. The following antibodies were used: mouse monoclonal antibodies against CD34 (Nichirei, Tokyo, Japan; 1:800 dilution), Ki-67 (Dako, Glostrup, Denmark; 1:40 dilution), and p53 (D07; Dako; 1:50 dilution). The number of CD34-positive vessels was counted in four selected hot spots in a 400x field (field area of 0.26 mm2). MVD was defined as the mean count of microvessels per 0.26 mm2 field area. The median numbers of CD34-positive vessels were evaluated, and the tumors in which the stained tumor cells comprised more than each median value were defined as positive.
For p53, a microscopic examination of the nuclear reaction product was performed and scored. Based on previous reports [18–20], p53 expression greater than 10 % of the tumor cells was defined as positive expression. A highly cellular area of the immunostained sections was evaluated for Ki-67, and the epithelial cells with nuclear staining of any intensity were defined as positive. Approximately 1000 nuclei were counted on each slide, and the proliferative activity was assessed as the percentage of Ki-67-stained nuclei (Ki-67 labeling index) in the sample. The median Ki-67 labeling index value was determined, and high expression in the tumor cells was defined as a value greater than the median. The sections were assessed using light microscopy in a blinded fashion by two experienced authors.
Statistical Analysis
Probability values of <0.05 indicated a statistically significant difference. Fisher’s exact test was used to examine the association of two categorical variables. The correlation between different variables was analyzed using the nonparametric Spearman’s rank test. Follow-up for these 143 patients was conducted using the patient medical records. The Kaplan-Meier method was used to estimate survival as a function of time, and survival differences were analyzed by the log-rank test. Overall survival (OS) was determined as the time from tumour resection to death from any cause. Progression-free survival (PFS) was defined as the time between tumour resection and the first disease progression or death. Multivariate analyses were performed using stepwise Cox proportional hazards model to identify independent prognostic factors. Statistical analysis was performed using GraphPad Prism 4 software (Graph Pad Software, San Diego, CA, USA) and JMP 8 (SAS, Institute Inc., Cary, NC, USA) for Windows.
Results
Patient’s Demographics and Immunohistochemical Analysis
Table 1 shows a patient’s characteristics according to the LAT1 expression. Age of the patients ranged from 50 to 88 years, and the median age was 69 years. Twenty-two patients (30 %) had stage III and fifty-one patients (70 %) stage IV. Forty-four patients (60 %) received postoperative adjuvant chemotherapy with cisplatin, 5-FU (fluorouracil), docetaxel, or S-1 (Taiho Pharmaceutical Co., Ltd, Tokyo, Japan), oral administration of tegafur (a fluorouracil derivative drug), and/or radiotherapy. High expression of LAT1 was significantly associated with lymph node status, 4F2hc, ASCT2, Ki-67 and p53.
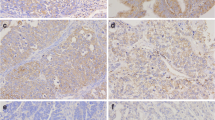
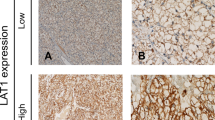
We performed an immunohistochemical staining on the 73 primary sites. Figure 1 shows the representative images of the immunohistochemical staining. Expression of LAT1 and ASCT2was predominantly localized at the plasma membrane of the carcinoma cells in the tumor tissues. Positive LAT1, 4F2hc and ASCT2 expression were found in 85 % (62/73), 80 % (59/73) and 45 % (33/73), respectively. Moreover, high LAT1, 4F2hc and ASCT2 expression were found in 48 % (35/73), 31 % (23/73) and 18 % (12/73), respectively. The positive expression rate of LAT1 was significantly higher than that of ASCT2 (p < 0.001), but there was no significant difference between LAT1 and 4F2hc (p = 0.66). The average scores of LAT1, 4F2hc and ASCT2 was 3.4 ± 0.9, 3.1 ± 0.9, and 2.3 ± 1.0, respectively, and the score of LAT1 was higher than that of ASCT2 (p < 0.001). However, no statistically significant difference was observed between the average scores of LAT1 and 4F2hc (p = 0.103). The median number of CD34-positive vessels was 13 (range, 2 to 30), and this value was chosen as a cutoff point. The median value of the Ki-67 labeling index was 28 (range, 2 to 81), and the value of 28 % was chosen as a cutoff point. Positive p53 expression was found in 41 % (30/73) (Table 2).
