Abstract
Although many consequences of climate change on marine and terrestrial ecosystems are well documented, the characterisation of estuarine ecosystems specific responses and the drivers of the changes are less understood. In this study, we considered the biggest Southwestern European estuary, the Gironde, as a model of a macrotidal estuary to assess the effects of both large- (i.e., North Atlantic basin-scale) and regional-scale climate changes. Using a unique set of data on climatic, physical, chemical and biological parameters for the period 1978–2009, we examined relations between changes in both the physical and chemical environments and pelagic communities (plankton and fish) via an end-to-end approach. Our results show that the estuary experienced two abrupt shifts (∼1987 and ∼2000) over the last three decades, which altered the whole system. The timing of these abrupt shifts are in accordance with abrupt shifts reported in both marine (e.g., in the North Sea, the Mediterranean Sea and along the Atlantic) and terrestrial (e.g., in European lakes) realms. Although this work does not allow a full understanding of the dynamical processes through which climate effects propagate along the different compartments of the ecosystem, it provides evidence that the dynamics of the largest estuary of Southwest Europe is strongly modulated by climate change at both regional and global scales.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It has been established that climate change has a major influence on marine ecosystems (Hatun et al. 2009; Cloern et al. 2010) affecting all biological compartments from phytoplankton to top predators, influencing biodiversity and the structure and functioning of ecosystems (Beaugrand et al. 2008; Luczak et al. 2011). Both phenological and biogeographical changes have been documented (Edwards and Richardson 2004; Perry et al. 2005). Climatic variability and global climate change also influence littoral ecosystems (Halpern et al. 2008; Goberville et al. 2010, 2011). For instance, Attrill and Power (2002) used the North Atlantic Oscillation (NAO) index, to show that climatic variability explains a large part of the changes observed in the diversity, abundance and growth of juvenile marine fishes in estuaries. Nicolas et al. (2011) provided evidence for a climate-induced biogeographical shift in estuarine fish species along the European West Atlantic coasts. Marine ecosystems are known to respond to temperature changes in a nonlinear way (Carpenter and Brock 2006). Abrupt ecosystem shifts were therefore identified. Those shifts mainly resulted from species’ critical thermal limits (Carpenter and Brock 2006; Beaugrand et al. 2008).
Temperature is among the most conspicuous factors governing changes in marine systems (Bopp et al. 2002; Scavia et al. 2002; Beaugrand et al. 2009; Goberville et al. 2010). However, it has become more evident that this parameter acts in synergy with others to alter the states of marine ecosystems (Goberville et al. 2011) including fishing and pollution from maritime traffic. For example, overfishing has a direct impact on the abundance of commercial species (Pauly 2003) and many works have provided evidence that the effect of climate change on fish increases when the stocks are weakened by overfishing (Beaugrand et al. 2009). Maritime traffic, via ballast waters, increases the number of invasive species such as copepods (David et al. 2007a, b) and shrimps (Beguer et al. 2008). Raitsos et al. (2010) showed that the appearance of tropical species in the eastern part of the Mediterranean Sea was highly positively correlated with sea surface warming. On a smaller scale, other anthropogenic local pressures may act in synergy with climatic forcing. For instance, damming and water pumping especially in summer, may have strong effects on river discharge (Rochard et al. 1990) and prevent the migration of some fish species (Imbert et al. 2008). Human infrastructures, as for example nuclear power plants that release warm waters, may locally modify both the abiotic and biotic components of the ecosystems (Bamber and Seaby 2004; David et al. 2005).
In this paper, we investigate the influence of both climatic variability and global climate change on long-term (multidecadal) changes of the Gironde estuary. First, we explore the correlations between both sea surface temperature (SST) and sea level pressure (SLP) in the North Atlantic and large-scale meteo-oceanic indices. Second, we examine the influence of both large-scale and regional climate change on the physical and chemical properties of the Gironde estuary and its zooplankton and fish diversity. Finally, we discuss the role of climate variability in the long-term changes of estuarine ecosystems, and highlight potential local direct anthropogenic influences that might exacerbate or mitigate their responses.
