Abstract
As mammalian Target of Rapamycin (mTOR) plays role in protein synthesis and metabolism, mTOR pathway activation is involved in the pathogenesis of several types of tumors. Our aim was to elucidate its role in medulloblastoma in terms of prognosis and as a therapeutic target. Members of activated mTOR complex 1 (mTORC1) pathway, phospho-mTOR (p-mTOR) and phospho-S6 (p-S6) were examined by immunohistochemistry in formalin fixed paraffin embedded samples of 40 patients with medulloblastoma, and results were compared to clinical features and survival of patients. In proliferation assays, Daoy and UW228–2 medulloblastoma cell lines were tested by rapamycin, an mTORC1 inhibitor, and NVP-BEZ235, a dual mTOR and phosphatidylinositol 3-kinase (PI3K) inhibitor, each in monotherapy and in combination with cytostatic drugs (cisplatin, etoposide). Components of mTORC1 and mTORC2 complexes were also examined in these cell lines. Neither presence of p-mTOR (32.5 %) nor p-S6 (32.5 %) correlated with age, gender or histological subtype. In 22.5 % of cases simultaneous expression of p-mTOR and p-S6 was shown. Kaplan-Meier analysis showed inferior survival of patients expressing both marker proteins, but it was not statistically significant, probably due to low case number. UW228–2 cells had greater sensitivity to mTOR inhibitors, possibly due to its higher mTORC1 specific protein expression levels, compared to Daoy cells. In both cell lines antiproliferative effect of cytostatic drugs was significantly enhanced by mTOR inhibitors (p < 0.05). Based on our in vitro and clinicopathological studies mTOR inhibitors may have a role in the future treatment of a subset of patients with medulloblastoma.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medulloblastoma (MB) – a type of embryonal tumors – is the most frequent malignant brain tumor in children. The name covers a heterogeneous group of variants, different in origin, histological appearance and molecular characteristics. According to the World Health Organization (WHO) there are classic, desmoplastic, extensive nodular, anaplastic and large cell variants [1, 2]. The current treatment (surgery, radiation and chemotherapy) of MB is often accompanied by serious side effects including neurocognitive impairment, psychological and behavioral disturbances, secondary malignancies and endocrinopathies [3, 4]. Five-year overall survival is 70–90 %, which is even worse in high risk patients [5, 6]. New diagnostic tools and targeted strategies may improve survival and long-term quality of life.
Due to its central role in growth and metabolism, mammalian Target of Rapamycin (mTOR) seems to be an important factor in many types of cancer, making it a possible target for cancer therapy. It is activated by receptor tyrosine kinases via phosphatidylinositol 3-kinase (PI3K)-Akt pathway [7, 8]. mTOR – a serin-threonin protein kinase - can operate in two complexes called mTORC1 and mTORC2 [8, 9]. These two complexes have different components: mTORC1 is characterized by Raptor and mTORC2 is characterized by Rictor [7–9]. The mTORC1 complex is rapamycin sensitive, participates in protein and lipid synthesis, ribosome biogenesis and autophagy, mainly mediated by p70 ribosomal protein S6 kinase (rpS6K) and eukaryotic translation initiation factor 4E binding protein 1 (4EBP1) [7–10]. mTORC2 can directly phosphorylate Akt, and has a role in actin cytoskeleton controlling and cell survival. It was thought to be resistant to rapamycin but long-term rapamycin treatment may have indirect inhibitory effect on mTORC2. Function and regulation of mTORC2 and its response to rapamycin remain unclear, and may vary in different cell types [7–11]. Rapalogs (rapamycin analogues) and newly developed mTORC1/mTORC2 inhibitors and dual PI3K/mTOR inhibitors were applied in several completed and ongoing clinical trials in different types of cancer [8–10, 12].
