Abstract
Modeling the distribution and habitat capacities of key estuarine species can be used to identify hot spots, areas where species density is significantly higher than surrounding areas. This approach would be useful for establishing a baseline for evaluating future environmental scenarios across a landscape. We developed species distribution models for early juvenile life stages of brown shrimp (Farfantepenaeus aztecus), white shrimp (Litopenaeus setiferus), blue crab (Callinectes sapidus), and spotted seatrout (Cynoscion nebulosus) in order to delineate the current coastal hot spots that provide the highest quality habitat conditions for these estuarine-dependent species in Louisiana. Response curves were developed from existing long-term fisheries-independent monitoring data to identify habitat suitability for fragmented marsh landscapes. Response curves were then integrated with spatially explicit input data to generate species distribution models for the coastal region of Louisiana. Using spatial autocorrelation metrics, we detected clusters of suitable habitat across the Louisiana coast, but only 1% of the areas were identified as true hot spots with the highest habitat quality for nekton. The regions identified as hot spots were productive fringing marsh habitats that are considered the most vulnerable to natural and anthropogenic impacts. The species distribution models identify the coastal habitats which currently provide the greatest capacity for key estuarine species and will be used in the Louisiana coastal planning process to evaluate how species distributions may change under various environmental and restoration scenarios.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High primary and secondary productivity stimulated by plumes from the Mississippi and Atchafalaya Rivers support ecologically and economically important nekton (fishes and decapod crustaceans) in coastal Louisiana. Juvenile life stages of several estuarine-dependent species rely on flooded marsh edge habitats for survival and growth and then move into deeper estuarine and shelf waters as they mature and grow large enough to avoid more predators and search for larger prey (Baltz et al. 1998; Chesney et al. 2000; Minello et al. 2003; Rozas and Minello 2010; Rozas and Minello 2015; Zimmerman et al. 2000). However, marshes in coastal Louisiana have shown extensive loss as a result of subsidence, storms, human modifications to the Mississippi River, and other natural and anthropogenic influences (Blum and Roberts 2012; Day et al. 2011), resulting in differing hypotheses on how fisheries may respond in the future (Cowan et al. 2008). As Louisiana enacts large-scale management plans in the region to restore and protect ecosystems (e.g., The Louisiana Comprehensive Master Plan for a Sustainable Coast [herein as Louisiana Master Plan], (CPRA 2012), modeling current species distributions and habitat capacities is needed to identify significant productive areas and to establish a baseline for evaluation of future scenarios across the coast.
Habitat Suitability Index (HSI) models have a long and wide history of use for describing the capacity of aquatic habitats to support key nekton (U.S. Fish and Wildlife Service 1980). They have also been used in determining ecological impacts (Kapustka 2005; Rand and Newman 1998) and defining essential or important habitats in restoration studies because they are simple to construct and communicate, and are solidly supported by species life history information and presence-absence data (Barnes et al. 2006; Barnes et al. 2007). HSI models consist of simplified (usually trapezoidal) relationships that relate key environmental variables to the quality of the habitat for the species. The individual relationships for each environmental variable, termed suitability indices (SI), are standardized on a 0 to 1 scale with 1 being the most favorable conditions and 0 being unsuitable. The relationships may be developed from observations of species presence or abundance sampled over a range of environmental predictor variables. In this way, species presence or abundance (typically as catch per unit effort [CPUE]) is assumed to be already constrained by biotic interactions, limiting resources (i.e., realized Hutchinsonian niche), and suitable environmental conditions (i.e., fundamental Grinellian niche), as described by Guisan and Thuiller (2005) and Pulliam (2000). The integration of HSIs with spatially explicit inputs in a geographic information system (GIS) framework can then be used to produce species distribution models (SDMs), a form of static, comparative models used in lieu of mechanistic models of growth and survival to explain patterns of species occurrence and investigate their response to environmental change (Corsi et al. 2000; Guisan and Zimmermann 2000).
SDMs have been increasingly used in coastal systems to gain insights to species distributions across habitats and environmental gradients (e.g., Elith and Leathwick 2009; Froeschke and Froeschke 2011; Froeschke and Froeschke 2016; Rubec et al. 2016a; Rubec et al. 2016b; Starke et al. 2011). The development of SDMs for estuarine nekton requires consideration of their complex life histories that result in differential use of vegetated and open water habitats by life stage or size (Able 2005). The quantity and quality of suitable habitat are also driven by seasonal and interannual variation in environmental conditions within the estuary (Baltz and Jones 2003). Although individual SDMs promote understanding of species distributions across landscapes, aggregating species-specific SDMs into ecologically similar or economically relevant groups can help to summarize nekton community habitat requirements for coastal systems. Hot spot analysis of these aggregated groups over space can aid in identifying suitable habitats for multiple species with similar habitat requirements. Hot spots are spatially explicit areas where an observation or process differs from the surrounding areas. Identification of hot spots contributes towards understanding ecological processes that generate the typical and atypical patterns in species distributions (Nelson and Boots 2008). Hot spot analysis has been used to identify locations that contain rare or endangered species (Flather et al. 1998; Kissling et al. 2007; Prendergast et al. 1993), abundant taxa (Cocu et al. 2005; Nelson and Boots 2008), or areas that support high species richness (Gaston and David 1994; Gould 2000; Pittman et al. 2007). Application of hot spot analysis to SDMs for ecological and economically important nekton of coastal Louisiana is needed for identifying the important habitats for multiple species and for effectively tracking the changes in these identified areas over time.
In coastal Louisiana, large-scale numerical modeling analyses are currently being used to inform the Louisiana Master Plan by evaluating various environmental scenarios (e.g., sea level rise) and testing different combinations of coastal protection and restoration projects over the next 50 years in order to predict how coastal conditions may change (CPRA 2012). The Louisiana Master Plan is updated every 5 years and includes models that represent hydrologic, water quality, vegetation, and key fish and shellfish populations and communities. Here, we present the two-stage approach for developing new SDMs for the Louisiana Master Plan and using them to identify important coastal habitats for commercially and ecologically important early juvenile stages of nekton. Long-term fisheries-independent monitoring data were used to develop HSIs for the early juvenile life stages of brown shrimp (Farfantepenaeus aztecus), white shrimp (Litopenaeus setiferus), blue crab (Callinectes sapidus), and spotted seatrout (Cynoscion nebulosus). The HSI models were dynamically coupled with the Louisiana Master Plan modeling framework to create a spatially explicit coastwide aggregated SDM. Geospatial analysis of the aggregated SDM was then used to identify estuarine hot spots representative of shared habitat usage and peak intensity of suitable habitats. The outcomes of our study define specific areas of significance for the coastal planning process under existing environmental conditions.
