Abstract
The use of tidal currents by fishes for movements to and from onshore spawning, foraging, and nursery grounds is well documented. However, fishes’ use of the water column in tidal currents frequently exceeding 1.5 m · s-1 is largely unknown. With growing interest in extracting energy from the tides, understanding animal use of these dynamic environments has become essential to determining environmental effects of tidal energy devices. To assess the effects of a tidal energy device on fishes, we used down-looking single-beam hydroacoustic technology to collect pre-deployment data on the presence and vertical distribution of fishes at a pilot project site and a control site in Cobscook Bay, ME. Twenty-four-hour stationary surveys were conducted in each season of 2010 and 2011. Relative fish density and vertical distribution were analyzed for variation with respect to site, year, month, and diel and tidal cycles. A seasonal pattern in fish density was apparent in both years at both sites, with maxima in spring and late fall. Fish density was generally highest near the sea floor. Diel changes in vertical distribution were frequently observed, but changes in distribution related to tidal cycle were inconsistent. Results from the project and control sites were very similar, demonstrating that the control site provides a reference for quantifying changes in fish density and vertical distribution related to the tidal device. This approach and baseline dataset will be used to compare hydroacoustic data collected at the project and control sites after device deployment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tidal currents help shape coastal marine environments and play an essential role in the life cycles of many marine and diadromous fishes. Numerous fish species use the tides to gain access to valuable intertidal foraging habitat (Dadswell and Rulifson 1994; Hartill et al. 2003; Krumme 2004; Ribeiro et al. 2006), actively feed when moving against the tidal flow (Krumme and Saint-Paul 2003), or traverse several kilometers over the course of a tidal cycle by moving with the current (Aprahamian et al. 1998; Sakabe and Lyle 2010). Species demonstrated to select tides to aid their movements include American eel (Anguilla rostrata; McCleave and Kleckner 1982), Atlantic cod (Gadus morhua; Arnold et al. 1994), Atlantic salmon (Salmo salar; Stasko 1975; Aprahamian et al. 1998), sea trout (Salmo trutta; Moore et al. 1998), plaice (Pleuronectes platessa; de Veen 1978; Greer Walker et al. 1978), Atlantic mackerel (Scomber scombrus; Castonguay and Gilbert 1995), and Atlantic herring (Clupea harengus; Lacoste et al. 2001).
Areas of extreme tidal currents are also targeted by humans for energy extraction. Harvesting tidal energy involves large, in-stream hydrokinetic (HK) turbines installed in areas with fast tidal flows (Charlier and Finkl 2010). Unlike conventional or tidal barrage hydropower designs, HK turbines are free-standing, open structures installed in naturally flowing water currents. These devices have the potential to affect fishes using the same currents, but because of a scarcity of installed projects, effects on fishes remain unknown. Some high-priority unknowns include direct strike by turbine blades and injury because of pressure changes near the blades (Polagye et al. 2011). More indirectly, fish behavior in response to a device may result in a modified distribution of individuals in the water column, ultimately affecting the magnitude of more direct effects like blade strike. While tidal currents are used by many fish species, the distribution of fish within the flow are largely unstudied, partly because of the difficulty of working in these challenging environments (Gill 2005; Shields et al. 2008, 2009).
Cobscook Bay, ME is located at the mouth of the Bay of Fundy. It has a mean tidal range of 5.7 m (Brooks 2004) and current speeds exceeding 2 m · s-1 (4 knots) in the outer bay. A pilot tidal energy device, Ocean Renewable Power Company’s (ORPC’s) TidGen® Power System (Fig. 1), was installed in outer Cobscook Bay in August of 2012 (Fig. 2). The entire turbine structure is 31.2 m (102.3 ft) long and is positioned approximately 8.1 m (26.5 ft) above the sea floor by a solid steel frame. As with other tidal energy devices, the effects of the TidGen® system on fishes are unknown.
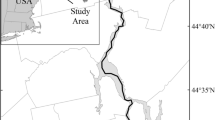
Left: Map of Cobscook Bay, ME. Right: Sampling sites in the outer bay, showing bottom depth (adapted from Kelley and Kelley 2004). Mean ebb and flood positions are indicated by the circles at each end of the white lines. CB1 is the Cobscook Bay 1 “project” site and CB2 is the Cobscook Bay 2 “control” site
Cobscook Bay is a productive ecosystem (Larsen and Campbell 2004), but the annual and seasonal presence and composition of pelagic fishes of the bay have not been studied. Most studies of the region have focused on benthic species vulnerable to trawling, and many of these studies are dated (Brawn 1960; Tyler 1971; MacDonald et al. 1984), therefore community composition could have changed and all of these surveys were conducted, not in Cobscook Bay, but in the adjacent Passamaquoddy Bay (Brawn 1960; Tyler 1971; MacDonald et al. 1984) or the Bay of Fundy proper (AECOM 2009). Key pelagic species expected to be in Cobscook Bay include Atlantic and blueback herring (C. harengus and Alosa aestivalis); alewife (Alosa pseudoharengus); rainbow smelt (Osmerus mordax); red, white, and silver hake (Urophycis chuss, Urophycus tenuis, and Merluccius bilinearis, respectively); threespine and blackspotted stickleback (Gasterosteus aculeatus and Gasterosteus wheatlandi), American eel (A. rostrata), and Atlantic mackerel (S. scombrus; Tyler 1971; MacDonald et al. 1984; Saunders et al. 2006; Athearn and Bartlett 2008; J. Vieser, unpublished information). Studies do not always agree on species seasonality, and the vertical distribution of species in the water column and the use of tidal currents are unknown. These missing data are critical to assessing the effects of tidal power devices on fishes in Cobscook Bay.
