Abstract
Recent studies have suggested that large rivers play important roles in mobilizing and transporting black carbon (BC) from land to the ocean. However, the influence of the Changjiang and Huanghe, the two largest rivers in China, on the fate of BC has not been determined. In this paper, we present measurements of BC in both the dissolved and particulate phases in the Changjiang and Huanghe Rivers and in the coastal waters of the East China Sea (ECS). Our results show that dissolved black carbon (DBC) accounted for 3.0 ± 0.4 % and 4.8 ± 3.6 % of the total DOC pool in the Changjiang and Huanghe Rivers and 3.4 ± 0.6 % of the DOC pool in the coast of the ECS. In addition, particulate black carbon (PBC) accounted for 13 ± 0.9 % and 22 ± 11 % of the POC pool in the Changjiang and Huanghe Rivers, respectively. We calculate that the Changjiang and Huanghe transported 4.7 × 1010 gC and 1.7 × 109 gC of DBC, and 2.0 × 1011 gC and 1.2 × 1010 gC of PBC to the ECS and Bohai Sea in 2015. The large amounts of BC transported by the two rivers represent a major sink of anthropogenically derived organic carbon and could have a significant impact on the ecosystem and carbon cycling in China’s marginal seas.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Black carbon (BC) is a class of combustion-derived, complex mixture of organic compounds that have a range of different compositions and reactivities along the combustion continuum (Masiello, 2004), and it is widely dispersed in the natural environment, especially in soils and marine sediments (LaFlamme and Hites, 1978; Goldberg, 1985). BC is defined as condensed carbonaceous residue produced from incomplete combustion of fossil fuel and biomass (Goldberg, 1985). It is estimated that globally, about 12–24 Tg (Tg = 1012 g) BC is produced from fossil fuel combustion (Penner et al., 1993) and 50–270 Tg BC is generated from biomass burning annually (Kuhlbusch and Crutzen, 1995). In a recent study, Santín et al. (2015) estimated that production of pyrogenic C from boreal forest wildfires could reach 100 Tg C year−1. After production, most BC will be preserved in soils as charcoal (Skjemstad et al., 2002; Cusack et al., 2012; Santín et al., 2015), but some very fine particulate BC could be transported long distances by rivers and atmospheric aerosols and eventually become deposited in the ocean (Masiello and Druffel, 1998; Mitra et al., 2002; Wagner et al., 2015; Wang et al., 2015). Many previous studies have determined the quantities of BC in coastal and deep sea sediments and have found that BC could account for 20–50 % and 5–20 % of organic carbon preserved in coastal and abyssal sediments (Verardo and Ruddiman, 1996; Gustafsson and Gschwend, 1998; Masiello and Druffel, 1998; Middelburg et al., 1999; Lohmann et al., 2009; Wang and Li, 2007; Huang et al., 2015). BC thus represents a significant fraction of the anthropogenic organic carbon sink in the global carbon cycle (Schmidt, 2004; Santín et al., 2015).
The presence of BC in the natural environment has been a topic of great concern based not only on its destructive nature to ecosystems and the serious health risks it poses to human beings (Rust et al., 2004; Lohmann et al., 2005; Kang et al., 2009) but also on its impact on climate and on its role as an important sink for the anthropogenic carbon (Druffel, 2004; Shrestha et al., 2010; Stubbins et al., 2012). Due to its stable chemical nature, BC could be preserved in soils and marine sediments for millions of years without being degraded (Masiello and Druffel, 1998; Santín et al., 2015). In the last 10 years, most studies conducted on BC have focused on its solid phase, such as in soils, marine sediments, and sinking particles (Czimczik and Masiello, 2007; Wang and Li, 2007; Ohlson et al., 2009; Cusack et al., 2012; Coppola et al., 2014). BC in the dissolved phase, however, has received less attention (Mannino and Harvey, 2004; Dittmar, 2008). Recent studies have emphasized the presence and potential importance of dissolved BC (DBC) as a missing pool in the global carbon cycle (Ziolkowski and Druffel, 2010; Dittmar et al., 2012; Ding et al., 2013; Coppola et al., 2015; Wagner et al., 2015). Jaffé et al. (2013) examined DBC in 15 world large rivers and concluded that about 26.5 ± 1.8 Tg of DBC could be transported annually by rivers into the ocean, and DBC accounts for about 10 % of the global riverine flux of dissolved organic carbon (DOC). Clearly, rivers play an important role in mobilizing BC from the land to the ocean.