Methods
Developing Response Curves for the Early Juveniles in Coastal Marshes
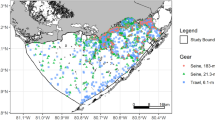
Response curves were constructed using catch data of early juvenile stages collected by the Louisiana Department of Wildlife and Fisheries (LDWF) long-term fisheries-independent coastal monitoring program. LDWF personnel use 15.24-m bag seines to sample small juvenile shrimps, crabs, and fishes monthly to twice per month by collecting a single sample at each site in shallow shoreline and marsh edge habitats (Fig. 1). The seines are deployed perpendicular to the vegetated shoreline in the shallow (≤1.5 m) soft-bottom habitat. The ends of the seine are affixed to posts set 15.24 m from each other at the shoreline or edge; the seine net is dropped 100 m from the shoreline and then two boats reverse from the posts to pull the seine net into the marsh edge (LDWF 2002). The number of fish and invertebrate species collected in the net is recorded and individual lengths and weights are measured for a subsample of up to 50 individuals per species. CPUE for brown shrimp, white shrimp, blue crab, and spotted seatrout was estimated as total catch at each sample event from 1986 to 2014.
An exploratory analysis of the seine data was performed to determine seasonal use of the marsh edge habitat and size range of each species sampled. We estimated the mean monthly CPUE for each year from all samples to determine when each species moves into the estuary to settle in the marsh habitats (Online Resource 1). The species-specific datasets were subset to the months of consistently high catch (Table 1) to construct response curves that reflect the habitat conditions experienced by each of the species when they were present in the marsh habitats and eliminate false zeros in the CPUE data for time periods when early juveniles were not present. Length-frequency distributions of the species were plotted for the months of peak occurrence (Online Resource 2) to determine the range and median size of individuals and to confirm that seines were collecting species representative of the early juvenile life stage. The early juvenile life stage is defined as including the size range (measured in total length = TL or carapace width = CW) between newly metamorphosed juveniles and juveniles large enough to begin moving out of shallow marsh edge habitats and into deeper estuarine waters. Table 1 lists the size range that represents the early juvenile life stage for each species defined in the literature (e.g., Ditty and Shaw 1994; Lassuy 1983; Minello 1999; Patillo et al. 1997).
LDWF personnel measure salinity and temperature at the top and bottom of the water column at each seine sample site (LDWF 2002, Fig. 1). Inspection of the data revealed minimal differences between these measurements because the samples from the shoreline or vegetated edge were taken in shallow (<1.5 m) water. The average of these two measurements was used for each sample site.
The CPUE data for each species were natural log transformed (ln [CPUE + 1]) to help meet the assumptions of homogeneous variance and heteroscedacity for multiple regression analysis of the CPUE with salinity and temperature. Polynomial regression was used to accommodate curvature in the CPUE response and account for interactions between salinity and temperature. Visual inspection of diagnostic plots (e.g., residuals versus predicted values and QQ plots of residuals) to assess homogeneity of variance and linearity of the model revealed the natural log transformation worked generally well to meet the assumptions of the regression analysis.
Salinity and temperature terms, including linear, polynomial, and their two-way interactions, that explained the CPUE for each species at an alpha = 0.05 were selected using PROC GLMSelect (SAS Institute Inc. version 9.4). General linear models were then generated for this reduced set of significant effects for each species using SAS PROC MIXED. The resulting polynomial regression model describes the early juvenile ln(CPUE + 1), V S where S is species, in terms of all significant effects (p < 0.05) from salinity (S) and temperature (T), their squared terms, and their interactions as follows:
Vbrownshrimp = 1.33615 + 0.22030(S) + 0.02229(T) – 0.00629(S2) + 0.00054(T2) – 0.00007(S*T2)Vwhiteshrimp = 2.41242 – 0.03471(S) + 0.009624(T) + 0.01385(S*T) – 0.00264(S2) – 0.00115(T2)-0.00034(S*T)V bluecrab = 0.65264 – 0.03677(S) + 0.06561(T) + 0.000312(S2) – 0.00182(T2)Vspottedseatrout = − 0.20458 + 0.0251(S) + 0.07216(T) – 0.00077(S2) – 0.00000085(S2*T2) – 0.00168(T2)
The regression equations were standardized to a 0–1 scale to provide a water quality suitability index function, WQ s , that can be used with other standardized SIs to generate an overall HSI for the early juvenile stage of each species. Polynomial response functions were standardized by dividing the predicted response variables (V S ) by the maximum predicted values from the surface responses. The maximum predicted V S for each species was found by running the polynomial model through combinations of salinity and temperature that fall within the observed ranges. The maximum response values were 3.51 for early juvenile brown shrimp, 3.25 for early juvenile white shrimp, 1.24 for early juvenile blue crab, and 0.71 for juvenile spotted seatrout.
An additional SI was developed to represent the marsh edge and shallow shoreline habitat at each seine sample site. The SI for marsh vegetation (V m ) was modeled from Minello and Rozas (2002) using the percent of emergent marsh vegetation versus open water within a specified geographic area. The same SI for marsh vegetation was used for each species assuming all four species similarly access and use the shallow marsh habitat and gain the same benefits. The highest V m score (1.0) for each species is assigned when marsh vegetation comprises between 25 and 80% of the area (Fig. 2). Vegetated marsh between 25 and 80% is correlated to high amounts of marsh edge which have been shown to support higher densities of penaeid shrimps and blue crab compared to solid unfragmented marsh and open water (Minello and Rozas 2002). V m predicts habitat capacity to decrease linearly as marsh vegetation decreases below 25% and converts to open water or increases beyond 80% towards solid marsh (Fig. 2).