Few studies have examined the vertical distribution of fishes in tidal flows strong enough for tidal power generation. Many studies of tidally dynamic areas have focused on species composition and habitat use at low and high slack tides to demonstrate that fishes move with the tides, but details about their use of the water column during those movements are lacking (Morrison et al. 2002; Ribeiro et al. 2006; Jovanovic et al. 2007). The best details concerning vertical distribution of fishes in tidal currents come from studies of selective tidal stream transport. These studies show that certain species move into the water column during tides that flow in the desired direction of movement and out of the water column during opposing tides to avoid being swept in an undesirable direction (Greer Walker et al. 1978; de Veen 1978; McCleave and Kleckner 1982). Other studies of fish vertical distribution have been carried out at ocean sites with little current (Brawn 1960; Neilson et al. 2003; Jensen et al. 2011), estuaries with moderate current (Bennett et al. 2002), lakes (Clark and Levy 1988; Levy 1990), and rivers (Kubecka and Duncan 1998). Many of these studies document significant diel or tidal differences in the vertical distribution of fish, with additional variation related to time of year, location, and species.
We conducted stationary, down-looking hydroacoustic surveys to characterize patterns in fish presence and vertical distribution, with fine vertical and temporal resolution, at the Cobscook Bay project area. A preliminary study conducted in Minas Passage concluded that hydroacoustic technology could be used for this purpose (Melvin and Cochrane 2012). Using hydroacoustics allows noninvasive, continuous sampling of nearly the entire water column despite this difficult, high-velocity environment. Our study design included a project site (where the device has been located since August 2012) and a control site, with data collected prior to and after device installation. The use of a control site and before-and-after data collection are both critical to detecting the effects of the device on fish density and vertical distribution over time; the use of a control site will allow the assessment of any changes due to turbine installation to be discriminated from natural variation.
We hypothesize that the overall density and vertical distribution of fishes at the project site will change when the device is installed. Therefore, effects of the turbine will be assessed as statistically detectable differences in fish density or vertical distribution, such as (1) overall reduced fish density at the project site compared to density recorded previously at the project site (e.g., in the same month of the previous year) and compared to data collected concurrently at the control site, or (2) a detectable change in vertical distribution of fish in the water column after turbine installation compared to previously collected data at the project site or compared to data collected at the control site. This manuscript details natural variation in these parameters prior to turbine deployment.
Two years of pre-deployment data from the project site and the control site were collected and analyzed. Our goals were to describe the patterns in fish density and vertical distribution at the test and control sites on varying temporal scales (seasonal, diel, and tidal cycles) and to verify that the control site is similar to the project site and therefore useful for detecting effects of the device.
Methods
Data were collected in outer Cobscook Bay at the proposed pilot project site and a control site (Fig. 2). The project site, CB1, was located mid-channel at 44°54.60′ N, 67°2.74′ W; the control site, CB2, was approximately 1.6 km seaward, also mid-channel, at 44°54.04′ N, 67°1.71′ W. For data collection, a 12.2 m boat was moored at these two sites. The boat swung around its mooring at each slack tide as the direction of tidal flow changed; this movement was minimal for most months (205 m mean difference at CB1, 147 m mean difference at CB2). Under normal conditions, water depth at CB1 averaged 24.5 m at low tide to 32.3 m at high tide, and at CB2 depth averaged 33.8 to 41.3 m. However, positioning of the mooring at CB1 in May 2010 caused the boat to swing into much deeper water during most of the ebbing tide; ebb tide data for that month were subsequently omitted from analyses. At CB1, average current speed (water column mean) was 1.01 m · s-1 (2.0 knots), with a maximum of 2.06 m · s-1 (4.0 knots). At CB2, average current speed was 0.87 m · s-1 (1.7 knots), with a maximum of 1.78 m · s-1 (3.5 knots).
A single-beam Simrad ES60 echosounder was used with a circular transducer (38/200 Combi W) mounted 1 m below the surface over the port side of the vessel, facing downward. The transducer insonified a volume of water approximately conical in shape, extending from the transducer to the sea floor. The echosounder operated at 200 kHz and 38 kHz simultaneously, at a rate of 2 pings s-1, with a half-power beam angle of 31° for both frequencies. A wide beam angle was used to maximize the volume sampled. The pulse duration was 0.512 ms for all surveys except the first two (May and June 2010 1.024- and 0.256-ms pulse lengths, respectively), when trial and error were used to reduce electrical noise in the dataset.
The echosounder was calibrated using copper calibration spheres (13.7-mm diameter with -45.00 dB nominal target strength (TS) for 200 kHz; 60-mm diameter with -33.60 dB nominal TS for 38 kHz) as recommended by Foote et al. (1987). On-axis calibrations were conducted in situ at slack tide during each sampling session to ensure consistent system functionality. To obtain accurate calibration offsets for use during data processing, on-axis calibrations were carried out each winter (January 2011 and February 2012) on a frozen lake, where the water was sufficiently still to allow precise positioning of the sphere within the acoustic beam. These calibrations were carried out for each pulse duration used during the surveys.