To quantitatively determine BC in the natural environment, however, a big challenge still remains largely due to the inconsistency of the different methods employed by different laboratories (Currie et al., 2002; Hammes et al., 2007). Due to the complexity of BC associated with different types of environmental samples, there has been no single standard technique used to separate and quantify BC. Hammes et al. (2007) compared seven methods used by 17 laboratories for 12 different environmental samples. They found that for the same samples, different methods could give a wide range of BC values because different types of BC had been measured. For example, the thermal-oxidation method usually identifies highly condensed aromatic structures as BC, so it is best suited for quantifying the most condensed forms of BC and gives relatively lower BC values. The chemical oxidation method, on the other hand, is good for soot, char, and bituminous coal BC determination but often overestimates BC values due to incomplete removal of non-BC in sediment and soil samples (Masiello, 2004; Hammes et al., 2007). Nevertheless, in spite of the differences of the BC values reported by different methods, the presence of BC in the environment still warrants it being an important component of the global carbon cycle.
The Changjiang and Huanghe are the two largest rivers in China and the third and sixth longest rivers in the world (Milliman and Meade, 1983). The two rivers together drain one third of China’s land and deliver about 1.9 and 1.6 Tg of POC and DOC annually to the coasts of the East China Sea (ECS) and Bohai Sea (Wang et al., 2012). The middle and lower basins of the Changjiang and Huanghe also pass through the most populated southeast and northeast regions of China, so both rivers were severely influenced by human activities (Zhang et al., 2013, 2014). It is expected that these two large rivers could transport significant amounts of BC to the coasts of the ECS and Bohai Sea, where high BC contents have been measured in the sediments (Wang and Li, 2007; Kang et al., 2009; Huang et al., 2015). However, no studies have been conducted to determine the quantity and flux of BC in the two rivers. In this paper, we present the first BC data set for both dissolved and particulate phases in the Changjiang and Huanghe Rivers. The goal of this study was to quantitatively determine and compare the BC transported in the Changjiang and Huanghe Rivers and to assess its significance to the riverine carbon cycle.
Methods
Study Site and Sample Collection
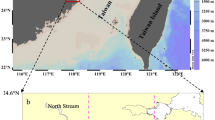
Water samples for this study were collected from the lower reach of the Huanghe River, the lower reach of the Changjiang River and its estuary, and the coast of the ECS, as shown in Fig. 1. We conducted a 12-month study for the Huanghe River. River water was collected on a floating bridge about 30 km upstream from the Huanghe Estuary in the middle of each month during 2015. Fifteen liters of water was collected using a stainless steel bucket (pre-cleaned and rinsed several times with river water before taking samples) and stored in well-cleaned polyethylene bottles (cleaned with soapy water then soaked with 10 % HCl solution for 24 h, and then rinsed with Milli-Q water 4 times before use). Water was filtered through 0.7-μm Whatman GF/F filters (prebaked in 550 °C for 5 h). The filtered water was then acidified using high-purity HCl to pH 2 and kept refrigerated in pre-baked glass bottles for further processing. Suspended particles were frozen for solid phase analysis. Six water samples were collected in the Changjiang River and its estuary and the coast of the ECS. River waters were collected in the main stream using a fishing boat on July 21, 2015 (CR03), and March 20, 2016 (CR01 and CR02). Seawater from the coast of the ECS (P02, P04, and P06) was collected during a cruise onboard the R/V Dongfanghong-2 in October 2015. Subsurface water (1.5 m) was collected using an oil-free in-situ pump (SFP-900P). After collection, 15 L of river water and 50 L of seawater were filtered through pre-baked 0.7-μm Whatman GF/F filters immediately and acidified to pH 2, and the water was kept in polyethylene bottles (pre-cleaned as described above) at a low temperature for further processing. Suspended particles from the Changjiang River sample were frozen for solid phase analysis.
DOC Extraction by SPE Method
In order to analyze DBC, DOC needs to be extracted first from the water samples. We used a solid phase extraction (SPE) method for concentrating DOC from the water samples. Recent studies have shown that the SPE technique is a simple and effective method for extraction of DOC for DBC analysis in both river water and seawater (De jesus, 2008; Dittmar et al., 2008; Ding et al., 2013; Coppola et al., 2015; Wagner et al., 2015). Studies have shown that the PPL cartridges had the highest extraction efficiency and were contamination-free for DOC compared with other types of SPE cartridges (Dittmar, 2008; Dittmar et al., 2008). For our study, we used pre-packed 60-ml volume PPL cartridges (Agilent Technologies Mega Bond Elute) that contain 5 g styrene divinyl benzene polymer as sorbent (pore size 150 Å) with the capacity to process a large volume of water samples. Before use, the cartridge was well cleaned according to the manufacturer’s guidelines. It was first rinsed with two bed-volumes of high purity methanol (B&J), which was then followed by at least 2000 mL Milli-Q high purity water to clean the sorbent. For sample extraction (within a week after collection), acidified water was kept in a 2-L glass funnel, dropped directly into the top of the cleaned PPL cartridge, and passed through. The extraction flow rate was kept at 5 ml/min. Permeate passing through the cartridge was not saved, but we collected samples for DOC analysis to calculate the extraction efficiency. For all samples, 15 L of river water and 50 L seawater were extracted.