Previous data suggest that there are some cases of MBs with disregulated Akt and Erk pathways which may activate mTOR signaling. Elevated Erk pathway was found by Western blot, whereas Akt seems to be slightly upregulated. However few cases were included in these studies [13–15]. Deletions of tuberous sclerosis 1 (TSC1), a growth inhibitory protein, that indirectly inhibit mTOR were also found in a small subset of medulloblastoma resulting in release of mTOR inhibition [16, 17]. Data suggest that insulin-like growth factor-mTOR signaling and Sonic hedgehog activation in cerebellar granular precursor cells could interact and enhance tumor formation [16–19]. mTORC1 inhibitor rapamycin and its derivates seem to be effective in certain MB cell lines, its effectiveness was also supported by in vivo experiments [20–22]. We aimed to clarify the activity of mTORC1 by examination of the presence of involved proteins (p-mTOR and p-S6) and its correlation with histology and survival data of patients with medulloblastoma. Effect of mTOR inhibitors in medulloblastoma cell cultures, and mTOR pathway related proteins were examined.
Methods
Human Tissue Samples and Clinical Data
Formalin fixed paraffin embedded MB tumor samples were obtained from National Institute of Neurosciences (Budapest, Hungary) and 1st Institute of Pathology and Experimental Cancer Research, Semmelweis University (Budapest, Hungary). The specimens were diagnosed according to the criteria of the WHO. Primary tumors of 40 patients operated between 2004 and 2010 were examined. Clinical data of patients were obtained from Hungarian Pediatric Cancer Registry. For survival analysis, Kaplan Meier estimated survival was applied. Investigations were approved by the Institutional Ethical Review Board (TUKEB no. 100/2012 and 155/2012).
In vitro Cell Cultures
Two human medulloblastoma cell lines were used: Daoy was purchased from ATCC, UW228-2 was kindly provided Dr. J. Silber (University of Washington, Seattle, WA, USA).
Daoy and UW228–2 cell lines were maintained in Minimum Essential Medium Eagle, Alpha Modification (M8042, Sigma, St. Louis, USA) supplemented with 10 % FCS (Gibco), Gentamycin (Sandoz), sodium-pyruvate, non-essential-amino acid solution and L-glutamine (Sigma, St. Louis, USA) at 37 °C in humidified 5 % CO2 air.
Construction of Tissue Microarrays
Representative areas of human MB samples were selected by reviewing haematoxylin and eosin slides of each case. In addition as control, normal cerebellar tissues were included in Tissue Microarray (TMA) blocks. We used computer controlled TMA Master (3DHISTECH, Budapest, Hungary) instrument, core diameter was 2 mm.
Immunohistochemistry
TMA sections (4 μm) were deparaffinized and slides were boiled in citrate buffer (pH = 6.0) by an electric pressure cooker for 20 min, cooled and washed in PBS. p-mTOR (#2976, Cell Signaling, USA; dilution 1:100) and p-S6 (#2211, Cell Signaling, USA; dilution 1:150) primary antibodies were diluted in 3 % normal horse serum containing PBS and incubated at room temperature for 1.5 h. Novolink Polymer Detection System (Novocastra, Wetzlar, Germany) was applied to develop antigens according to the manufacturer’s instructions. Diaminobenzidine (DAB) (DAKO, Denmark) substrate was used and slides were counterstained with haematoxylin. Pannoramic Scan instrument and Pannoramic Viewer program with TMA module software (3DHISTECH, Budapest, Hungary) were used to digitalize slides and analysis. For positive control colon tumor and lymphoma tissues were used.
Daoy and UW228–2 cell cultures were trypsinized and 100,000 cells were centrifuged on slides by Cytospin (Shandon Scientific LTD). Cells were fixed in 80 % methanol for 10 min. Then samples were incubated with primary antibodies at 4 °C overnight in a humidified chamber. p-mTOR (ab51044, Abcam, Cambridge, UK), Rictor (ab56578, Abcam, Cambridge, UK), Raptor (ab40768, Abcam, Cambridge, UK) and p-S6 (#2211, Cell Signaling, USA) antibodies were diluted 1:100 in 3 % normal horse serum containing PBS, detection was done by Novolink Polymer Detection System (Novocastra, Wetzlar, Germany) and DAB (DAKO, Denmark) substrate followed by nuclear counterstaining with haematoxylin.
Evaluation of Immunohistochemical and Immunocytochemical Reactions
Cytoplasmic presence of p-mTOR and p-S6 was evaluated in human tumor samples. In each case 1,000 cells were counted and percentages of immunopositive cells were calculated. For further analysis, slides were regarded as positive if immunopositive cells are ≥ 1 %.