Current speed measurements were obtained using a Marsh–McBirney flow meter from May 2010 to May 2011, and an acoustic doppler current profiler (ADCP) from June 2011 to November 2011. These devices were also mounted over the side of the vessel, 1 m below the surface (flow meter to starboard; ADCP to port, aft of the Simrad transducer). The flow meter recorded surface current speed only, while the ADCP recorded current speeds throughout the water column with 1-m vertical resolution.
Data Collection
Twenty-four-hour stationary surveys were carried out at the two sites at least once each season beginning May 2010 (Table 1). Surveys were scheduled in order to sample nearly two complete tidal cycles: one at night and one during the day. Depending on the time of year, this was not always possible; in May and June, nights encompassed only one tidal stage, and in March, this was true for days. Surface current speed was manually recorded every half hour when using the flow meter; current speed throughout the water column was automatically measured and recorded every half hour for 1 min when using the ADCP. Surface salinity was measured using a handheld refractometer in all months surveyed in 2011 and was 32.00 ± 0.45 (average ± standard error). It was assumed that salinity variation with depth and over the course of the year would be negligible in this very well-mixed area (Brooks 2004; Larsen and Campbell 2004; G. Staines, unpublished information).
Data Analyses
Data collected with the 200-kHz frequency were used in this study, as smaller objects may be detected with 200 than with 38 kHz, and we were interested in all sizes of fish. Hydroacoustic data were processed using Echoview® software (5.1, Myriax, Hobart, Australia), and statistical analyses were carried out in R (2.15.2, R Core Team, Vienna, Austria).
Hydroacoustic data processing began with calibrating the raw data according to results from the winter calibration sessions. The Simrad ES systematic triangle wave error was investigated in calibration files and determined to be negligible. The bottom line was automatically detected with the Echoview best bottom-line-pick algorithm, then manually corrected for errors and offset by 0.5 m. Backscatter data were then visually scrutinized, and areas of noise (for instance, from electrical interference, a passing boat’s depth sounder, or interference from the ADCP) or high boat motion were manually excluded from analyses. June 2010 data collected at CB1 were excluded because of excessive electrical interference. Acoustic interference from entrained air was common in the upper 10 m of the water column. Data analyses were therefore limited to the lowest 15 m of the water column at both sites (though CB2 was deeper). The lower 15 m of water spanned the future position of the TidGen® turbine, which occupies the space from 6.7 to 9.5 m above the sea floor.
Acoustic signals from unwanted targets (such as plankton, krill, and fish larvae) were then excluded by eliminating acoustic returns with TS less than -60 dB. Most fish have TS between -60 and -20 dB, but this varies greatly with fish anatomy and orientation (Simmonds and MacLennan 2005). This variability, combined with the TS uncertainty inherent in single-beam systems, meant that some fish with actual TS greater than the -60 dB threshold may have been excluded, depending on their position within the beam (Simmonds and MacLennan 2005).
Data from slack tides were removed from analyses because flowing tides (when the turbine would be rotating) were the focus of this study. Focusing on flowing tides also greatly reduced the potential for the same fish to make multiple passes through the hydroacoustic beam and eliminated periods of time when the boat was swinging around its mooring (and therefore moving over the seafloor, instead of remaining stationary). The ORPC TidGen® stops rotating when current speeds fall below 0.5 m · s-1 and remains nonrotating until current speeds rise above 1.0 m · s-1. These nonrotating periods were considered slack tides, and mean water column current speed was used to define slack tide start and end points. Mean current was obtained for each half hour by averaging ADCP current data from surface to seafloor. When only surface current data were available (collected with the flow meter), a correction was applied to approximate the water column average. This correction was obtained for each site using data collected concurrently with the ADCP and the flow meter in August of 2011. The average slack tide spanned 2.9 h (±0.1 standard error) at CB1 (the project site) and 2.2 h (±0.1) at CB2.
Once slack tide data were removed, the remaining hydroacoustic data were divided into a grid with columns 30 min wide and rows 1 m high. Time segments of 30 min were large enough to minimize autocorrelation but still obtain an accurate measure of the variation in density that occurred over the course of each survey. The 1 m water column layers were measured upward from the sea floor, rather than downward from the surface, as the turbine is located at a fixed distance above the bottom. Echoview was used to calculate the mean volume backscatter strength (S v) of each 30 min column of the grid and the area backscattering coefficient (s a) for each 30 min × 1 m grid cell. Volume backscatter is a measure of the sound scattered by a unit volume of water and is an index of fish density (Foote 1983). It is represented in the linear scale as the volume backscattering coefficient (s v, with units of m2 · m-3) or in the logarithmic scale as volume backscattering strength (S v in dB re 1 m-1; Simmonds and MacLennan 2005). The area backscattering coefficient, s a, is the acoustic energy returned from a given layer within the water column and has units of m2 · m-2. s a is the integration of S v with respect to depth and so is also an index of fish density. The normalized vertical distribution of fish was constructed for each 30-min grid column by calculating the proportion of the column’s total s a contributed by each 1-m layer of water.