After extraction, the cartridge sorbent was rinsed with one bed-volume of 0.01 M HCL twice to remove salts, then dried with high-purity nitrogen gas. Once the cartridge was dry, 80 mL high-purity methanol was used to elute the DOC extracted from the sorbent into a glass bottle. We determined that >98 % of DOC absorbed on the sorbent was eluted in the methanol phase, consistent with the results of other studies using the same SPE sorbent (Dittmar et al., 2008; Coppola et al., 2015). The extracted DOC in methanol was then condensed to 5 mL using a Buchi Multivapor P-12 Evacuator. The condensed DOC was defined as SPE-DOC and kept in the refrigerator for further DBC determination. All glassware used in sample processing and storage were soaked in 10 % HCL acid for 24 h, rinsed with Milli-Q water and baked at 550 °C for 5 h to remove any organic carbon prior to use.
Blanks associated with SPE extraction were determined. We considered two types of blanks during the extraction: a sorbent blank and a methanol elution blank. To test the blanks from the sorbent, we continuously collected Milli-Q water permeate for every 500 mL that passed through the pre-cleaned cartridge for the same extraction volume (15 L) to measure DOC concentrations. To check the blanks during methanol elution, we collected 80 mL of methanol eluent, dried it with high purity N2 gas, re-dissolved it in high purity Milli-Q water, and measured its DOC concentration. In both cases, we found no detectible DOC (within the detection limit ~3 μM) associated with either the sorbent or methanol, consistent with the results reported in previous studies (Dittmar, 2008; Dittmar et al., 2008).
DOC and BC Measurement
The concentration of DOC was analyzed by the high temperature catalytic oxidation (HTCO) method using a Shimadzu TOC-L analyzer equipped with an ASI-L auto-sampler. The instrument was calibrated using 5-point calibration curves derived from a carbon standard solution of potassium hydrogen phthalate (KHP) and DOC values were checked against low-carbon water and deep-seawater reference materials (CRM, University of Miami, Rosenstiel School of Marine and Atmospheric Sciences). Blank subtraction was carried out using high purity Milli-Q water that was analyzed before every five samples. Average blanks associated with DOC measurements were about 5 μM, and analytic precision on triplicate injections was <3 %.
The concentrations of both DBC and particulate black carbon (PBC) were determined by the thermal-oxidation method based on Gustafsson and Gschwend (1998) and Gustafsson et al. (2001). Previous studies have shown that this method was precise and reproducible for BC analysis in marine sediments and particles (Gustafsson et al., 2001; Reddy et al., 2002, Wang and Li, 2007). It has also been used to determine DBC in HMW-DOM samples with good recovery, tested using standard BC materials (Mannino and Harvey, 2004). It should be pointed out that by using the thermal-oxidation method, we likely determine the soot-type highly condensed aromatic BC in our samples (Hammes et al., 2007). For DBC analysis, 1.0–2.0 mL of condensed SPE-DOC was added into a 9-mm OD × 200-mm quartz tube (prebaked at 850 °C for 2 h) and dried with a high-purity N2 stream. The quartz tube was then placed in an oven and the SPE-DOC was thermally oxidized at 375 °C for 24 h with a continuous air supply to remove non-BC OC. The organic carbon left after thermal oxidation was defined as DBC. For PBC determination, suspended particles were first dried at 60 °C and ground to fine powder. About 1–2 g ground particles were placed in a very thin layer (2–3 mm) in petri dishes and heated at 375 °C for 24 h in an oven with continuous air supply. Organic carbon left after the thermal oxidation was defined as PBC. Following thermal oxidation, DBC was oxidized again in evacuated, flame-sealed quartz tubes (with CuO and Ag wire added) at 850 °C for 2 h in a muffle furnace (Druffel et al., 1992). Particles before and after thermal oxidation were acidified with 10 % HCl to remove inorganic carbon. After being dried in the oven at 60 °C, 200–500 mg was placed in prebaked 9-mm OD quartz tubes and flame-sealed under a vacuum (with CuO and Ag wire added). Both POC and PBC in the sealed quartz tubes were combusted to CO2 in a muffle furnace at 850 °C for 2 h. The resultant CO2 from combustion was collected cryogenically and quantitatively measured on vacuum line. The amounts of DBC, POC, and PBC were calculated based on the volume of the resultant CO2. As part of this study, we also measured carbon isotope (14C and 13C) compositions for DOC, DBC, POC, and PBC. These results are presented in a separate paper (Wang et al., 2016).
Results and Discussion
DBC in the Changjiang and Huanghe Rivers
The results of all chemical measurements are summarized in Table 1. The extraction efficiency of SPE-DOC was 59 ± 6 % for the fresh river water and 42 ± 3 % for the coastal seawater. We observed that the extraction efficiency of seawater was actually about the same as for the river freshwater until 20 L of seawater passed through, when the extraction efficiency decreased (data not shown). This was probably due to the high amount of salt retained on the sorbent at that point. The 42 ± 3 % was an overall extraction efficiency for seawater calculated based on a 50 L extraction volume. These results, in general, are in good agreement with the values obtained by Dittmar et al. (2008) and Coppola et al. (2015). Using the same PPL cartridge, Dittmar et al. (2008) reported 62–65 % DOC extraction efficiency for river and salt marsh waters and 43 % for high-salinity seawater DOC. Coppola et al. (2015) determined a 43 ± 6 % extraction efficiency for seawater using a modified SPE method with the same sorbent.