Cells in each cell culture were stained homogeneously, evaluation of reactions in MB cell culture based on the intensity of staining (0/negative; +/weak; ++/moderate and +++/strong).
Drugs and Treatments
Daoy or UW228–2 cells were seeded in 96-well plates (3 × 103 cells/well). After 24 h media was refreshed and cells treated for 72 h by drugs. The mTORC1 inhibitor rapamycin (Sigma) (applied doses were 0.5, 5 and 50 ng/ml) and the NVP-BEZ235 (Cayman Europe, Estonia) PI3K and mTORC1/C2 inhibitor (applied doses: 0.1 and 1 μM) were tested. Slightly effective doses were applied, selection of concentration was based on pre-experiments. Drugs were applied in combination with cytostatic drugs: cisplatin (Ebewe Pharma, Austria) 1 μM for both cell lines and etoposide (Ebewe Pharma, Austria) 0.1 μM for UW228–2 or 1 μM for Daoy according to their IC10–20, based on pre-experiments. Each experiment was performed in triplicate.
Proliferation Assays
Cell proliferation was evaluated by MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (Sigma, St. Louis, USA) assay after 72 h by method described in manufacturer’s protocol.
Statistical Analysis
Statistical analyses were carried out with Statistica 9.0 software (StatSoft). Correlation between p-mTOR and p-S6 was tested by Spearman rank order test. We analyzed correlation of p-mTOR and p-S6 expression with age (Mann–Whitney U-test), gender and histological subtype (Fisher’s exact test) according. At survival analysis for statistical evaluation log rank test was applied. Samples with co-expression of both markers were defined as with activated mTORC1 pathway, any other cases were considered as samples without mTORC1 activity.
In cell culture study for comparison of different treatments two tailed t-test was used. Statistically significant difference was regarded as p < 0.05.
Results
Patients’ Characteristics
Median age of patients was 8.2 years (from 1.1 years through 28.7 years). Female to male rate was 21:19. Classic, desmoplastic and large cell/anaplastic medulloblastoma were 62.5 %, 32.5 % and 5.0 % of samples, respectively. Median follow up time of survival was 4.0 years (survival dates were available in 92.5 % of cases). All patients were treated according to the Hungarian MBL2004/2008 schedule [23]. (Patients data are shown in Table 1.)
mTOR Activity in Human Samples
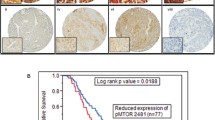
Expression of p-mTOR was observed in 32.5 % of cases and the ratio of expression of p-S6 was the same. Simultaneous positivity (p-mTOR and p-S6) was present in 9 samples (22.5 %). Representative stains are shown in Fig. 1.
Expression of p-mTOR and p-S6 showed strong correlation (R = 0.55; p = 0.0002). Neither of them showed correlation with age (p = 0.97 and p = 0.59, respectively) or gender (p = 0.74 and p = 1.00, respectively). 81.8 % of p-mTOR positive cases were classic, and 18.2 % were desmoplastic, 63.6 % of p-S6 positive cases were classic MB and 36.4 % were desmoplastic. Both of large cell/anaplastic samples were positive for p-mTOR and p-S6. Correlations of these proteins with histological subtype could be evaluated only in classic and desmoplastic subtypes. Expression of p-mTOR and p-S6 did not show correlation with histological subtypes (p = 0.27 and p = 1.00, respectively) (Table 2).
Nuclear p-mTOR staining (18 cases out of 40) was also observed. Several authors describe nuclear mTOR presence, but its role desires further investigation [24–26], hence, our analysis did not focus on nuclear staining.