Permutation analysis of variance tests (R package lmPerm; Wheeler 2010) were done to assess variation in water column S v, as the data did not meet the assumption of normality (significance level 0.05). When significant factor effects were found, nonparametric Tukey-type multiple comparisons (R package nparcomp; Konietschke 2012) were used to determine significant differences among groups (significance level 0.05). After testing for the effect of year on S v, 2010 and 2011 were treated separately, as we believe the year effect was largely due to an improved electrical system used in 2011. We then tested for the effect of month and site on water column S v in each year and for effects of diel condition (day or night) and tidal stage (ebb or flood) on water column S v in 2011 only.
To assess changes in fish vertical distribution, the mean vertical distribution of s a was obtained for each survey in 2010 and 2011, as well as for each diel and tidal stage of each survey in 2011 (day, night; day ebb, day flood, night ebb, and night flood). Survey distributions from the project site were compared to those from the control site to test for site differences. Diel and tidal stage comparisons were done for each 2011 survey. Statistical testing consisted of the linear regression of one mean vertical distribution onto the other. If distributions were similar, the fitted line was significant (p < 0.05) and had a positive slope. If distributions were not similar, the fitted line had either a nonsignificant fit (p > 0.05) or a negative slope (indicating opposite distributions).
Results
Fish Density Index: Water Column Sv
Water column S v (an index of fish density) was significantly greater in 2010 than 2011 at both sites (p < 0.05). In 2010, S v was significantly affected by month and month’s interaction with site (p < 0.05), indicating that fish density varied significantly over the course of the year, but not in the same way at both sites. Multiple comparisons for 2010 data indicated that sites were significantly different from each other only in September. At CB1 (project), water column fish density was highest in November, followed by May, then September, August, and October (Fig. 3). At CB2 (control), May, June, and September had the greatest density, followed by November, October, and August (Fig. 3). The effects of diel and tidal stage on water column fish density were not tested for 2010.
Mean water column (0–15 m above sea floor) volume backscatter, S v, of each survey. Horizontal lines are median S v, filled rectangles indicate the interquartile range, and whiskers extend to the 5th and 95th percentile. Month of survey is shown along horizontal axis. Significant differences between the sites (project CB1, control CB2) are indicated by *. May 2010 (†) data were limited to the flood tides, in June 2010 (X) data were collected, but not usable, and in November 2010 (‡), only the first half of the CB1 survey was conducted due to inclement weather
In 2011, S v varied significantly by month and site (p < 0.05), but the interaction had no effect. S v was slightly higher at CB2 when all surveys were grouped together; however, multiple comparisons revealed that within each survey, densities at CB1 and CB2 were never significantly different. Fish density was highest in May, followed by November (Fig. 3). Diel condition, tidal stage, and their interaction with month significantly affected S v, indicating diel condition and tidal stage had different effects on fish density in different months (Fig. 4). Multiple comparisons carried out within each survey showed fish density during the ebb tide was equal to or higher than during flood tide (Fig. 4). Density also appeared more variable during the day than at night (except March 2011 at CB2; Fig. 4). Water column S v at CB1 and CB2 showed similar patterns with respect to tidal stage during most surveys (Fig. 4).
Mean water column (0–15 m above sea floor) volume backscatter, S v, of each tidal stage sampled (day ebb, day flood, night ebb, and night flood) in 2011. Horizontal lines are median S v; filled rectangles indicate the interquartile range, and whiskers extend to the 5th and 95th percentile. Month of survey is shown along the horizontal axis. Significant differences, determined for each survey using multiple comparisons, are indicated with letters a and b, with group a having the highest values and group b having the lowest. May and June lack night flood data because nights were too short to encompass two complete tidal stages
Vertical Distribution: Proportional sa
The proportion of backscatter (fish density) contributed by each layer of water generally increased toward the sea floor (0–5 m above the bottom) at both sites in both years (Fig. 5). This was true in all months surveyed except May 2011 CB1, when fish density was highest in the upper layers analyzed (11–15 m above the bottom). This was the only month in which vertical distributions at the test and control sites were significantly different (though note that only the lower 15 m of CB2 could be used in the statistical comparison to CB1; density increased near the upper layers at CB2 as well).
Mean vertical distribution of fish for each survey in 2010 and 2011 at CB1 and CB2. Vertical axis is distance above bottom (m). Each horizontal bar represents the proportion of the water column’s total area backscatter (s a ) within each 1 m layer. Error bars show one standard error. Data are for the lower 15 m at CB1 and for the lower 26 m at CB2, which is a deeper site. Horizontal dashed lines indicate the depth spanned by the tidal energy device’s turbine (not present during this study). A blank box indicates no data collected; “X” indicates data were collected, but not usable (June 2010, CB1). May 2010 (†) data were limited to the flood tides, and in November 2010 (‡), only the first half of the CB1 survey was conducted due to inclement weather. The mean water column s a for each survey is shown in the upper right of each box (units of m2 · m-2)
Vertical distributions of fish in 2011 differed significantly between day and night in May and June at CB1, but not at CB2 (Fig. 6). In these instances, the fish density index was higher in the upper layers analyzed during the day, but shifted toward the sea floor during the night (Fig. 6). Though the difference between day and night was not statistically significant in the other surveys, fish appeared to be more evenly distributed throughout the water column at night than during the day, with generally lower variation and fewer peaks in density (Fig. 6).