The suitability response index for salinity and temperature and the suitability index for the percent marsh vegetation were combined. An overall HSI score was thereby generated for the early juvenile stage of each species as follows:
HSIS = (WQS * VM)0.5
Model Coupling and SDMs
The application of HSIs as an SDM for the entire coast required spatially explicit input data to generate HSI scores for the four early juvenile stages of species across coastal Louisiana. The data used as inputs to generate HSI scores (salinity, temperature, percent emergent marsh vegetation) were produced by a coastwide mass-balance model designed for running 50-year simulations, the Integrated Compartment Model (ICM), for the Master Plan (Meselhe et al. 2015). The ICM serves as the central modeling platform for the Louisiana Master Plan and was developed by dynamically coupling three pre-existing models of the coastal Louisiana environment (i.e., hydrodynamic, vegetation, morphology) into one modeling framework. Hydrodynamic data were modeled using a mass-balance link-node model, with the offshore Gulf of Mexico region defining the tidally bounded downstream condition and upstream boundary conditions consisting of freshwater sources (e.g., tributaries and Mississippi River distributaries). Environmental drivers such as precipitation, evapotranspiration, winds, temperatures, and tide levels were the primary data used to drive the hydrodynamic model. The hydrodynamic model output was linked directly to the morphological and vegetation models that predicted the amount of open water to land and the relative success or mortality of vegetation species based on statistical relationships of survival due to salinity and water level variability. Vegetation species were grouped into predominant vegetation types for every 500 m × 500 m grid cell, either fresh forested wetlands, fresh herbaceous marsh, intermediate herbaceous marsh, brackish herbaceous marsh, saline herbaceous marsh, upland, or bare ground. The ICM was calibrated for 2010–2013 and validated against observed data for 2006–2010. Additional details on the development of the ICM, calibration, and validation can be found in a series of technical reports (Meselhe et al. 2015 and attachments therein).
In our study, mean monthly salinity and temperature and annual emergent marsh vegetation (includes all simulated marsh vegetation types from the vegetation model and excludes upland and bare habitats) and open water area were extracted from the 2011, 2012, and 2013 calibration periods of the ICM at the 500 m × 500 m grid cell resolution to represent present-day conditions and account for interannual variability associated with model output. For each year, mean monthly salinity and temperature outputs were averaged over the species-specific months of occurrence (Table 1) analyzed in the polynomial regressions. The species-specific mean seasonal salinity and temperature were then used as inputs for the standardized equations to calculate each WQ S index for each 500 m × 500 m cell in each year. The area of marsh vegetation in each grid cell for each year was used to calculate the V m index. For fresh forested wetlands, the V m index was assigned a default score of 0 as the four species were not sampled in that habitat. The geometric mean of the two indices was then used to generate an HSI score for the early juvenile life stage of each species for the years 2011, 2012, and 2013. Exploratory analysis of the mean salinity and temperature conditions for those years revealed minimal differences among years; thus, a 3-year average of the HSI for each species was used as a representation of their habitat capacity for present-day conditions.
Hot Spot Analysis
The species-specific annual HSI scores resulting from the mean seasonal (Table 1) salinity and temperature and annual percent marsh vegetation were summed for each 500 m × 500 m grid cell to create an aggregate score for the four species (i.e., one score per cell). The Local Moran’s I and Getis-Ord Gi* statistics were used to measure spatial autocorrelation of the aggregated scores among cells (Anselin 1995; Ord and Getis 1995). Local Moran’s I measured similarity of aggregated HSI values between a grid cell and its neighboring cells. The statistic was used to identify significant high-high clusters, i.e., high scores surrounded by other high scores, and significant low-low clusters, i.e., low scores surrounded by other low scores, as an indication of shared habitat usage among species. A 1-km Euclidian distance band was applied to the Local Moran’s I to ensure each cell had one neighbor to test for similarity in neighboring features. Similarly, the Getis-Ord Gi* statistic identified significant clusters of either high or low aggregated HSI scores for a grid cell and its neighboring cells. The Gi* statistic differed from Local Moran’s I in that the sum of scores within a cluster were then compared proportionally to the sum of scores across all grid cells (i.e., the coastwide mean). The statistic identified the significant peak intensities of scores, or “hot spots,” for a grid cell and neighboring cells relative to all cells combined. Likewise, “cold spots” were clusters of grid cells that possessed the significantly lowest scores relative to all other cells. Therefore, the Gi* statistic identified the areas across the Louisiana coast that have the highest (hot spot) and lowest (cold spot) habitat suitability for early juvenile life stages of the estuarine-dependent species. The detection of hot spots can vary in response to the scale of the spatial analysis (Palmer and White 1994). As a result, the Getis-Ord Gi* test was run at different spatial scales by testing five distance bands (1-, 5-, 10-, 15-, and 25-km length) to evaluate peak intensities within the context of neighboring cells and relative to all cells combined. For both the Local Moran’s I and Getis-Ord Gi*, inverse distance squared methods were applied such that the closest neighbors to a cell exert greater influence on the statistic, and the influence decreases sharply as the distance of the neighboring cells increases. z scores were used to represent the statistical significance of the spatial autocorrelation between a cell and its neighboring cells. z scores <−1.96 or >1.96 were considered significant at alpha of 0.05. Negative z scores refer to low-low clusters and cold spots, while positive z scores refer to high-high clusters and hot spots.
Results
Species Response to Environmental Conditions in Marsh and Open Water
Juvenile brown shrimp had the highest suitability at salinities of 10–15 and warm temperatures (>20 °C); the temperature range was indicative of the late spring-early summer months (Fig. 3). At cooler temperatures (<20 °C), the suitability function showed a slightly steeper rate of decline as salinity decreased below 10 or increased above 15 (Fig. 3). The highest suitability index score for juvenile white shrimp was detected at slightly higher salinities (18–22) and cooler temperatures 16–20 °C (Fig. 3) than juvenile brown shrimp. A strong temperature and salinity interaction was detected for juvenile white shrimp; the suitability function sharply decreased as both salinities and temperatures increased. The early juvenile blue crabs that overwinter in the shallow estuaries had the highest suitability in low-salinity waters, and suitability decreased as salinity increased (Fig. 3). The early juvenile spotted seatrout present in the fall had the highest suitability at a similar salinity range as the brown shrimp (13–18) and temperature around 18–22 °C (Fig. 3).