The average abundances of DBC determined for the samples accounted for 3.0 ± 0.4, 4.8 ± 3.6, and 3.4 ± 0.6 % of the total DOC pool in the Changjiang and Huanghe Rivers and ECS coastal seawaters (Table 1 and Fig. 2). For the Huanghe River, however, greatly variable DBC content was observed for different months in 2015. The values of DBC/DOC ranged from 1.2 ± 0.1 % in December to 13 ± 0.3 % in April. March (8.2 ± 0.4 %) and August (9.3 ± 0.5 %) also exhibited higher DBC values compared with other months. The DBC contents (3.4 ± 0.6 %) in the three coastal seawater samples are comparable, close to the DBC value (3.0 ± 0.4 %) in the Changjiang River (Fig. 2).
The DBC values in the Changjiang and Huanghe rivers and the coastal waters of the ECS that we determined are comparable to the results reported in other environments. Using the same SPE method, Dittmar (2008) reported DBC contents that accounted for 2.6 and 0.9 % of the DOC pools in the Gulf of Mexico and the Atlantic Ocean. DBC in Atlantic open ocean waters was much lower than that in coastal seawater. More recently, using the same sorbent SPE method, Coppola et al., (2015) extracted 4.2 ± 1.0 % DBC in the DOC pool for the North Pacific surface water. However, Dittmar (2008) and Coppala et al. (2015) used benzene polycarboxylic acids (BPCAs) in the chemical oxidation method to quantify DBC in the SPE extracted DOC, which is a different approach from the thermal-oxidation method we applied. The DBC/DOC values we determined in the Changjiang and Huanghe Rivers (3.0–4.8 %) and coastal waters of ECS (3.4 ± 0.6 %) are actually quite similar to the values they reported. As reviewed by Hammes et al. (2007), the thermal-oxidation method mainly identifies highly condensed aromatic structure BC and usually gives lower BC values. The BPCAs method also chemically oxidizes condensed aromatic BC in SPE-DOC to individual BPCAs which are subsequently analyzed using HPLC (Dittmar, 2008; Ziolkowski et al., 2011). These two methods, although one determines aromatic bulk BC and one measures molecular markers, likely determine structurally similar BC, resulting in comparable BC values in the different water samples. Even using a different ultrafiltration method, Mannino and Harvey (2004) found that the DBC comprised 8.9 ± 6.5 and 4–7 % of the DOC pool in the Delaware Estuary and the coastal Atlantic Ocean. These similarities in DBC contents in a different river, coastal water, and seawater could suggest that a similar mechanism or process controls the dissolution and cycling of BC in the DOC pool. As reported by Jaffé et al. (2013), a strong positive linear correlation (r 2 = 0.95) exists between DBC and DOC concentrations in the 15 world rivers they studied. They estimated that about 26.5 ± 1.8 Tg of DBC could be transported by the world’s rivers into the ocean annually and this DBC could account for about 10 % of the global riverine flux of DOC. Our results clearly demonstrate that both the Changjiang and Huanghe Rivers play important roles in mobilizing and transporting DBC to coastal waters. As shown in Fig. 3a, a strong positive correlation (r 2 = 0.86) between DBC and DOC exists in the Changjiang and its estuarine coastal waters. However, for the Huanghe River, such a relationship is insignificant (r 2 = 0.15, Fig. 3b). This is largely due to the seasonal variations of DBC observed in the Huanghe River (Fig. 2), suggesting that regional point source contributions of DBC could be important (Hockaday et al., 2007; Dittmar et al., 2012). The decomposition processes and rates of DBC and DOC could also be different (Bird et al., 1999; Nguyen et al., 2010). Recent studies have shown that photodegradation preferentially removes DBC in river and coastal waters (Stubbins et al., 2012; Wagner and Jaffé, 2015), so the abundance of DBC in a particulate river basin may not be necessarily controlled directly by DOC concentration.
PBC in the Changjiang and Huanghe Rivers
In contrast to DBC, the PBC contents were much higher in the POC pools in the two rivers (Table 1). PBC accounted for 21.8 ± 11.1 % of the POC in the Huanghe and 13 ± 0.9 % of POC in the Changjiang. The monthly distributions of PBC in the Huanghe River also showed large variations during 2015, with high values in April (41.9 %) and June (45.2 %) and lower values in September (13.4 %) and October (9.0 %), respectively (Fig. 2). The PBC in the Changjiang River (13 %) was lower, but similar to the September and October values in the Huanghe River.