Mean vertical distribution of fish for each tidal stage at CB1 and CB2 in 2011. Day distributions are shown in gray, night in black. Vertical axis is distance above bottom. Each horizontal bar represents the proportion of the water column’s total area backscatter (s a) within each 1 m layer during that tidal stage. Error bars show one standard error. Data are for the lower 15 m at CB1 and for the lower 26 m at CB2, which is a deeper site. Horizontal dashed lines indicate the depth spanned by the tidal energy device’s turbine (not present during this study). Blank boxes in May and June indicate no data collected during that time period because nights were too short. The mean water column s a for each time period is shown in the upper right of each box (units of m2 · m-2)
In 2011, the vertical distributions of fish were affected by tidal stage during the day in March and June at CB1 and June at CB2 (Fig. 6). In March at CB1, fish density was higher near the bottom during the daytime ebb, but more evenly distributed in the water column during the daytime flood. In June at CB1, fish density was highest between 10 and 15 m above the bottom during the ebb, but highest between 1 and 5 m above the bottom during the flood. At CB2 in June, fish were more evenly distributed in the water column during the daytime ebb (with variable peaks mid-column), but closer to the bottom during the flood.
Discussion
Understanding the interactions between the environment and its biological constituents in tidally dynamic coastal regions is essential for informing tidal power development. Research and monitoring in these areas is limited because of the strong tidal currents. Recent interest in tidal power extraction in Cobscook Bay provided the opportunity to develop an approach to investigate variation in fish abundance (density index) and vertical distribution, both expected to change with the installation of an obstacle (e.g., tidal turbine) occupying a specific layer within the water column. The Bay’s complicated bathymetry combines with a large tidal range to create high current speeds and flow patterns that vary greatly with location and tide (Brooks 2004; H. Xue, unpublished information). Multiple fish species pass through the strong currents of the outer bay to move between deeper ocean habitats and the extensive inshore habitats of the inner bays. Given the extreme variation in currents over time and space and the mixed seasonal and year-round fish community structure, hydroacoustic measures of fish density were expected to vary widely in relation to season and location. Our 2 years of hydroacoustic assessment demonstrate that while fish density is indeed variable, changes in fish density and vertical distribution on seasonal, diel, and tidal time scales are similar at the project and control sites. Therefore, site comparisons in the future can be used to statistically examine the effects of devices on overall fish density and vertical distribution at the project site.
Water column fish density varied by year and month in a similar manner at both sites. The decrease in density and its variability related to site and month from 2010 to 2011 may have been natural variation. However, it is also likely related to changes to the electrical system used in 2011 that resulted in cleaner data, especially at greater depths. This is supported by preliminary data collected in 2012 (unpublished) which appear very similar to 2011, with little difference between sites. Despite the change from 2010 to 2011, similar seasonal patterns were discernible at both sites in both years, with fish density higher in May and November than other months. The increase in May is likely linked to the springtime movements of anadromous fish (e.g., alewife; Saunders et al. 2006) and other species (e.g., threespine and blackspotted stickleback) into the bay, and the presence of large schools of larval and juvenile Atlantic herring (J. Vieser, unpublished information). The slight increase in November may reflect an inverse movement of summer residents out of the bay.
The vertical distribution of fish did not show a distinct seasonal pattern. Fish density almost always increased toward the sea floor, regardless of time of year. The main exception was May 2011 at CB1 (project), when dense schools were present in the middle and upper water column (mostly during the day), causing the density index to increase toward the surface. This effect was also seen during the daytime ebb tide in June 2011 at CB1, and during the day in May and June 2011 at CB2. These higher densities in the upper water column were likely due to the large numbers of larval and juvenile herring that were present in the spring, which have been found to move toward the surface during the day and toward the bottom at night (Jensen et al. 2011).
A relatively consistent difference between day and night was observed in the vertical distributions, though only two of the surveys tested showed a significant difference. At night, fewer peaks in density throughout the water column and lower variation in each layer reflected a general dispersal of dense clusters (e.g., schools of fish). This was also evidenced by the higher variation in water column density during the day than during the night (Fig. 4). The dispersion of fish at night has been observed by others (Gauthier and Rose 2002; Knudsen et al. 2009) and may be linked to visibility as schooling fish often depend on vision to remain in formation (Pitcher 2001). Regardless of the reason, this diel change in fish use of the water column may be important to consider when assessing the potential effects of tidal turbines on fishes. Viehman and Zydlewski (this issue) found that schooling fish may have a greater likelihood of avoiding a test tidal turbine than individual fish and that fish were more likely to enter the turbine at night than during the day. If fish in the area of a tidal device spread out at night, a fish’s chance of entering the turbine may increase.
The vertical distribution of fish lacked a consistent pattern related to tidal stage. This was not surprising, considering different species of fish may be using different tidal currents depending on the time of year or even time of day (Tyler 1971; Morrison et al. 2002). Diel and tidal fish behaviors are often species- and age-specific (Weinstein et al. 1980; Levy and Cadenhead 1995; Neilson and Perry 2001), and the fish community of Cobscook Bay is composed of many species and age classes which change over the course of the year. The species composition of sampled fish may even differ between ebb and flood tides due to the difference in flow pattern through the bay. The ability to isolate a specific group of fish within a mixed fish community is limited when using hydroacoustics. This is especially true for single-beam systems, which cannot provide accurate TS values without detailed knowledge of the beam pattern and additional data processing methods (such as deconvolution; Simmonds and MacLennan 2005). Without isolating groups of fish, movement patterns of one group may be obscured by the movements of others. In this study, one of the best examples of a diel pattern in vertical fish distribution occurred in May 2011 at CB1 and CB2 and in June 2011 at CB2, when fish were in the upper layers of the water column during the day but spread throughout the lower layers at night (Fig. 6). This difference may have been because of large numbers of larval and juvenile herring during these surveys compared to other fish species. The fish community was likely to be more evenly mixed during the other surveys.