Correlation Between LAT1 / ASCT2 Expression and Different Variables
We analyzed any correlation between amino acid transporters (LAT1 and ASCT2) and the other biomarkers. The expression of LAT1 was significantly correlated with ASCT2, 4F2hc, cell proliferation and MVD determined by CD34. ASCT2 expression yielded a statistically significant correlation with 4F2hc (Table 2)
Prognosis of patients after surgical resection of advanced laryngeal cancer shown by Kaplan-Meier analysis of overall survival (OS) according to the LAT1 and ASCT2 expression. A statistically significant difference was observed between the patients with high and low LAT1 expression (a) and also between the patients with high and low ASCT2 expression
Univariate and Multivariate Survival Analysis
All patients revealed a median survival time (MST) and 5-year survival rate for OS of 917 days and 42 %, respectively, and the MST and 5-year survival rate for PFS were 840 days and 43 %, respectively. Table 3 shows results of the univariate and multivariate analysis in all patients with laryngeal cancer. By the univariate analysis, only the disease stage was significantly associated with poor OS, but we could not find any factors predicting poor PFS (Fig. 2). Multivariate analysis demonstrated that disease stage was an independent and significant factor for predicting poor outcome.
Discussion
We had recently reported that the LAT1 expression of was correlated with malignant features such as vascular invasion and lymph node metastases in a variety of human neoplasms [7, 12–19]. Although the expression status was significantly different according to the histological types of cancer, its mechanism was remained to be detailed [7, 12–19]. In patients with lung cancer, positive LAT1 expression was significantly higher in squamous cell carcinoma (SQC) (91 %) than in adenocarcinoma (AC) (29 %). The patients with SQC of esophagus or oral cavity also yielded a positive LAT1 expression rate of approximately 70–90 %, whereas, a positive LAT1 expression was observed in approximately 30–60 % of patients with AC such as pancreatic cancer, biliary tract cancer and breast cancer [7, 12–19, 24]. LAT1 requires a corporative expression of 4F2hc to function as an amino acid transporter [13, 19]. In a variety of human neoplasms, LAT1 plays a crucial role in the development and progression of tumor cells, and the previous clinicopathological data indicated that LAT1 expression was closely associated with cell proliferation and angiogenesis [13, 19]. Clinical significance of LAT1 has been described to be an independent and significant marker for predicting poor survival after treatment [7, 12–19, 24]. In the present study, we found that LAT1 was highly expressed in patients with advanced laryngeal cancer and yielded a significant correlation with 4F2hc, ASCT2, cell proliferation and angiogenesis. But, no statistically significant difference in the prognosis was recognized between high and low LAT1 expression. The results of our study suggest that LAT1 has a significant malignant feature associated with tumor cell progression and metastasis for patients with advanced laryngeal SQC.
In the present study, LAT1 and 4F2hc were positively expressed in 85 and 80 % of laryngeal SQC, respectively. These expressions were closely correlated, although the positive expression of ASCT2 (45 %) was significantly lower than that of LAT1. By the recent research, both LAT1 and ASCT2 are cooperatively expressed in primary human cancers and several cell lines, and these amino acid transporters are shown to play essential role in the cell growth and survival [19–21]. Additionally, the recent studies had documented that the protein expression of LAT1 and ASCT2 could be an independent and negative prognostic biomarker in various human neoplasms [19–21]. Both LAT1 and ASCT2 expressions have a prognostic significance in cancer patients with a histology of adenocarcinoma [13, 19–21], although these amino acid transporters yielded a higher expression in SQC compared to AC. The prognosis may not be predicted according to the expression level of these transporters in patients with SQC. Our study also indicates that the expression level of LAT1 and ASCT2 may not exactly predict a survival after surgery in the advanced laryngeal SQC. Moreover, the previous studies showed that the expression level of LAT1 was closely correlated with the outcome after surgery in patients with early-stage disease, but was not correlated with the outcome in the advanced-stage disease [24, 25]. These findings were corresponding to the results of the present study. However, little is known about the detailed mechanisms of different relationship of the expression and prognosis between SQC and AC, and between early- and advanced-stage. Further study is needed to examine the prognostic role of the amino acid transporters such as LAT1 and ASCT2 according to histological types and disease stage.