The amount of PBC we determined accounted for 13 and 22 ± 11 % of the POC in the Changjiang and Huanghe Rivers. Obviously, BC in the suspended particles in the two rivers is much higher than DBC in the dissolved phase. The Huanghe River POC contained higher PBC than the Changjiang River POC. This is in good agreement with the results we reported previously for BC measured in the coastal sediments of China’s marginal seas (Wang and Li, 2007; Kang et al., 2009). In the Huanghe River-influenced Bohai Sea, BC contents in the surface sediments ranged from 27 to 41 %, which is much higher than the range of 5–26 % in the Changjiang River-influenced ECS coastal sediments. As compared with the values reported for the other environments, it appears that particulate organic matter serves as an important matrix for transporting BC. In their earlier study, Masiello and Druffel (2001) measured PBC in POC samples collected in the Santa Clara River in California (USA). They found that 7.9–17 % of the POC was in the form of PBC. For the Mississippi River Estuary, Mitra et al. (2002) reported that 16–17 % of river POC and 25–28 % of the shelf water POC were identified as PBC in 1999. These values are comparable to the PBC values we measured for the Changjiang and Huanghe Rivers. The relatively high PBC values in the Huanghe River are not surprising considering that the Huanghe is one of the most turbid rivers in the world (Milliman and Meade, 1983). Its flow rate is only 1 % of the Changjiang River, but the average value of annual total suspended sediment (TSS) in the Huanghe River was 2.52 ± 2.98 kg/1000 L, 19 times higher than the value of 0.134 ± 0.067 kg/1000 L in the Changjiang River. The POC content of the TSS in the Huanghe River, however, was much lower (0.48 ± 0.14 %) compared to the average value (1.21 ± 0.21 %) in the Changjiang (Wang et al., 2012). Also, the radiocarbon ages of POC in the Huanghe River were much older (average 6170 years BP) than the POC ages (average 920 years BP) in the Changjiang River (Wang et al., 2012). Most of the particles transported in the Huanghe are derived from the highly decomposed soil, clay minerals, and quaternary loess in the middle basin of the river (Zhang et al., 1995). These fine particles could have a high adsorption capacity for BC. Cusack et al. (2012) have reported that strong adsorption by minerals was a major control for BC preservation in soils. Direct evidence is given by a positive correlation between POC and PBC in the Changjiang and Huanghe Rivers, as shown in Fig. 3c, indicating that PBC is strongly bound to POC and mineral surfaces (Czimczik and Masiello, 2007; Cusack et al., 2012). However, it should be pointed out that the PBC contents in the months of April (41.9 %) and June (45.2 %) in the Huanghe River seem unusually high. We do not have a clean explanation for these two high values but expect that they could be affected by some local point sources of BC, or some non-BC organic matter was not totally removed during thermal-oxidation process for these two samples (Hammes et al., 2007).
Flux of DBC and PBC in the Changjiang and Huanghe Rivers
Based on the DBC and PBC measured for the Huanghe River and its flow rate recorded at the Lijin Hydrographic Station (60 km upstream from the estuary) (www.sdhh.gov.cn/hhsq), we calculated monthly fluxes of DBC and PBC and plotted them in Fig. 4. The flow rate showed a very high peak in July that was caused by an anthropogenically controlled high flood event (Fig. 4a). Since 2002, annual water management regulations during June–July require opening the dam gate at the Xiaolangdi Reservoir, which is 600 km upstream from the estuary, to let a high flow of water remove the sediment deposited on the river bed during previous months. Therefore, a high load of sediment was observed during this high flood period. The relatively high flow rate during April to June was related to the rainy season. The calculated monthly fluxes ranged from 0.13–5.92 × 108 gC for DBC and 0.3–32.7 × 108 gC for PBC in the Huanghe River during 2015. The flux of PBC was much higher than that of DBC, and it mainly occurred in the high flow period during April to the end of July (Fig. 4b). The annual flux of DBC and PBC was 1.7 × 109 gC and 1.2 × 1010 gC, and this accounted for 5.4 and 28 % of the DOC and POC fluxes in the Huanghe River in 2015 (Xue, 2016). For the Changjiang River, since we only measured three DBC values and one PBC value, we use the value of 3.0 ± 0.4 and 13.0 ± 0.9 % to represent the average DBC and PBC contents in the Changjiang River. Based on the annual fluxes of DOC (1.58 × 1012 gC) and POC (1.52 × 1012 gC) we measured in 2009 for the river (Wang et al., 2012). We calculated that the fluxes of DBC and PBC in the Changjiang could reach 4.7 × 1010 gC and 2.0 × 1011 gC annually. The PBC flux in the Changjiang (2.0 × 1011 gC) is comparable with the PBC flux (5 × 1011 gC) measured for the Mississippi River in 1999 (Mitra et al., 2002). These large fluxes of DBC and PBC carried by the Changjiang and Huanghe rivers could have a major influence on carbon cycling and preservation in the Bohai Sea and ECS. Our radiocarbon measurements of DBC and PBC collected in this study have demonstrated that DBC and PBC transported by the Changjiang and Huanghe Rivers represent two distinctive BC pools with PBC (2550–12,600 years BP) being much older than DBC (475–1510 years BP) (Wang et al., 2016). Using 14C measurements of organic biomarkers (n-alkanes), Tao et al. (2015) estimated that 10–31 % of the POC in the Huanghe River was BC derived from fossil fuel combustion. Due to its stable chemical nature (Bird et al., 1999; Cusack et al., 2012), it is expected that most of the PBC will be deposited and preserved in coastal sediments, consistent with the high BC contents measured in the sediments of the Bohai Sea and ECS (Wang and Li, 2007; Kang et al., 2009; Huang et al., 2015). Thus, PBC transported by the Changjiang and Huanghe Rivers represents an important sink of anthropogenically derived carbon that could be preserved in sediments for millennia. It is not well known at present whether riverine aged DBC could be microbially degraded or photo-decomposed when it enters the ocean. A previous study has shown that >2 % of the DOC in the ocean has a heat-induced molecular signature (Dittmar and Paeng, 2009), suggesting that some riverine DBC could last for a long time in the ocean, and thus could contribute to the old DOC pool (Coppola et al., 2015; Wang et al., 2016). Future studies are certainly needed to address this question.