Hot Spot Detection
The aggregated SDMs had the lowest scores in the upper reaches of the estuaries (Fig. 4, top panel), which included non-fragmented marsh, forested wetlands, and low salinity. Moving down the estuaries towards the Gulf of Mexico, a transition from low to high scores was evident for the fringing marshes of the mid and low estuaries that have a high degree of fragmentation and brackish salinities. Moderate scores were observed coastwide in the open water bays and coastal waters extending beyond the barrier islands. The Local Moran’s I statistic detected significant spatial clusters of low and high habitat suitability scores along the coastline (Fig. 4, middle panel). Large bands of fringing marsh in the mid and lower estuaries were identified as significant high-high clusters (i.e., grid cells with high suitability adjacent to other grid cells with high suitability). Most of the significant low-low clusters were detected for interior fresh marshes or fresh forested wetlands, areas along the coast, or in areas with unfragmented (solid) marsh on constructed barrier islands and sand bars (Fig. 4, middle panel). The Getis-Ord Gi* detection capability was scale-dependent; significant hot and cold spots were detectable in select areas across the coast only with distance bands ≥15 km (Fig. 4, bottom panel and Fig. 5). The habitat suitability scores for grid cells and their neighboring grid cells at these larger distance bands were found to be significantly higher (hot) than the mean of all areas along the Louisiana coast (Fig. 4, bottom panel). At smaller distance bands (<15 km), hot spots could not be detected against the coastwide mean within the 95% confidence interval (Fig. 5). Although Local Moran’s I indicated high-high clusters of cells within the smaller distance band (Fig. 4, middle panel), the statistic does not compare the scores to the coastwide mean, as is done in the Getis-Ord Gi* statistic. At the largest distance band of 25 km, only 1% of the coast was classified as a significant hot spot (Fig. 5). These areas were characterized by grid cells with the highest habitat suitability for the species aggregate (Fig. 6, top panel), with brackish salinity, temperatures around 23–24 °C, and a high degree of marsh fragmentation showing about 30–60% marsh vegetation for a given area (Fig. 6). The cold spots identified at the 25-km distance band were comprised of grid cells with zero salinity (fresh water) and primarily consisted of nearly solid marsh or fresh forested wetlands with very little open water habitat (Fig. 6).
Results of a aggregated SDM scores for early juvenile brown shrimp, white shrimp, blue crab, and spotted seatrout, b significant clusters identified by the Local Moran’s I statistic at the 1-km scale, and c significant hot spots identified by the Getis-Ord Gi* at the 25-km scale. Significance was attributed to p < 0.05. Hot spots are located in Terrebonne Bay (1), Barataria Bay (2), and Biloxi Marsh (3)
Cumulative probabilities of Getis-Ord Gi* z scores calculated for different distance bands applied to the aggregated SDM scores in coastal Louisiana. Vertical reference lines mark one standard deviation used for significance testing. Shaded areas in gray denote where the probability function of the largest distance band, 25 km, intersected the vertical reference lines and indicate the total proportion of all grid cells that are statistically significant hot or cold spots at alpha = 0.05
Box plot of cumulative HSI scores, salinity, temperature, and percent emergent vegetation for areas identified by Getis-Ord Gi* as hot and cold spots. Dashed line indicates mean; solid line indicates median; points represent 5th and 95th percentiles; sample size (n) = 2913 for hot spots; n = 13,650 for cold spots
Discussion
The role of salinity and temperature in defining the distribution of species within estuaries is complex (Rozas et al. 2005). Selection of habitats is determined not only by the species physiological preferences and tolerances, but also efforts to minimize predation risk, and the quality and availability of prey to maximize growth potential (e.g., Dunson and Travis 1991; Huey 1991; Lima and Dill 1990; Martin 2001; McIvor and Odum 1988; Werner et al. 1983). As a result, large ranges of salinity and temperature preferences by life stage are reported for the four species analyzed in our study (Clark et al. 2004; Flaherty and Guenther 2011; Froeschke and Froeschke 2011; Minello 1999; Patillo et al. 1997; Rozas and Minello 2011; Thomas 1999). For instance, the dome-shaped salinity and temperature response for juvenile brown shrimp is consistent with preferences previously reported in the literature (Adamack et al. 2012; Foret et al. 2003; Rozas and Minello 2011; Patillo et al. 1997) and also supports earlier HSI models developed for juvenile brown shrimp (Baltz 2012; Turner and Brody 1983). Others have reported high densities or relative preferences of juvenile brown shrimp in salinities >15 (Clark et al. 1999; Clark et al. 2004; Doerr et al. 2016; Longley 1994; Minello 1999), while some studies using abundance data have found high densities at low salinities (Thomas 1999). Seasonal timing of juvenile brown shrimp migrations in and out of the estuaries along the northern Gulf of Mexico, the use of different gear types and periods of collection, as well as species accessibility to different habitats within the estuaries could also describe the differences in apparent salinity preferences. However, the LDWF seine data used in our study were subset to the months when the highest abundances of early juvenile brown shrimp are present in Louisiana coastal marshes.
The predicted highest suitability of salinity and temperature for juvenile white shrimp was similar to the combined suitability reported in tidal marsh creeks in Georgia (Webb and Kneib 2002) and marshes of the northern Gulf of Mexico (Baker and Minello 2010; Rozas and Minello 2010). As with brown shrimp, some discrepancies exist in the literature with regards to apparent salinity preference of white shrimp. Juvenile white shrimp abundance has been reported to be high in low-salinity waters (Sable and Villarrubia 2011; Thomas 1999); other studies have not found a significant relationship between salinity and white shrimp preferences (Clark et al. 1999; Doerr et al. 2016). Marsh complexity may be more important than salinity in explaining the variation in their distribution (Webb and Kneib 2002).
Blue crabs are considered euryhaline and like white shrimp may spend their entire life cycle within Louisiana estuaries. Similarly, spotted seatrout generally spend their entire life cycle in inshore coastal waters within and near their natal estuary (Comyns et al. 2008; Saucier and Baltz 1993; Wagner 1973); less than 30% of the adult population moves between estuaries (Callihan et al. 2013; Hendon et al. 2002; Killiam et al. 1992). The response functions for both species agreed with the literature. Juvenile blue crab abundance is typically the highest in low-salinity waters (Flaherty and Guenther 2011; Patillo et al. 1997; Rozas and Minello 2006; Sable and Villarrubia 2011), and the highest suitability on the response surface fit to the LDWF CPUE data was also found in low salinities (<5). Likewise, our salinity suitability ranges and dome-shaped response fit to juvenile spotted seatrout CPUE also agrees well with the bell-shaped response function for seatrout described by Kupschus (2003) and Froeschke and Froeschke (2011).