LAT1 imports essential amino acids in exchange for the glutamine that has been imported by ASCT2. LAT1 and ASCT2 are cooperatively provides amino acids for protein synthesis through glutamine exchange. The present results disclosed the correlation of 4F2hc expression with the expression of both LAT1 and ASCT2. The covalent association of 4F2hc was required for LAT1 function in the plasma membrane. Amino acid such as leucine regulates cell signaling via mammalian target of rapamycin (mTOR). LAT1 and ASCT2 are closely associated with the mTOR pathway in the human tumors [4, 26, 27]. The inhibition of amino acid transporters has been shown to reduce phosphorylation of mTOR and downstream proteins by several in vitro studies [28].
In conclusion, LAT1, 4F2hc and ASCT2 were highly expressed in patients with advanced laryngeal cancer. The expression of LAT1 was closely associated with the metastasis and tumor progression, although LAT1 expression was not predictive as a prognostic factor. Further study is warranted to evaluate the prognostic significance of these amino acid transporters in the with early-stage laryngeal cancer.
References
Ragin CC, Modugno F, Gollin SM (2007) The epidemiology and risk factors of head and neck cancer: a focus on human papillomavirus. J Dent Res 86:104–114
Thephamongkhol K, Browman G, Hodson I, Oliver T, Zuraw L, members of the Head and Neck Cancer Disease Site Group (2004) Chemotherapy with radiotherapy for nasopharygeal cancer: a clinical practice guidelineevidence-based Series #5-7. Cancer Care Ontario, Program in Evidence-Based Care, Toronto
Christensen HN (1990) Role of amino acid transport and countertransport in nutrition and metabolism. Physiol Rev 70:43–77
Fuchs BC, Bode BP (2006) Amino acid transporters ASCT2 and LAT1 in cancer: partners in crime? Semin Cancer Biol 15:254–266
Kanai Y, Segawa H, Miyamoto K et al (1998) Expression cloning and characterization of a transporter for large neutral amino acids activated by the heavy chain of 4F2 antigen (CD98). J Biol Chem 273:23629–23632
Yanagida O, Kanai Y, Chairoungdua A et al (2001) Human L-type amino acid transporter 1 (LAT 1): characterization of function and expression in tumor cell lines. Biochim Biophys Acta 1514:291–302
Nawashiro H, Otani N, Shinomiya N et al (2006) L-type amino acid transporter 1 as a potential molecular target in human astrocytic tumors. Int J Cancer 119:484–492
Kim CS, Cho SH, Chun HS et al (2008) BCH, an inhibitor of system L amino acid transporters, induces apoptosis in cancer cells. Biol Pharm Bull 31:1096–1100
Kim CS, Moon IS, Park JH et al (2010) Inhibition of L-type amino acid transporter modulates the expression of cell cycle regulatory factors in KB oral cancer cells. Biol Pharm Bull 33:1117–1121
Kekuda R, Prasad PD, Fei YJ et al (1996) Cloning of the sodium-dependent, broad-scope, neutral amino acid transporter Bo from a human placental choriocarcinoma cell line. J Biol Chem 271:18657–18661
Fuch BC, Finger RE, Onan MC et al (2007) ASCT2 silencing regulates mammalian target of rapamycin growth and survival signaling in human hepatoma cells. Am J Physiol Cell Physiol 293:C55–C63
Kobayashi H, Ishii Y, Takayama T (2005) Expression of L-type amino acid transporter 1 (LAT1) in esophageal carcinoma. J Surg Oncol 90:233–238
Kaira K, Oriuchi N, Imai H et al (2008) Prognostic significance of L-type amino acid transporter 1 expression in resectable stage I-III nonsmall cell lung cancer. Br J Cancer 98:742–748