References
Bird, M.I., C. Moyo, E.M. Veenendaal, L. Lloyd, and P. Frost. 1999. Stability of elemental carbon in a savanna soil. Global Biogeochemical Cycles 13(4): 923–932.
Coppola, A.I., B.D. Walker, and E.R. Druffel. 2015. Solid phase extraction method for the study of black carbon cycling in dissolved organic carbon using radiocarbon. Marine Chemistry 177: 697–705.
Coppola, A.I., L.A. Ziolkowski, C.A. Masiello, and E.R. Druffel. 2014. Aged black carbon in marine sediments and sinking particles. Geophysical Research Letters 41(7): 2427–2433.
Currie, L.A., B.A. Benner Jr., J.D. Kessler, and D.B. Klinedinst. 2002. A critical evaluation of interlaboratory data on total, elemental, and isotopic carbon in the carbonaceous particle reference material, NIST SRM 1649a. Journal of Research of the National Institute of Standards and Technology 107(3): 279.
Cusack, D.F., O.A. Chadwick, W.C. Hockaday, and P.M. Vitousek. 2012. Mineralogical controls on soil black carbon preservation. Global Biogeochemical Cycles 26(2). doi:10.1029/2011GB004109.
Czimczik, C.I., and C.A. Masiello. 2007. Controls on black carbon storage in soils. Global Biogeochemical Cycles 21(3). doi:10.1029/2006GB002798.
De Jesus, R.P. 2008. Natural abundance radiocarbon studies of dissolved organic carbon (DOC) in the marine environment, PhD thesis, Univ. of Calif., San Diego.
Ding, Y., Y. Yamashita, W.K. Dodds, and R. Jaffé. 2013. Dissolved black carbon in grassland streams: is there an effect of recent fire history? Chemosphere 90(10): 2557–2562.
Dittmar, T. 2008. The molecular level determination of black carbon in marine dissolved organic matter. Organic Geochemistry 39(4): 396–407.
Dittmar, T., B. Koch, N. Hertkorn, and G. Kattner. 2008. A simple and efficient method for the solid-phase extraction of dissolved organic matter (SPE-DOM) from seawater. Limnology and Oceanography: Methods 6(6): 230–235.
Dittmar, T., and J. Paeng. 2009. A heat-induced molecular signature in marine dissolved organic matter. Nature Geoscience 2(3): 175–179.
Dittmar, T., J. Paeng, T.M. Gihring, I.G. Suryaputra, and M. Huettel. 2012. Discharge of dissolved black carbon from a fire-affected intertidal system. Limnology and Oceanography 57(4): 1171.
Druffel, E.R. 2004. Comments on the importance of black carbon in the global carbon cycle. Marine Chemistry 92(1): 197–200.
Druffel, E.R., P.M. Williams, J.E. Bauer, and J.R. Ertel. 1992. Cycling of dissolved and particulate organic matter in the open ocean. Journal of Geophysical Research: Oceans 97(C10): 15639–15659.
Goldberg, E.D. 1985. Black carbon in the environment: properties and distribution. New York: Wiley.
Gustafsson, Ö., T.D. Bucheli, Z. Kukulska, M. Andersson, C. Largeau, J.N. Rouzaud, and T.I. Eglinton. 2001. Evaluation of a protocol for the quantification of black carbon in sediments. Global Biogeochemical Cycles 15(4): 881–890.
Gustafsson, Ö., and P.M. Gschwend. 1998. The flux of black carbon to surface sediments on the New England continental shelf. Geochimica et Cosmochimica Acta 62(3): 465–472.