Modeling species distribution is confounded by the fact that many species regularly occur in unsuitable habitat, while oftentimes, species are absent in otherwise suitable habitats (Pulliam 2000). This pattern was observed in the LDWF long-term dataset where a proliferation of zero CPUE values were observed under suitable salinity and temperature conditions while high catches were apparent at the extreme ends of the salinity and temperature conditions. These apparent data anomalies could be due to other environmental variables not included in the current HSIs such as tides (water levels) or climatic events that can affect the timing of migration and settlement location of juveniles in estuaries (Piazza et al. 2010). Changes in water levels influence the timing and degree of marsh flooding and ultimately, whether densities of nekton will be concentrated along the marsh edge or on the marsh surface (i.e., out of reach of the sampling gear; Peterson and Turner 1994). The analysis generally accounted for the seasonal timing of the early juveniles settling in the shallow shoreline and marsh habitats of the estuaries, but there is still interannual variation in environmental conditions as well as timing of recruitment that is not accounted for in the data analysis. For instance, recruitment into suitable habitat may be limited by factors occurring during early life stages, such as community interactions (e.g., predator-prey interactions) or abiotic factors that influence the number of offspring produced (Etherington and Eggleston 2000; Garvey et al. 2002; Haas et al. 2001; Kneib 1997). Alternatively, source-sink theory suggests that unsuitable habitats serve as sinks if immigration from source areas is sufficiently high (Crowder et al. 2000; Dunning et al. 1992). Nonetheless, the response curves and spatially integrated SDM agree well with the habitat preferences and known distributions of these four species.
We assumed all emergent marsh vegetation was functionally equivalent and provided the same habitat capacity for the early juvenile life stage of all the species in this analysis. This general assumption and suitability function (Fig. 2) for the early juvenile life stage of these four estuarine-dependent species that are highly abundant in Louisiana coastal estuaries is well supported by over 30 years of observations and habitat comparisons (Baltz et al. 1998; Beck et al. 2001; Chesney et al. 2000; Minello 1999; Minello et al. 1994; Minello et al. 2003; Peterson and Turner 1994; Rozas and Minello 1998; Rozas and Minello 2010; Rozas and Reed 1993; Rozas and Zimmerman 2000; Zimmerman et al. 2000; Zimmerman and Minello 1984).
The spatial analyses of the aggregated SDM identified clusters of high suitability for fringing marshes (typical salinity range of 5 to 20) characterized by shallow, turbid waters, micro-tidal ranges (∼30 cm), and complex substrate and micro-topography from Spartina stems and rhizomes (Chesney et al. 2000). Rozas and Minello (2010) and Mace and Rozas (Population dynamics and secondary production of juvenile white shrimp (Litopenaeus setiferus) along an estuarine salinity gradient, unpublished) have similarly characterized marshes in the brackish and saline zones of estuaries to be the zones of highest shrimp abundance. Peak intensities (i.e., hot spots) of aggregated SDM scores were detected for three regions within Terrebonne Bay, Barataria Bay, and Biloxi Marsh (Fig. 4) that have historically been known for, or designated as, highly productive nursery grounds for penaeid shrimps, blue crab, and early juvenile stages of fish species such as spotted seatrout, Atlantic croaker (Micropogonias undulates), and red drum (Scianops ocellatus) (Baltz et al. 1993; Brown et al. 2013; Minello 1999; O’Connell et al. 2009; Rozas and Minello 2010; Rozas and Reed 1993). These regions are also some of the most rapidly changing wetland areas in coastal Louisiana (Couvillion et al. 2011) and are currently within areas targeted for mid- to large-scale river diversions and marsh restoration projects by the Louisiana Master Plan (CPRA 2012). The general agreement between the reported literature and this current SDM, which only accounts for salinity and temperature conditions in marsh edge habitats, supports its use as a simplified tool to evaluate whether annual changes in the distribution of the early juvenile stage of these four species may occur in response to proposed restoration projects, as well as sea level rise scenarios and other climate change effects.
Conclusions
Long-term fisheries-independent monitoring data were used to develop nonlinear response curves and describe habitat suitability for the early juvenile life stage of brown shrimp, white shrimp, blue crab, and spotted seatrout given the temperature and salinity conditions in the Louisiana coastal marshes. Integrating the response curves with spatially explicit data resulted in an aggregated SDM that represents a relative suitability for juveniles of these species across coastal habitats. As part of the Louisiana Master Plan, 50-year simulations will be run to examine the SDM response to environmental scenarios, such as sea level rise, and restoration actions. The outcomes of this research determined coastal regions of significance for key nekton and will assist in establishing a baseline of species distributions for this large-scale coastal planning process. The spatial autocorrelation metrics can also be applied to future simulated SDM to detect clusters and hot spots of habitat suitability under future scenarios of environmental change and to examine whether current hot spots shift, disappear, or are newly detected across a changing landscape. Our method to identify significant hot spots of highly productive nursery grounds is a novel approach that can be used to indicate how groups of species may shift under future climate and restoration scenarios.
References
Able, K.W. 2005. A re-examination of fish estuarine dependence: evidence for connectivity between estuarine and ocean habitats. Estuarine, Coastal and Shelf Science 64: 5–17.
Adamack, A.T., C.A. Stow, D.M. Mason, L.P. Rozas, and T.J. Minello. 2012. Predicting the effects of freshwater diversions on juvenile brown shrimp growth and production: a Bayesian-based approach. Marine Ecology Progress Series 444: 155–173.
Anselin, L. 1995. Local indicators of spatial association-LISA. Geographical Analysis 27: 93–115.
Baker, R., and T.J. Minello. 2010. Growth and mortality of juvenile white shrimp Litopenaeus setiferus in a marsh pond. Marine Ecology Progress Series 413: 95–104.
Baltz, D.M. 2012. Appendix D16—brown shrimp habitat suitability index. Baton Rouge, LA: Coastal Protection and Restoration Authority.
Baltz, D.M., and R.F. Jones. 2003. Temporal and spatial patterns of microhabitat use by fishes and decapod crustaceans in a Louisiana estuary. Transactions of the American Fisheries Society 132: 662–678.
Baltz, D.M., C.F. Rakocinski, and J.W. Fleeger. 1993. Microhabitat use by marsh-edge fishes in a Louisiana estuary. Environmental Biology of Fishes 36: 109–126.
Baltz, D.M., J.W. Fleeger, and C.F. Rakocinski. 1998. Food, density, and microhabitat: factors affecting growth and recruitment potential of juvenile saltmarsh fishes. Environmental Biology of Fishes 53: 89–103.
Barnes, T.K., F.J. Mazzotti, L.G. Pearlstine, and A.K. Volety. 2006. Habitat evaluation in coastal Southwest Florida: a policy decision model for the blue crab, Callinectes sapidus. Florida Scientist 69: 140–151.
Barnes, T.K., A.K. Volety, K. Chartier, F.J. Mazzotti, and L. Pearlstine. 2007. A habitat suitability index model for the eastern oyster (Crassostrea virginica), a tool for restoration of the Caloosahatchee Estuary, Florida. Journal of Shellfish Research 26: 949–959.