Kaplan-Meier estimated survival shows that patients presenting mTORC1 activity (N = 9; 24.3 %) have lower overall survival rate compared to patients with inactive mTORC1 (N = 28; 76.7 %) without statistical significance (p = 0.12) (Fig. 2).
mTOR Activity in Medulloblastoma Cell Lines
Proliferation Assay
Inhibitory effect of mTORC1 inhibitor rapamycin and dual inhibitor NVP-BEZ235 were tested in two MB cell lines (Daoy and UW228–2) with doses selected according to previous studies [21]. UW228–2 cell line shows higher sensitivity to rapamycin and NVP-BEZ235 than Daoy cell line. Rapamycin has only slight proliferation inhibitory effect even at 50 ng/ml dose in Daoy cells (Fig. 3). mTOR inhibitors were combined with cytostatic drugs, cisplatin and etoposide. Rapamycin and NVP-BEZ235 significantly enhanced their cytostatic effect in both cell lines, especially of cisplatin in Daoy cell line (p < 0.05) (Fig. 4).
Effect of mTOR inhibitors, rapamycin (a) and NVP-BEZ235 (b) in MB cell lines (Daoy and UW228–2) compared to untreated control. UW228–2 cells show higher sensitivity for rapamycin and NVP-BEZ235 than Daoy cells. Daoy cells were not sensitive for rapamycin in applied doses (Graphs show one representative experiment of the triplicate)
Combined treatment of mTOR inhibitors (rapamycin and NVP-BEZ235) and cytostatic drugs (cisplatin and etoposide) in MB cell lines (Daoy and UW228–2). Proliferation was compared to untreated cells. In both cell lines proliferation was significantly inhibited by combined treatment: (a) cisplatin combined with rapamycin (b) cisplatin combined with NVP-BEZ235 (c) etoposide combined with rapamycin (d) etoposide combined with NVP-BEZ235 (Graphs show one representaive experiment of the triplicate evaluated by two-tailed t-test) *denotes p < 0.05
Immunocytochemistry
Immunocytochemical analysis of p-mTOR and p-S6, Rictor and Raptor were performed in both MB cell lines. Daoy cells showed strong p-mTOR, moderate p-S6, weak Raptor and strong Rictor positivity. UW228–2 cells were strongly positive for all examined proteins. These results indicate weak mTORC1 and elevated mTORC2 activity in Daoy cells, whereas both complex activities were elevated in UW228–2 cells.
Discussion
Medulloblastoma is a heterogeneous group of childhood brain tumors and several pathways were implicated in its development like Shh, Wnt, Notch and receptor tyrosine kinase signaling [1, 2, 27]. Here, we examined mTORC1 activation in MB. The upstream signaling for mTOR activation is derived from growth factors through tyrosine kinase receptors or altered metabolic status. mTORC1 could be activated through Akt or Erk transmitted signals [8, 28] which could contribute to MB development [27, 29].
In our cohort of primary MBs we have found a small subset showing mainly weak mTORC1 activity (with co-expression of p-mTOR and p-S6). There was a tight correlation between expression of p-mTOR and p-S6 suggesting mTORC1 activity in samples expressing these proteins. Neither p-mTOR nor p-S6 expression showed correlation with age or gender. Additionally, mTORC1 activity did not correlate with histological subtype.
Patients with active mTORC1 pathway have worse outcome than those with inactive pathway, the difference was not significant. Due to the limitation of cases with same treatment in Hungary, this question could not be answered more accurately. mTOR pathway activation was described as a poor prognostic factor in several type of tumors: glioma [30], glioblastoma [31], leiomyosarcomas [32], diffuse large B-cell lymphomas [33], colorectal cancer [34] and oral cancer [35].
MB tumor cell lines show different sensitivity to mTOR inhibitors [21, 36]. This was also observed in other type of tumors, for instance in breast cancer cells [37]. We tested rapamycin (bind to FKBP-12 protein, thereby inhibit Raptor/mTOR association and prevent mTORC1 action) and NVP-BEZ235 (dual PI3K/mTOR kinase inhibitor joining ATP-binding site of these enzymes, blocking both mTORC1 and mTORC2 complexes) in two MB cell lines, Daoy and UW228–2 [8, 38]. According to published data both cell lines expected to be sensitive [21, 36]. UW228–2 cells showed greater sensitivity to both drugs than Daoy cells. In contrast to Geoerger et al. [21] we have found that Daoy cell line was resistant to rapamycin.