Hammes, K., M.W. Schmidt, R.J. Smernik, L.A. Currie, W.P. Ball, T.H. Nguyen, and G. Cornelissen. 2007. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochemical Cycles 21(3). doi:10.1029/2006GB002914.
Hockaday, W.C., A.M. Grannas, S. Kim, and P.G. Hatcher. 2007. The transformation and mobility of charcoal in a fire-impacted watershed. Geochimica et Cosmochimica Acta 71(14): 3432–3445.
Huang, L., J. Zhang, Y. Wu, and J. Wang. 2015. Distribution and preservation of black carbon in the East China Sea sediments: perspectives on carbon cycling at continental margins. Deep Sea Research Part II 124: 43–52.
Jaffé, R., Y. Ding, J. Niggemann, A.J. Vähätalo, A. Stubbins, R.G.M. Spencer, J. Campbell, and T. Dittmar. 2013. Global charcoal mobilization from soils via dissolution and riverine transport to the oceans. Science 340(6130): 345–347.
Kang, Y., X.C. Wang, M.H. Dai, H. Feng, A. Li, and Q. Song. 2009. Black carbon and polycyclic aromatic hydrocarbons (PAHs) in surface sediments of China’s marginal seas. Chinese Journal of Oceanology and Limnology 27: 297–308.
Kuhlbusch, T., and T. Crutzen. 1995. Toward a global estimate of black carbon in residues of vegetation fires representing a sink of atmospheric CO2 and a source of O2. Global Biogeochemical Cycles 9(4): 491–501.
Laflamme, R.E., and R.A. Hites. 1978. The global distribution of polycyclic aromatic hydrocarbons in recent sediments. Geochimica et Cosmochimica 42(3): 289–303.
Lohmann, R., K. Bollinger, M. Cantwell, J. Feichter, I.F. Bruns, and M. Zabel. 2009. Fluxes of soot black carbon to South Atlantic sediments. Global Biogeochemical Cycles 23(1). doi:10.1029/2008GB003253.
Lohmann, R., J.K. MacFarlane, and P.M. Gschwend. 2005. Importance of black carbon to sorption of native PAHs, PCBs, and PCDDs in Boston and New York harbor sediments. Environmental Science and Technology 39(1): 141–148.
Mannino, A., and H.R. Harvey. 2004. Black carbon in estuarine and coastal ocean dissolved organic matter. Limnology and Oceanography 49(3): 735–740.
Masiello, C., and E.R.M. Druffel. 1998. Black carbon in deep-sea sediments. Science 280(5371): 1911–1913.
Masiello, C.A., and E.R.M. Druffel. 2001. Carbon isotope geochemistry of the Santa Clara River. Global Biogeochemical Cycles 15(2): 407–416.
Masiello, C.A. 2004. New directions in black carbon organic geochemistry. Marine Chemistry 92(1–4): 201–213.
Middelburg, J.J., J. Nieuwenhuize, and P. van Breugel. 1999. Black carbon in marine sediments. Marine Chemistry 65(3): 245–252.
Milliman, J.D., and R.H. Meade. 1983. World-wide delivery of river sediment to the oceans. The Journal of Geology 91: 1–21.
Mitra, S., T.S. Bianchi, B.A. McKee, and M. Sutula. 2002. Black carbon from the Mississippi River: quantities, sources, and potential implications for the global carbon cycle. Environmental Science and Technology 36(11): 2296–2302.
Nguyen, B.T., J. Lehmann, W.C. Hockaday, S. Joseph, and C.A. Masiello. 2010. Temperature sensitivity of black carbon decomposition and oxidation. Environmental Science and Technology 44(9): 3324–3331.
Ohlson, M., B. Dahlberg, T. Økland, K.J. Brown, and R. Halvorsen. 2009. The charcoal carbon pool in boreal forest soils. Nature Geoscience 2(10): 692–695.
Penner, J.E., H. Eddleman, and T. Novakov. 1993. Toward the development of a global inventory for black carbon emissions. Atmospheric Environment 27(8): 1277–1295.
Reddy, C.M., A. Pearson, L. Xu, A.P. McNichol, B.A. Benner, S.A. Wise, and T.I. Eglinton. 2002. Radiocarbon as a tool to apportion the sources of polycyclic aromatic hydrocarbons and black carbon in environmental samples. Environmental Science and Technology 36(8): 1774–1782.
Rust, A.J., R.M. Burgess, A.E. McElroy, M.G. Cantwell, and B.J. Brownawell. 2004. Influence of soot carbon on the bioaccumulation of sediment-bound polycyclic aromatic hydrocarbons by marine benthic invertebrates: an interspecies comparison. Environmental Toxicology and Chemistry 3(11): 2594–2603.
Santín, C., S.H. Doerr, C.M. Preston, and G. González-Rodríguez. 2015. Pyrogenic organic matter production from wildfires: a missing sink in the global carbon cycle. Global Change Biology 21(4): 1621–1633.