Beck, M.W., K.L. Heck, K.W. Able, D.L. Childers, D.B. Eggleston, B.M. Gillanders, B. Halpern, C.G. Hays, K. Hoshino, T.J. Minello, R.J. Orth, P.F. Sheridan, and M.P. Weinstein. 2001. The identification, conservation, and management of estuarine and marine nurseries for fishes and invertebrates. Bioscience 51: 633–641.
Blum, M.D., and H.H. Roberts. 2012. The Mississippi Delta region: past, present, and future. Annual Review of Earth and Planetary Sciences 40: 655–683.
Brown, H., T.J. Minello, G.A. Matthews, M. Fisher, E.J. Anderson, R. Reidel, D.L. Leffler 2013. Nekton from fishery independent trawl samples in Estuaries of the US Gulf of Mexico: a Comparative Assessment of Gulf Estuarine Systems (CAGES). U.S. Deptartment of Commerce NOAA Technical Memorandum NMFS-SEFSC-647.
Callihan, J.L., J.H. Cowan Jr., and M.D. Harbison. 2013. Sex differences in residency of adult spotted seatrout in a Louisiana estuary. Marine and Coastal Fisheries 5: 79–92.
Chesney, E.J., D.M. Baltz, and R.G. Thomas. 2000. Louisiana estuarine and coastal fisheries and habitats: perspectives from a fish’s eye view. Ecological Applications 10: 350–366.
Clark, R.D., T.J. Minello, J.D. Christensen, P.A. Caldwell, M.E. Monaco, G.A. Matthews 1999. Modeling nekton habitat use in Galveston Bay, Texas: An approach to define essential fish habitat (EFH). Vol. Biogeography Program Technical Report Number 17. NOAA/NOS Biogeography Program, Silver Spring, MD, and NMFS, Galveston, TX.
Clark, R.D., J.D. Christensen, M.E. Monaco, P.A. Caldwell, G.A. Matthews, and T.J. Minello. 2004. A habitat-use model to determine essential fish habitat for juvenile brown shrimp (Farfantepenaeus aztecus) in Galveston Bay, Texas. Fishery Bulletin 102: 264–277.
Coastal Protection and Restoration Authority (CPRA). 2012. Louisiana’s comprehensive master plan for a sustainable coast. Baton Rouge, LA: Coastal Protection and Restoration Authority.
Cocu, N., R. Harrington, M. Hullé, and M.D.A. Rounsevell. 2005. Spatial autocorrelation as a tool for identifying the geographical patterns of aphid annual abundance. Agricultural and Forest Entomology 7: 31–43.
Comyns, B.H., C.F. Rakocinski, M.S. Peterson, and A.M. Shiller. 2008. Otolith chemistry of juvenile spotted seatrout Cynoscion nebulosus reflects local natal regions of coastal Mississippi, USA. Marine Ecology Progress Series 371: 243–252.
Corsi, F., de J. Leeuw, A. Skidmore 2000. Modeling species distributions with GIS. In Research techniques in animal ecology: controversies and consequences, ed. Luigi Boitani and T. K. Fuller, 389–434. Methods and Cases in Conservation Science. New York: Columbia University Press.
Couvillion, B.R., J.A. Barras, G.D. Steyer, W. Sleavin, M. Fischer, H. Beck, N. Trahan, B. Griffin, D. Heckman 2011. Land area change in coastal Louisiana from 1932 to 2010. Scientific Investigations Map 3164. U.S. Geological Survey.
Cowan, J.H. Jr., C.B. Grimes, and R.F. Shaw. 2008. Life history, history, hysteresis, and habitat changes in Louisiana’s coastal ecosystem. Bulletin of Marine Science 83: 197–215.
Crowder, L.B., S.J. Lyman, W.F. Figueira, and J. Priddy. 2000. Source-sink population dynamics and the problem of siting marine reserves. Bulletin of Marine Science 66: 799–820.
Day, J.W. Jr., G.P. Kemp, D.J. Reed, D.R. Cahoon, R.M. Boumans, J.M. Suhayda, and R. Gambrell. 2011. Vegetation death and rapid loss of surface elevation in two contrasting Mississippi delta salt marshes: the role of sedimentation, autocompaction and sea-level rise. Ecological Engineering 37: 229–240.
Ditty, J.G., R.F. Shaw 1994. Preliminary guide to the identification of the early life history stages of sciaenid fishes from the western central Atlantic. NOAA Tech. Memo. NMFSSEFSC-349.
Doerr, J.C., H. Liu, and T.J. Minello. 2016. Salinity selection by juvenile brown shrimp (Farfantepenaeus aztecus) and white shrimp (Litopenaeus setiferus) in a gradient tank. Estuaries and Coasts 39: 829–838.
Dunning, J.B., B.J. Danielson, and H.R. Pulliam. 1992. Ecological processes that affect populations in complex landscapes. Oikos 65: 169.
Dunson, W.A., and J. Travis. 1991. The role of abiotic factors in community organization. The American Naturalist 138: 1067.
Elith, J., and J.R. Leathwick. 2009. Species distribution models: ecological explanation and prediction across space and time. Annual Review of Ecology, Evolution, and Systematics 40: 677–697.
Etherington, L.L., and D.B. Eggleston. 2000. Large-scale blue crab recruitment: linking postlarval transport, post-settlement planktonic dispersal, and multiple nursery habitats. Marine Ecology Progress Series 204: 179–198.
Flaherty, K.E., and C.B. Guenther. 2011. Seasonal distribution and abundance of blue crabs (Callinectes sapidus) in the Tampa Bay estuary. Gulf of Mexico Science 2: 91–110.
Flather, C.H., M.S. Knowles, I.A. Kendall 1998. Threatened and endangered species geography. BioScience: 365–376.
Foret, J.D., J.N. Nyman, L.P. Rozas, K.H. Rose, J.H. Cowan, D. Baltz 2003. Habitat Use Module. In Coastal Louisiana Ecosystem Assessment and Restoration (CLEAR) Model of Louisiana Coastal Area (LCA) Comprehensive Ecosystem Restoration Plan. Volume I: Tasks 1–8., ed. R.R. Twiley. Baton Rouge, LA.
Froeschke, J.T., and B.F. Froeschke. 2011. Spatio-temporal predictive model based on environmental factors for juvenile spotted seatrout in Texas estuaries using boosted regression trees. Fisheries Research 111: 131–138.
Froeschke, J.T., and B.F. Froeschke. 2016. Two-stage boosted regression tree model to characterize southern flounder distribution in Texas estuaries at varying population sizes. Marine and Coastal Fisheries 8: 222–231.