In the aspect of personalized and targeted therapy, a crucial question is which markers are able to predict therapeutic response [39]. Expression of mTOR pathway components may predict sensitivity to mTOR inhibitors, hence we tested these markers on MB cell lines. Raptor and p-S6 expression were higher in UW228–2 cell line than in Daoy cells, indicating that UW228–2 cell line is more sensitive to mTORC1 inhibitor rapamycin. The amount of Rictor is high in both cell lines suggesting that NVP-BEZ235 (inhibits not only mTORC1 but also mTORC2 complexes) may have antiproliferative effect in both cell lines. NVP-BEZ235 was more effective in UW228–2 cells. The results of proliferation tests showed the expected effects based on the expression of these proteins.
Rapamycin and NVP-BEZ235 significantly enhanced the effect of etoposide and cisplatin in Daoy and UW228–2 cell lines. The strongest interaction was observed in Daoy cells where cisplatin was combined with mTOR inhibitors. These results suggest that co-administration of mTOR inhibitors with certain cytostotic drugs may have rationale in future treatment in a subgroup of MB patients.
In conclusions, mTORC1 pathway is activated in a small subset of MB mostly with weak activity. Our survival analysis suggests prognostic role of the presence of activated mTOR pathway in MB, which should be confirmed in a higher cohort of patients. Our in vitro results support previous findings, and indicate that mTOR inhibitors augment the effect of chemotherapy in MB in presence of activated mTORC1 pathway in a synergistic way, which should be proved in further preclinical experiments.
References
Hatten ME, Roussel MF (2011) Development and Cancer of the Cerebellum. Trends Neurosci 34:134–142
Roussel MF, Hatten ME (2011) Cerebellum Development and Medulloblastoma. Curr Top Dev Biol 94:235–282
Crawford JR, MacDonald TJ, Packer RJ (2007) Medulloblastoma in Childhood: new Biological Advances. Lancet Neurol 6:1073–1085
Massimino M, Giangaspero F, Garrè ML, Gandola L, Poggi G, Biassoni V, Gatta G, Rutkowski S (2011) Childhood Medulloblastoma. Crit Rev Oncol Hematol 79:65–83
Gottardo NG, Gajjar A (2008) Chemotherapy for Malignant Brain Tumors of Childhood. J Child Neurol 23:1149–1159
Packer RJ, Zhou T, Holmes E, Vezina G, Gajjar A (2013) Survival and Secondary Tumors in Children with Medulloblastoma Receiving Radiotherapy and Adjuvant Chemotherapy: Results of Children’s Oncology Group trial A9961. Neuro Oncol 15:97–103
Dazert E, Hall MN (2011) mTOR Signaling in Disease. Curr Opin Cell Biol 23:744–755
Willems L, Tamburini J, Chapuis N, Lacombe C, Mayeux P, Bouscary D (2012) PI3K and mTOR Signaling Pathways in Cancer: New Data on Targeted Therapies. Curr Oncol Rep 14:129–138
Guertin DA, Sabatini DM (2007) Defining the Role of mTOR in Cancer. Cancer Cell 12:9–22
Dunlop EA, Tee AR (2009) Mammalian target of Rapamycin Complex 1: Signalling Inputs, Substrates and Feedback Mechanisms. Cell Signal 21:827–835
Akcakanat A, Singh G, Hung MC, Meric-Bernstam F (2007) Rapamycin Regulates the Phosphorylation of Rictor. Biochem Biophys Res Commun 362:330–333
Fouladi M, Laningham F, Wu J, O’Shaughnessy MA, Molina K, Broniscer A, Spunt SL, Luckett I, Stewart CF, Houghton PJ, Gilbertson RJ, Furman WL (2007) Phase I Study of Everolimus in Pediatric Patients with Refractory Solid Tumors. J Clin Oncol 25:4806–4812