Nakanishi K, Ogata S, Matsuo H et al (2007) Expression of LAT1 predicts risk of progression of transitional cell carcinoma of the upper urinary tract. Virchows Arch 451:681–690
Sakata T, Ferdous G, Tsuruta T et al (2009) L-type amino acid transporter 1 as a novel biomarker for high-grade malignancy in prostate cancer. Pathol Int 59:7–18
Ichinoe M, Mikami T, Yoshida T et al (2011) High expression of L-type amino-acid transporter 1 (LAT1) in gastric carcinomas: comparison with non-cancerous lesions. Pathol Int 61:281–289
Furuya M, Horiguchi J, Nakajima H et al (2012) Correlation of L-type amino acid transporter 1 and CD98 expression with triple negative breast cancer prognosis. Cancer Sci 103:382–389
Kaira K, Oriuchi N, Takahashi T et al (2011) LAT1 expression is closely associated with hypoxic markers and mTOR in resected non-small cell lung cancer. Am J Transl Res 3:468–478
Kaira K, Sunose Y, Arakawa K et al (2012) Prognostic significance of L-type amino acid transporter 1 expression in surgically resected pancreatic cancer. Br J Cancer 107:632–638
Shimizu K, Kaira K, Tomizawa Y et al (2014) ASC amino acid transporter 2 (ASCT2) as a novel prognostic marker in non-small cell lung cancer. Br J Cancer 110:2030–2039
Toyoda M, Kaira K, Ohshima Y et al (2014) Prognostic significance of amino acid transporter expression (LAT1, ASCT2 and xCT) in surgically resected tongue cancer. Br J Cancer 110:2506–2513
Li R, Younes M, Frolov A et al (2003) Expression of neutral amino acid transporter ASCT2 in human prostate. Anticancer Res 23:3413–3418
Whitte D, Ali N, Carlson N et al (2002) Overexpression of the neutral amino acid transporter ASCT2 in human colorectal adenocarcinoma. Anticancer Res 22:2555–2557
Kaira K, Oriuchi N, Imai H et al (2009) CD98 expression is associated with poor prognosis in resected non-small-cell lung cancer with lymph node metastases. Ann Surg Oncol 16:3473–3481
Kaira K, Oriuchi N, Imai H et al (2009) Prognostic significance of L-type amino acid transporter 1 (LAT1) and 4F2 heavy chain (CD98) expression in stage I pulmonary adenocarcinoma. Lung Cancer 66:120–126
Namikawa M, Kakizaki S, Kaira K, et al (2014) Expression of amino acid transporters (LAT1, ASCT2 and xCT) as clinical significance in hepatocellular carcinoma. Hepatol Res in press
Hassanein M, Hoeksema MD, Shiota M et al (2013) SLC1A5 mediates glutamine transport required for lung cancer cell growth and survival. Clin Cancer Res 19:560–570
Imai H, Kaira K, Oriuchi N et al (2010) Inhibition of L-type amino acid transporter 1 has antitumor activity in non-small cell lung cancer. Anticancer Res 30:4819–4828
Acknowledgments
This work was supported in part by Grant 23591750 (K. K) and Grant 23592523 (K. C) from the Ministry of Education, Culture, Sports, Science and Technology, Japan, and National Hospital Organization Policy Based Medical Services. We thank Ms. Chizuko Tomioka for their technical assistance of immunohistochemical analysis.
Conflicts of Interest
We, all authors, have no financial or personal relationships with other people or organizations that could inappropriately influence our work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nikkuni, O., Kaira, K., Toyoda, M. et al. Expression of Amino Acid Transporters (LAT1 and ASCT2) in Patients with Stage III/IV Laryngeal Squamous Cell Carcinoma. Pathol. Oncol. Res. 21, 1175–1181 (2015). https://doi.org/10.1007/s12253-015-9954-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-015-9954-3