Garvey, J.E., R.A. Stein, R.A. Wright, and M.T. Bremigan. 2002. Exploring ecological mechanisms underlying largemouth bass recruitment along environmental gradients. American Fisheries Society Symposium 31: 7–23.
Gaston, K.J., and R. David. 1994. Hotspots across Europe. Biodiversity Letters 2: 108–116.
Gould, W. 2000. Remote sensing of vegetation, plant species richness, and regional biodiversity hotspots. Ecological Applications 10: 1861–1870.
Guisan, A., and W. Thuiller. 2005. Predicting species distribution: offering more than simple habitat models. Ecology Letters 8: 993–1009.
Guisan, A., and N.E. Zimmermann. 2000. Predictive habitat distribution models in ecology. Ecological Modelling 135: 147–186.
Haas, H.L., E.C. Lamon III, K.A. Rose, R.F. Shaw 2001. Environmental and biological factors associated with the stage-specific abundance of brown shrimp (Penaeus aztecus) in Louisiana: applying a new combination of statistical techniques to long-term monitoring data. Canadian Journal of Fisheries and Aquatic Sciences: 2258–2270.
Hendon, J.R., J.R. Warren, J.S. Franks, and M.V. Buchanan. 2002. Movements of spotted seatrout (Cynoscion nebulosus) in Mississippi coastal waters based on tag-recapture. Gulf of Mexico Science 20: 91–97.
Huey, R.B. 1991. Physiological consequences of habitat selection. The American Naturalist 137: S91.
Kapustka, L.A. 2005. Assessing ecological risks at the landscape scale: opportunities and technical limitations. Ecology and Society 10: 11.
Killiam, K.A., R.J. Hochberg, and E.C. Rzemien. 1992. Spotted seatrout (Cynoscion nebulosus). In Synthesis of basic life historites of Tampa Bay species, 340–357. St. Petersburg, FL: Tampa Bay National Estuary Program Technical Publication 10–92.
Kissling, M.L., M. Reid, P.M. Lukacs, S.M. Gende, and S.B. Lewis. 2007. Understanding abundance patterns of a declining seabird: implications for monitoring. Ecological Applications 17: 2164–2174.
Kneib, R.T. 1997. Early life stages of resident nekton in intertidal marshes. Estuaries 20: 214–230.
Kupschus, S. 2003. Development and evaluation of statistical habitat suitability models: an example based on juvenile spotted seatrout Cynoscion nebulosus. Marine Ecology Progress Series 265: 197–212.
Lassuy, D.R. 1983. Species profiles: life histories and environmental requirements (Gulf of Mexico)—brown shrimp. U.S. Fish and Wildlife Service Biological Report (TREL-82-4 No. FWS/OBS-82/11.1) (p. 15). U.S. Army Corps of Engineers.
Lima, S.L., and L.M. Dill. 1990. Behavioral decisions made under the risk of predation: a review and prospectus. Canadian Journal of Zoology 68: 619–640.
Longley, W.L. 1994. Freshwater inflows to Texas bays and estuaries: ecological relationships and methods for determination of needs. Texas Water Development Board and Texas Parks and Wildlife Department.
Louisiana Department of Wildlife and Fisheries (LDWF). 2002. Marine Fisheries Division Field Procedure Manual. Ver. 02–1. Baton Rouge, LA.
Martin, T.E. 2001. Abiotic vs. biotic influences on habitat selection of coexisting species: climate change impacts? Ecology 82: 175–188.
McIvor, C.C., and W.E. Odum. 1988. Food, predation risk, and microhabitat selection in a marsh fish assemblage. Ecology 69: 1341–1351.
Meselhe, E., D.J. Reed, and A.O. Grace. 2015. 2017 coastal master plan: appendix C: modeling. Version I. (p. 99). Baton Rouge, Louisiana: Coastal Protection and Restoration Authority.
Minello, T.J. 1999. Nekton densities in shallow estuarine habitats of Texas and Louisiana and the identification of essential fish habitat. American Fisheries Society Symposium g: 43–75.
Minello, T.J., and L.P. Rozas. 2002. Nekton in gulf coast wetlands: fine-scale distributions, landscape patterns, and restoration implications. Ecological Applications 12: 441–455.
Minello, T.J., R.J. Zimmerman, and R. Medina. 1994. The importance of edge for natant macrofauna in a created salt marsh. Wetlands 14: 184–198.
Minello, T.J., K.W. Able, M.P. Weinstein, and C.G. Hays. 2003. Salt marshes as nurseries for nekton: testing hypotheses on density, growth and survival through meta-analysis. Marine Ecology Progress Series 246: 39–59.
Nelson, T.A., and B. Boots. 2008. Detecting spatial hot spots in landscape ecology. Ecography 31: 556–566.
O’Connell, M.T., A.M.U. O’Connell, and R.W. Hastings. 2009. A meta-analytical comparison of fish assemblages from multiple estuarine regions of southeastern Louisiana using a taxonomic-based method. Journal of Coastal Research SI54: 101–112.
Ord, J.K., and A. Getis. 1995. Local spatial autocorrelation statistics: distributional issues and an application. Geographical Analysis 27: 286–306.
Palmer, M.W., P.S. White 1994. Scale dependence and the species-area relationship. American Naturalist: 717–740.
Patillo, M.E., T.E. Czapla, D.M. Nelson, M.E. Monaco 1997. Distribution and abundance of fishes and invertebrates in Gulf of Mexico estuaries volume II: Species life history summaries. 11. NOAA’s Estuarine Living Marine Resources Program. U.S. Department of Commerce, Nation Oceanic and Atmosperic Administration, National Ocean Service.
Peterson, G.W., and R.E. Turner. 1994. The value of salt marsh edge vs interior as a habitat for fish and decapod crustaceans in a Louisiana tidal marsh. Estuaries 17: 235–262.
Piazza, B.P., M.K. La Peyre, and B.D. Keim. 2010. Relating large-scale climate variability to local species abundance: ENSO forcing and shrimp in Breton Sound, Louisiana, USA. Climate Research 42: 195–207.
Pittman, S.J., J.D. Christensen, C. Caldow, C. Menza, and M.E. Monaco. 2007. Predictive mapping of fish species richness across shallow-water seascapes in the Caribbean. Ecological Modelling 204: 9–21. doi:10.1016/j.ecolmodel.2006.12.017.