Jozwiak J, Sontowska I, Bikowska B, Grajkowska W, Galus R, Roszkowski M (2011) Favourable Prognosis in Medulloblastoma with Extensive Nodularity is Associated with Mitogen-activated Protein Kinase Upregulation. Folia Neuropathol 49:257–261
Wlodarski P, Grajkowska W, Lojek M, Rainko K, Jozwiak J (2006) Activation of Akt and Erk Pathways in Medulloblastoma. Folia Neuropathol 44:214–220
Wlodarski PK, Boszczyk A, Grajkowska W, Roszkowski M, Jozwiak J (2008) Implication of Active Erk in the Classic Type of Human Medulloblastoma. Folia Neuropathol 46:117–122
Bhatia B, Northcott PA, Hambardzumyan D, Govindarajan B, Brat DJ, Arbiser JL, Holland EC, Taylor MD, Kenney AM (2009) Tuberous Sclerosis Complex Suppression in Cerebellar Development and Medulloblastoma: Separate Regulation of Mammalian Target of Rapamycin Activity and p27 Kip1 Localization. Cancer Res 69:7224–7234
Bhatia B, Nahlé Z, Kenney AM (2010) Double Trouble: when Sonic Hedgehog Signaling Meets TSC Inactivation. Cell Cycle 9:456–459
Mainwaring LA, Kenney AM (2011) Divergent Functions for eIF4E and S6 Kinase by Sonic Hedgehog Mitogenic Signaling in the Developing Cerebellum. Oncogene 30:1784–1797
Parathath SR, Mainwaring LA, Fernandez-L A, Campbell DO, Kenney AM (2008) Insulin Receptor Substrate 1 is an Effector of Sonic Hedgehog Mitogenic Signaling in Cerebellar Neural Precursors. Development 135:3291–3300
Buonamici S, Williams J, Morrissey M, Wang A, Guo R, Vattay A, Hsiao K, Yuan J, Green J, Ospina B, Yu Q, Ostrom L, Fordjour P, Anderson DL, Monahan JE, Kelleher JF, Peukert S, Pan S, Wu X, Maira SM, Garcia-Echeverria C, Briggs KJ, Watkins DN, Yao YM, Lengauer C, Warmuth M, Sellers WR, Dorsch M (2010) Interfering with resistance to smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci Transl Med 2:51ra70
Geoerger B, Kerr K, Tang CB, Fung KM, Powell B, Sutton LN, Phillips PC, Janss AJ (2001) Antitumor Activity of the Rapamycin Analog CCI-779 in Human Primitive Neuroectodermal Tumor/Medulloblastoma Models as Single Agent and in Combination Chemotherapy. Cancer Res 61:1527–1532
Pei Y, Moore CE, Wang J, Tewari AK, Eroshkin A, Cho YJ, Witt H, Korshunov A, Read TA, Sun JL, Schmitt EM, Miller CR, Buckley AF, McLendon RE, Westbrook TF, Northcott PA, Taylor MD, Pfister SM, Febbo PG, Wechsler-Reya RJ (2012) An animal Model of MYC-driven Medulloblastoma. Cancer Cell 21:155–167
Hauser P, Jakab Z, Kiss C, Szegedi I, Bárdi E, Batyik K, Ottóffy G, Kajtár P, Szűcs R, Nagy K, Cservák J, Masát P, Bálint K, Kordás M, Bognár L, Kocsis B, Vízkeleti J, Kriván G, Kállay K, Benyó G, Schuler D, Garami M (2009) Előzetes eredmények a medulloblastoma/primitív neuroektodermális tumor (PNET) kezelésében a magyar MBL 2004 kezelési sémával. Magyar Belorvosi Archívum, pp 196–201
Back JH, Kim AL (2011) The expanding Relevance of Nuclear mTOR in Carcinogenesis. Cell Cycle 10:3849–3852
Malik AR, Urbanska M, Macias M, Skalecka A, Jaworski J (2013) Beyond Control of Protein Translation: what we Have Learned About the Non-canonical Regulation and Function of Mammalian Target of Rapamycin (mTOR). Biochim Biophys Acta 1834:1434–1448
Rosner M, Hengstschläger M (2008) Cytoplasmic and Nuclear Distribution of the Protein complexes mTORC1 and mTORC2: Rapamycin Triggers Dephosphorylation and Delocalization of the mTORC2 Components riCtor and Sin1. Hum Mol Genet 17:2934–2948