Fish density almost always increased near the sea floor, regardless of any diel or tidal variation in the vertical distribution of fish. Therefore, factors besides diel condition and tidal stage may have been shaping the vertical distribution of fish. The increase in fish density in the lower layers may correspond to the species present, which include winter flounder (Pseudopleuronectes americanus), tomcod (Microgadus tomcod), red, white, and silver hake, sculpins, and other species generally associated with the bottom (J. Vieser, unpublished information). Pelagic fish might also seek shelter in the lower current speeds near the sea floor in the same way that species exhibiting selective tidal stream transport resist backward movement (McCleave and Kleckner 1982; Auster 1988). Auster (1988) described this behavior in cunner (Tautogolabrus adspersus) feeding on current-exposed surfaces: increasing current speed resulted in increasingly larger fish remaining above the seabed to forage, while smaller fish moved toward shelter on the bottom. If fish seek refuge near the sea floor during strong currents at the sites studied, analyzing behaviors during the slack tide (when pelagic species may leave the bottom layers) could aid in distinguishing diel behavioral patterns suppressed during the flowing tide. This could allow the identification of pelagic and benthic species based on behavior as well as acoustic properties.
The timing of these surveys within each month likely affected the data collected and the patterns observed. Sampling continuously for 1 day month-1 provided a wealth of information for that day on a fine time scale, but data collected over 24 h may not be representative of an entire month or season. In such a dynamic environment, there is likely to be variability from one day to the next which is difficult to quantify without sampling multiple sequential days. To achieve a more accurate understanding of fish vertical distribution and what causes it to change over time would likely require sampling multiple days throughout each month, although sampling the control site should help identify abnormal occurrences at the project site. One option for consideration in future studies is to deploy a surface- or bottom-mounted echosounder that automatically collects data on a preset, semicontinuous schedule. This would reduce the long-term cost and effort required to collect the data, while allowing the collection of high-resolution data over a long period of time. High-resolution data collected over long time periods is essential for quantifying natural variability and separating it from turbine effects.
Data collected to date were sufficient to characterize the relative abundance and vertical distribution of fish at the site that could be affected by the installation of a tidal turbine, despite any influence of survey timing and natural variation. The use of a control site, which has exhibited similar changes in fish density and vertical distribution as the project site (especially in 2011 and 2012; unpublished data), overcomes the potential for natural variation to mask effects of the turbine. The rotating blades of ORPC’s TidGen® span the area between 6.7 and 9.5 m above the sea floor, directly in the center of the range analyzed (0–15 m above the bottom). Within this range, fish density was generally greatest near the bottom (0–5 m). While this is below the height of the rotating turbine, these fish are still likely to interact with the solid support frame and foundation, and this could affect their vertical distribution post-deployment. Using the tests presented, we should be able to detect such changes in the future.
Continued data collection at the pilot project and control sites will improve understanding of the seasonal movements of fishes through the region, fishes’ use of the water column in fast tidal currents, and any changes to fish density or vertical distribution associated with the introduction of the pilot device. The use of a split-beam echosounder (as in Melvin and Cochrane 2012) concurrently with the single-beam system (which must continue to be used for comparison to past data) will allow estimation of fish abundance and length, as well as direction of movement. Fish size has been found to affect their reactions to an HK turbine (Viehman and Zydlewski this issue), so if the number and length of detected fish could be approximated, the potential rate of fish encounters with the turbine and supporting structures as well as their likely reactions could be estimated. Furthermore, analyzing the vertical distribution of various size groups may improve knowledge of the temporal and spatial use of the water column by different fish species relative to various environmental factors. This information is of particular interest to federal and state agencies making decisions concerning habitat protection (e.g., Essential Fish Habitat) and species regulations (e.g., the US Endangered Species Act) in coastal zones and can aid in management efforts as tidal power development continues.
References
AECOM. 2009. Appendix 9: Commercial fisheries studies—phase I and phase II. In: Fundy Ocean Research Centre for Energy (Minas Basin Pulp and Power Co. Ltd.) Environmental Assessment.
Aprahamian, M.W., G.O. Jones, and P.J. Gough. 1998. Movement of adult Atlantic salmon in the Usk estuary, Wales. Journal of Fish Biology 53: 221–225.
Arnold, G.P., M. Greer Walker, L.S. Emerson, and B.H. Holford. 1994. Movements of cod (Gadus morhua L.) in relation to the tidal streams in the southern North Sea. ICES Journal of Marine Science 51: 207–232.
Athearn, K. and C. Bartlett. 2008. Saltwater fishing in Cobscook Bay: Angler profile and economic impact. Maine Sea Grant Marine Research in Focus 6.
Auster, P.J. 1988. A review of the present state of understanding of marine fish communities. Journal of Northwest Atlantic Fishery Science 8: 67–75.
Bennett, W.A., W.J. Kimmerer, and J.R. Burau. 2002. Plasticity in vertical migration by native and exotic estuarine fishes in a dynamic low-salinity zone. Limnology and Oceanography 47(5): 1496–1507.