Schmidt, M.W. 2004. Biogeochemistry: carbon budget in the black. Nature 427(6972): 305–307.
Skjemstad, J.O., D.C. Reicosky, A.R. Wilts, and J.A. McGowan. 2002. Charcoal carbon in US agricultural soils. Soil Science Society of America Journal 66(4): 1249–1255.
Shrestha, G., S.J. Traina, and C.W. Swanston. 2010. Black carbon’s properties and role in the environment: a comprehensive review. Sustainability 2(1): 294–320.
Stubbins, A., E. Hood, P.A. Raymond, G.R. Aiken, R.L. Sleighter, P.J. Hernes, and H.A. Abdulla. 2012. Anthropogenic aerosols as a source of ancient dissolved organic matter in glaciers. Nature Geoscience 5(3): 198–201.
Tao, S., T.I. Eglinton, D.B. Montluçon, C. McIntyre, and M. Zhao. 2015. Pre-aged soil organic carbon as a major component of the Yellow River suspended load: regional significance and global relevance. Earth and Planetary Science Letters 414: 77–86.
Verardo, D.J., and W.F. Ruddiman. 1996. Late Pleistocene charcoal in tropical Atlantic deep-sea sediments: climatic and geochemical significance. Geology 24(9): 855–857.
Wagner, S., K.M. Cawley, F.L. Rosario-Ortiz, and R. Jaffé. 2015. In-stream sources and links between particulate and dissolved black carbon following a wildfire. Biogeochemistry 124(1–3): 145–161.
Wagner, S., and R. Jaffé. 2015. Effect of photodegradation on molecular size distribution and quality of dissolved black carbon. Organic Geochemistry 86: 1–4.
Wang, X.C., and A. Li. 2007. Preservation of black carbon in the shelf sediments of the East China Sea. Chinese Science Bulletin 52(22): 3155–3161.
Wang, X.C., C.L. Xu, E.M. Druffel, Y.J. Xue, and Y. Z. Qi. 2016. Two black carbon pools transported by the Changjiang and Huanghe Rivers. Global biogeochemical cycles, (in review).
Wang, X.C., H. Ma, R. Li, Z. Song, and J. Wu. 2012. Seasonal fluxes and source variation of organic carbon transported by two major Chinese rivers: the Yellow River and Changjiang (Yangtze) river. Global Biogeochemical Cycles 26(2). doi:10.1029/2011GB004130.
Wang, X.C., T. Ge, C. Xu, Y. Xue, and C. Luo. 2015. Carbon isotopic (14C and 13C) characterization of fossil-fuel derived dissolved organic carbon in wet precipitation in Shandong Province, China. Journal of Atmospheric Chemistry: 73, 207–221. doi:10.1007/s10874-015-9323-3.
Xue, Y. J. 2016. Carbon isotopic (13C, 14C) studies of the sources, seasonal variation and flux of organic carbon transported by the Huanghe River, MS thesis, Ocean University of China (in Chinese).
Zhang, L.J., M. Xue, M. Wang, W.J. Cai, L. Wang, and Z.G. Yu. 2014. The spatiotemporal distribution of dissolved inorganic and organic carbon in the main stem of the Changjiang (Yangtze) river and the effect of the three gorges reservoir. Journal of Geophysical Research: Biogeosciences 119(5): 741–757.
Zhang, L.J., L. Wang, W.J. Cai, D.M. Liu, and Z.G. Yu. 2013. Impact of human activities on organic carbon transport in the Yellow River. Biogeosciences 10(4): 2513–2524.
Zhang, J., W.W. Huang, R. Letolle, and C. Jusserand. 1995. Major element chemistry of the Huanghe (Yellow River), China-weathering processes and chemical fluxes. Journal of Hydrology 168(1): 173–203.
Ziolkowski, L.A., and E.R. Druffel. 2010. Aged black carbon identified in marine dissolved organic carbon. Geophysical Research Letters 37(16). doi:10.1029/2010GL043963.
Ziolkowski, L.A., A.R. Chamberlin, J. Greaves, and E.R.M. Druffel. 2011. Quantification of black carbon in marine systems using the benzene polycarboxylic acid method: a mechanistic and yield study. Limnology and Oceanography: Methods 9: 140–149.
Acknowledgments
We thank Hailong Zhang for help with sample collection in the Changjiang River. We also thank Dr. Coppola at UCI for sharing her detailed SPE method for DOC extraction. We appreciate the two anonymous reviewers for their valuable and constructive comments. Financial support for this work was provided by the National Natural Science Foundation of China (Grant Nos 91428101, 41476057, and 41521064).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Zhanfei Liu
Rights and permissions
About this article
Cite this article
Xu, C., Xue, Y., Qi, Y. et al. Quantities and Fluxes of Dissolved and Particulate Black Carbon in the Changjiang and Huanghe Rivers, China. Estuaries and Coasts 39, 1617–1625 (2016). https://doi.org/10.1007/s12237-016-0122-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-016-0122-0