Jozwiak J, Grajkowska W, Wlodarski P (2007) Pathogenesis of Medulloblastoma and Current Treatment Outlook. Med Res Rev 27:869–890
Yecies JL, Manning BD (2011) mTOR links Oncogenic Signaling to Tumor Cell Metabolism. J Mol Med (Berl) 89:221–228
Del Valle L, Enam S, Lassak A, Wang JY, Croul S, Khalili K, Reiss K (2002) Insulin-like Growth Factor I Receptor Activity in Human Medulloblastomas. Clin Cancer Res 8:1822–1830
Yang J, Liao D, Wang Z, Liu F, Wu G (2011) Mammalian Target of Rapamycin Signaling Pathway Contributes to Glioma Progression and Patients’ Prognosis. J Surg Res 168:97–102
Pelloski CE, Lin E, Zhang L, Yung WK, Colman H, Liu JL, Woo SY, Heimberger AB, Suki D, Prados M, Chang S, Barker FG, Fuller GN, Aldape KD (2006) Prognostic Associations of Activated Mitogen-activated Protein Kinase and Akt Pathways in Glioblastoma. Clin Cancer Res 12:3935–3941
Setsu N, Kohashi K, Endo M, Yamamoto H, Tamiya S, Takahashi Y, Yamada Y, Ishii T, Matsuda S, Yokoyama R, Iwamoto Y, Oda Y (2013) Phosphorylation of Signal Transducer and Activator of Transcription 3 in Soft Tissue Leiomyosarcoma is Associated with a Better Prognosis. Int J Cancer 132:109–115
Sebestyen A, Sticz TB, Mark A, Hajdu M, Timar B, Nemes K, Nagy N, Varadi Z, Kopper L (2012) Activity and Complexes of mTOR in Diffuse large B-cell Lymphomas–a Tissue Microarray Study. Mod Pathol 25:1623–1628
Tampellini M, Longo M, Cappia S, Bacillo E, Alabiso I, Volante M, Dogliotti L, Papotti M (2007) Co-expression of EGF Receptor, TGFalpha and S6 Kinase is Significantly Associated with Colorectal Carcinomas with Distant Metastases at Diagnosis. Virchows Arch 450:321–328
Chaisuparat R, Rojanawatsirivej S, Yodsanga S (2013) Ribosomal Protein S6 Phosphorylation is Associated with Epithelial Dysplasia and Squamous Cell Carcinoma of the Oral Cavity. Pathol Oncol Res 19:189–193
Baryawno N, Sveinbjörnsson B, Eksborg S, Chen CS, Kogner P, Johnsen JI (2010) Small-molecule Inhibitors of Phosphatidylinositol 3-Kinase/Akt Signaling Inhibit Wnt/beta-Catenin Pathway Cross-talk and Suppress Medulloblastoma Growth. Cancer Res 70:266–276
Noh WC, Mondesire WH, Peng J, Jian W, Zhang H, Dong J, Mills GB, Hung MC, Meric-Bernstam F (2004) Determinants of Rapamycin Sensitivity in Breast Cancer Cells. Clin Cancer Res 10:1013–1023
Maira SM, Stauffer F, Brueggen J, Furet P, Schnell C, Fritsch C, Brachmann S, Chène P, De Pover A, Schoemaker K, Fabbro D, Gabriel D, Simonen M, Murphy L, Finan P, Sellers W, García-Echeverría C (2008) Identification and Characterization of NVP-BEZ235, A New Orally Available Dual phosphatidylinositol 3-Kinase/Mammalian Target of Rapamycin Inhibitor with Potent in vivo Antitumor Activity. Mol Cancer Ther 7:1851–1863
Wolff JE, Brown RE, Buryanek J, Pfister S, Vats TS, Rytting ME (2012) Preliminary Experience with Personalized and Targeted Therapy for Pediatric Brain Tumors. Pediatr Blood Cancer 59:27–33
Acknowledgments
We thank Mária Csorba, Edit Parsch, Zsuzsanna Kaminszky and Renáta Kiss for excellent technical assistance and Dr. J. Silber (University of Washington, Seattle, WA, USA) for providing UW228–2 medulloblastoma cell line.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pócza, T., Sebestyén, A., Turányi, E. et al. mTOR Pathway As a Potential Target In a Subset of Human Medulloblastoma. Pathol. Oncol. Res. 20, 893–900 (2014). https://doi.org/10.1007/s12253-014-9771-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9771-0