Brawn, V.M. 1960. Seasonal and diurnal vertical distribution of herring (Clupea harengus L.) in Passamaquody Bay, NB. Journal of the Fisheries Research Board of Canada 17: 699–711.
Brooks, D.A. 2004. Modeling tidal circulation and exchange in Cobscook Bay, Maine. Fisheries Bulletin 11: 23–50.
Castonguay, M., and D. Gilbert. 1995. Effects of tidal streams on migrating Atlantic mackerel, Scomber scombrus L. ICES Journal of Marine Science 52: 941–954.
Charlier, R.H., and C.W. Finkl. 2010. Ocean energy: Tide and tidal power. Berlin: Springer-Verlag.
Clark, C.W., and D.A. Levy. 1988. Diel vertical migrations by juvenile sockeye salmon and the antipredation window. The American Naturalist 131(2): 271–290.
Dadswell, M.J., and R.A. Rulifson. 1994. Macrotidal estuaries: A region of collision between migratory marine animals and tidal power development. Biological Journal of the Linnean Society 51: 93–113.
de Veen, J.F. 1978. On selective tidal transport in the migration of North Sea plaice (Pleuronectes platessa) and other flatfish species. Netherlands Journal of Sea Research 12: 115–147.
Foote, K.G. 1983. Linearity of fisheries acoustics, with addition theorems. Journal of the Acoustical Society of America 73(6): 1932–1940.
Foote, K.G., H.P. Knudsen, G. Vestnes, D.N. MacLennan, and E.J. Simmonds. 1987. Calibration of acoustic instruments for fish density estimation: A practical guide. ICES Cooperative Research Report 144.
Gauthier, S., and G.A. Rose. 2002. Acoustic observation of diel vertical migration and shoaling behavior in Atlantic redfishes. Journal of Fish Biology 61: 1135–1153.
Gill, A.B.. 2005. Offshore renewable energy: Ecological implications of generating electricity in the coastal zone. Journal of Applied Ecology 42: 605–615.
Greer Walker, M., F.R. Harden Jones, and G.P. Arnold. 1978. The movements of plaice (Pleuronectes platessa L.) tracked in the open sea. Journal du Conseil International pour l'Exploration de la Mer 38: 58–86.
Hartill, B.W., M.A. Morrison, M.D. Smith, J. Boubée, and D.M. Parsons. 2003. Diurnal and tidal movements of snapper (Pagrus auratus, Sparidae) in an estuarine environment. Marine and Freshwater Research 54: 931–940.
Jensen, O.P., S. Hansson, T. Didrikas, J.D. Stockwells, T.R. Hrabik, T. Axenrot, and J.F. Kitchell. 2011. Foraging, bioenergetic and predation constraints on diel vertical migration: Field observations and modeling of reverse migration by young-of-the-year herring Clupea harengus. Journal of Fish Biology 78: 449–465.
Jovanovic, B., C. Longmore, A. O’Leary, and S. Mariani. 2007. Fish community structure and distribution in a macro-tidal inshore habitat in the Irish Sea. Estuarine, Coastal and Shelf Science 75: 135–142.
Kelley, J.T., and A.R. Kelley. 2004. Controls on surficial materials distribution in a rock-framed, glaciated, tidally dominated estuary: Cobscook Bay, Maine. Northeast Naturalist 11: 51–74.
Knudsen, F.R., A. Hawkins, R. McAllen, and O. Sand. 2009. Diel interactions between sprat and mackerel in a marine lough and their effects upon acoustic measurements of fish abundance. Fisheries Research 100: 140–147.
Konietschke, F. 2012. nparcomp: Perform multiple comparisons and compute simultaneous confidence intervals for the nonparametric relative contrast effects. R package version 2.0. http://CRAN.R-project.org/package=nparcomp.
Krumme, U. 2004. Patterns in tidal migration of fish in a Brazilian mangrove channel as revealed by a split-beam echosounder. Fisheries Research 70: 1–15.
Krumme, U., and U. Saint-Paul. 2003. Observations of fish migration in a macrotidal mangrove channel in Northern Brazil using a 200-kHz split-beam sonar. Aquatic Living Resources 16: 175–184.
Kubecka, J., and A. Duncan. 1998. Diurnal changes of fish behavior in a lowland river monitored by a dual-beam echosounder. Fisheries Research 35: 55–63.
Lacoste, K.N., J. Munro, M. Castonguay, F.J. Saucier, and J.A. Gagné. 2001. The influence of tidal streams on the pre-spawning movements of Atlantic herring, Clupea harengus L., in the St Lawrence estuary. ICES Journal of Marine Science 58: 1286–1298.
Larsen, P.F., and D.E. Campbell. 2004. Ecosystem modeling in Cobscook Bay, Maine: A summary, perspective, and look forward. Northeast Naturalist 11: 425–438.
Levy, D.A. 1990. Reciprocal diel vertical migration behavior in planktivores and zooplankton in British Columbia lakes. Canadian Journal of Fisheries and Aquatic Sciences 47: 1755–1764.
Levy, D.A., and A.D. Cadenhead. 1995. Selective tidal stream transport of adult sockeye salmon (Oncorhynchus nerka) in the Fraser River Estuary. Canadian Journal of Fisheries and Aquatic Sciences 52: 1–12.