Prendergast, J.R., R.M. Quinn, J.H. Lawton, B.C. Eversham, and D.W. Gibbons. 1993. Rare species, the coincidence of diversity hotspots and conservation strategies. Letters to Nature 365: 335–337.
Pulliam, H.R. 2000. On the relationship between niche and distribution. Ecology Letters 3: 349–361.
Rand, G.M., and J.R. Newman. 1998. The applicability of habitat evaluation methodologies in ecological risk assessment. Human and Ecological Risk Assessment: An International Journal 4: 905–929. doi:10.1080/10807039891284875.
Rozas, L.P., and T.J. Minello. 1998. Nekton use of salt marsh, seagrass, and nonvegetated habitats in a south Texas (USA) estuary. Bulletin of Marine Science 63: 481–501.
Rozas, L.P., and T.J. Minello. 2006. Nekton use of Vallisneria americana Michx. (wild celery) beds and adjacent habitats in coastal Louisiana. Estuaries and Coasts 29: 297–310.
Rozas, L.P., and T.J. Minello. 2010. Nekton density patterns in tidal ponds and adjacent wetlands related to pond size and salinity. Estuaries and Coasts 33: 652–667. doi:10.1007/s12237-009-9258-5.
Rozas, L.P., and T.J. Minello. 2011. Variation in penaeid shrimp growth rates along an estuarine salinity gradient: implications for managing river diversions. Journal of Experimental Marine Biology and Ecology 397: 196–207. doi:10.1016/j.jembe.2010.12.003.
Rozas, L.P., and T.J. Minello. 2015. Small-scale nekton density and growth patterns across a saltmarsh landscape in Barataria Bay, Louisiana. Estuaries and Coasts 38: 2000–2018.
Rozas, L.P., and D.J. Reed. 1993. Nekton use of marsh-surface habitats in Louisiana (USA) deltaic salt marshes undergoing submergence. Marine Ecology Progress Series 96: 147–157.
Rozas, L.P., and R.J. Zimmerman. 2000. Small-scale patterns of nekton use among marsh and adjacent shallow nonvegetated areas of the Galveston Bay Estuary, Texas (USA). Marine Ecology Progress Series 193: 217–239.
Rozas, L.P., T.J. Minello, I. Munuera-Fernández, B. Fry, and B. Wissel. 2005. Macrofaunal distributions and habitat change following winter-spring releases of freshwater into the Breton Sound estuary, Louisiana (USA). Estuarine, Coastal and Shelf Science 65: 319–336.
Rubec, P.J., R. Kiltie, E. Leone, R.O. Flamm, L. McEachron, and C. Santi. 2016a. Using delta-generalized additive models to predict spatial distributions and population abundance of juvenile pink shrimp in Tampa Bay, Florida. Marine and Coastal Fisheries 8: 232–243.
Rubec, P.J., J. Lewis, D. Reed, C. Santi, R.H. Weisberg, L. Zheng, C. Jenkins, C.F. Ashbaugh, C. Lashley, and S. Versaggi. 2016b. Linking oceanographic modeling and benthic mapping with habitat suitability models for pink shrimp on the West Florida Shelf. Marine and Coastal Fisheries 8: 160–176.
Sable, S., and C. Villarrubia. 2011. Analysis of fisheries-independent data for evaluation of species distribution responses to the Caernarvon Freshwater Diversion. Final report. Baton Rouge, LA: Coastal Protection and Restoration Authority.
Saucier, M.H., and D.M. Baltz. 1993. Spawning site selection by spotted seatrout, Cynoscion nebulosus, and black drum, Pogonias cromis, in Louisiana. Environmental Biology of Fishes 36: 257–272.
Starke, A., J.S. Levinton, and M. Doall. 2011. Restoration of Crassostrea virginica (Gmelin) to the Hudson River, USA: a spatiotemporal modeling approach. Journal of Shellfish Research 30: 671–684.
Thomas, R.G. 1999. Fish habitat and coastal restoration in Louisiana. American Fisheries Society Symposium 22: 240–251.
Turner, R.E., M.S. Brody 1983. Habitat suitability index models: Northern Gulf of Mexico brown shrimp and white shrimp. Fish and Wildlife Service.
U.S. Fish and Wildlife Service. 1980. Habitat evaluation procedures (HEP). Department of the Interior: Division of Ecological Services, US Fish and Wildlife Service.
Wagner, P.R. 1973. Seasonal biomass, abundance, and distribution of estuarine dependent fishes in the Caminada Bay system of Louisiana. PhD dissertation. Baton Rouge, LA: Louisiana State University.
Webb, S.R., and R.T. Kneib. 2002. Abundance and distribution of juvenile white shrimp Litopenaeus setiferus within a tidal marsh landscape. Marine Ecology-Progress Series 232: 213–223.
Werner, E.E., J.F. Gilliam, D.J. Hall, and G.G. Mittelbach. 1983. An experimental test of the effects of predation risk on habitat use in fish. Ecology 64: 1540.
Zimmerman, R.J., and T.J. Minello. 1984. Densities of Penaeus aztecus, Penaeus setiferus, and other natant macrofauna in a Texas salt marsh. Estuaries 7: 421–433.
Zimmerman, R.J., T.J. Minello, and L.P. Rozas. 2000. Salt marsh linkages to productivity of penaeid shrimps and blue crabs in the northern Gulf of Mexico. In Concepts and controversies in tidal marsh ecology, ed. M.P. Weinstein and D.A. Kreeger, 293–314. Dordrecht: Kluwer Academic Publishers.
Acknowledgments
We thank the Louisiana Department of Wildlife and Fisheries who collected and provided all fisheries-independent data used in the analysis. We also thank Harry Blanchet, Mandy Green, Lisa Landry, Brian Lezina, Leland Moss, and Denise Reed who contributed to the development of results and thinking presented here. Tim Carruthers provided helpful comments on an earlier draft that resulted in additional refinements and clarity in the final manuscript. We appreciated the constructive comments by the associate editor, Lawrence P. Rozas, and two anonymous reviewers. This work was funded in part by the Coastal Protection and Restoration Authority of Louisiana under Cooperative Endeavor Agreement Number 2503-12-58, Task Order No. 03
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Lawrence P. Rozas
Rights and permissions
About this article
Cite this article
Hijuelos, A.C., Sable, S.E., O’Connell, A.M. et al. Application of Species Distribution Models to Identify Estuarine Hot Spots for Juvenile Nekton. Estuaries and Coasts 40, 1183–1194 (2017). https://doi.org/10.1007/s12237-016-0199-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-016-0199-5