MacDonald, J.S., M.J. Dadswell, R.G. Appy, G.D. Melvin, and D.A. Methven. 1984. Fishes, fish assemblages, and their seasonal movements in the lower Bay of Fundy and Passamaquoddy Bay. Fishery Bulletin 82: 121–139.
McCleave, J.D., and R.C. Kleckner. 1982. Selective tidal stream transport in the estuarine migration of glass eels of the American eel (Anguilla rostrata). ICES Journal of Marine Science 40: 262–271.
Melvin, G.D., and N.A. Cochrane. 2012. A preliminary investigation of fish distributions near an in-stream tidal turbine in Minas Passage, Bay of Fundy. Canadian Technical Report of Fisheries and Aquatic Sciences 3006. New Brunswick: St. Andrews.
Moore, A., M. Ives, M. Scott, and S. Bamber. 1998. The migratory behavior of wild sea trout (Salmo trutta L.) smolts in the estuary of the River Conwy, North Wales. Aquaculture 198: 57–68.
Morrison, M.A., M.P. Francis, B.W. Hartill, and D.M. Parkinson. 2002. Diurnal and tidal variation in the abundance of the fish fauna of a temperate tidal mudflat. Estuarine, Coastal and Shelf Science 54: 793–807.
Neilson, J.D., and R.I. Perry. 2001. Fish migration, vertical. In Encyclopedia of ocean sciences, vol 2, 2nd ed, ed. J.H. Steel, K.K. Turekian, and S.A. Thorpe, 411–416. Oxford: Academic Press.
Neilson, J.D., D. Clark, G.D. Melvin, P. Perley, and C. Stevens. 2003. The diel vertical distribution and characteristics of pre-spawning aggregations of Pollock (Pollachius virens) as inferred from hydroacoustic observations: The implications for survey design. ICES Journal of Marine Science 60: 860–871.
Pitcher, T.J. 2001. Fish schooling. In Encyclopedia of Ocean Sciences, vol 2, 2nd ed, ed. J.H. Steel, K.K. Turekian, and S.A. Thorpe, 432–444. Oxford: Academic Press.
Polagye, B., B. Van Cleve, A. Copping, and K. Kirkendall. 2011. Environmental effects of tidal energy development. U.S. Dept. Commerce, NOAA Tech. Memo. F/SPO-116.
Ribeiro, J., L. Bentes, R. Coelho, J.M.S. Gonçalves, P.G. Lino, P. Monteiro, and K. Erzini. 2006. Seasonal, tidal and diurnal changes in fish assemblages in the Ria Formosa lagoon (Portugal). Estuarine, Coastal and Shelf Science 67: 461–474.
Sakabe, R., and J.M. Lyle. 2010. The influence of tidal cycles and freshwater inflow on the distribution and movement of an estuarine resident fish Acanthopagrus butcheri. Journal of Fish Biology 77: 643–660.
Saunders, R., M. Hachey, and C.W. Fay. 2006. Maine’s diadromous fish community. Fisheries 31: 537–547.
Shields, M. A., A. T. Ford, and D. K. Woolf. 2008. Ecological considerations for tidal energy development in Scotland. 10th World Renewable Energy Conference, 19–25 July, Glasgow, Scotland.
Shields, M.A., L.J. Dillon, D.K. Woolf, and A.T. Ford. 2009. Strategic priorities for assessing ecological impacts of marine renewable energy devices in the Pentland Firth (Scotland, UK). Marine Policy 33: 635–642.
Simmonds, J., and D. MacLennan. 2005. Fisheries acoustics: Theory and practice, 2nd ed. Oxford: Blackwell Science.
Stasko, A.B.. 1975. Progress of migrating Atlantic salmon (Salmo salar) along an estuary, observed by ultrasonic tracking. Journal of Fish Biology 7: 329–338.
Tyler, A.V. 1971. Periodic and resident components in communities of Atlantic fishes. Journal of the Fisheries Research Board of Canada 28: 935–946.
Weinstein, M.P., S.L. Weiss, R.G. Hodson, and L.R. Gerry. 1980. Retention of three postlarval fishes in an intensively flushed tidal estuary, Cape Fear River, North Carolina. Fisheries Bulletin 78: 419–436.
Wheeler, B. 2010. lmPerm: Permutation tests for linear models. R package version 1.1–2. http://CRAN.R-project.org/package=lmPerm.
Acknowledgments
We would like to thank the employees of Ocean Renewable Power Company for their generous contribution of time and resources, Captain George Harris, Jr. and crew for their invaluable assistance in the field, and Donald Degan, Anna-Maria Mueller, and Michael Jech for their acoustics advice. We also thank everyone who volunteered field time, Jeffrey Vieser and Huijie Xue for sharing unpublished data, and the members of the Maine Tidal Power Initiative and Michael Peterson for their assistance. This work was supported by an award from the United States Department of Energy, project #DE-EE0003647. The views expressed herein are those of the authors and do not necessarily reflect the views of Ocean Renewable Power Company or any of its subagencies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Wayne S. Gardner
Rights and permissions
About this article
Cite this article
Viehman, H.A., Zydlewski, G.B., McCleave, J.D. et al. Using Hydroacoustics to Understand Fish Presence and Vertical Distribution in a Tidally Dynamic Region Targeted for Energy Extraction. Estuaries and Coasts 38 (Suppl 1), 215–226 (2015). https://doi.org/10.1007/s12237-014-9776-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-014-9776-7