Methods
Study Area
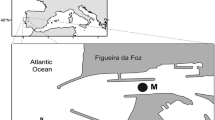
The Gironde estuary (latitude 45°20′ N, longitude 0°45′ W) is among the largest estuaries of the southern and western part of Europe, representing an area of about 625 km2 at high tide (Fig. 1; Lobry et al. 2003). This 70-km long estuary formed by the rivers Dordogne and Garonne drains 81,000 km2 (Allen et al. 1972) with a mean flow ranging from 250 (August–September) to 1,500 m3 (January–February). The Gironde is also one of the most turbid European estuaries with a mean suspended matter concentration higher than 500 mg l−1 (Sautour and Castel 1995; Abril et al. 1999).
The Gironde estuary is characterised by a strong spatio-temporal variability already documented by David et al. (2005). The authors described some significant long-term trends in the physical chemistry of the Gironde and related them to changes in environmental parameters (i.e., temperature, salinity, suspended matter concentration and active chlorophyll in the water column). Regarding Gironde zooplankton diversity, it is composed of only five dominant species including three calanoid copepod species (Eurytemora affinis, Acartia bifilosa and Acartia tonsa) and two mysids (Neomysis integer and Mesopodopsis slabberi; David et al. 2005). Biological shifts have also been documented. For example A. tonsa invaded the Gironde in 1983. The calanoid reached rapidly comparable densities to those of the dominant native species E. affinis (David et al. 2007a, b). Other studies on fish in the Gironde (Pronier and Rochard 1998; Nicolas et al. 2011; Pasquaud et al. 2012) also pointed out long-term trends possibly modulated by climate-driven environmental changes (e.g., the smelt Osmerus eperlanus, Pronier and Rochard 1998).
Large-Scale Climatic Parameters
The SST and SLP data were provided by the NOAA-CIRES Climate Diagnostics Center (Woodruff et al. 1987) and are part of the comprehensive ocean–atmosphere data set (COADS, 1° enhanced data). Monthly data, recorded in a 1° longitude × 1° latitude grid, are available for the period 1960–2009. An annual mean was calculated for each geographical cell of the spatial domain between 40° and 70° N of latitude and 80° W and 20° E of longitude (spatial resolution of 1° × 1°) for the period 1960–2009. These data were gathered in Matrix 1a.
Large-Scale Meteo-oceanic and Hydro-climatic Indices
We used three meteo-oceanic indices and one hydro-climatic index (in Matrix 1b) to examine potential relationships between long-term changes in annual SST and SLP and large-scale sources of hydro-meteorological or hydro-climatic variability.
The NAO is the most important mode of variability in the Northern Hemisphere atmospheric circulation. The atmospheric oscillation modulates climate variability (with an ∼8-year period) from the eastern seaboard of the USA to Siberia and from the Arctic to the subtropical Atlantic (Hurrell et al. 2001). It is a basin-scale alternation of atmospheric mass over the North Atlantic between the high pressures centred on the subtropical Atlantic and the low pressures around Iceland. This atmospheric oscillation, observed during the whole year, is particularly strong in winter, explaining an important part of the SLP variability from December to February (Hurrell 1995; Marshall et al. 2001). The changes in the pressure gradient from one index phase to another imply strong modifications in the winds speed and direction over the North Atlantic and also in the heat and moisture transport between the ocean and the surrounding continents.
The Atlantic Multidecadal Oscillation (AMO) is a large-scale mode of observed multidecadal climate variability (with a ∼70-year period) with alternated warm and cool phases over large parts of the Northern Hemisphere (Knight et al. 2005). Many examples of regional multidecadal climate variability have been related to the AMO, such as northeastern Brazilian rainfalls, or the North American and European summer climate. Its natural variability has a range of 0.4 °C (Enfield et al. 2001). This phenomenon has a strong impact on SST. The AMO index we used was built by computing a moving average on Atlantic (detrended) SST anomalies north of the equator. The National Centre for Atmospheric Research provided the NAO and AMO index data from 1860 to 2009.
The East Atlantic Pattern (EAP) is a low-frequency variability mode over the North Atlantic detected in all months but May–August. Similar to the NAO, this pattern consists of a North–South dipole of anomaly centres near 55° N, 20–35° W, 25–35° N and 0–10° W that spans the entire North Atlantic Ocean from East to West (Barnston and Livezey 1987; Decastro et al. 2006).
The Northern Hemisphere Temperature (NHT) anomalies over the period 1950–2009 were provided by the NOAA's Climate Prediction Centre and downloaded from the website: ftp://ftp.cpc.ncep.noaa.gov/.
Local Meteorological and Hydrological Parameters
Monthly data of local air temperature (in degrees Celsius) and precipitation (in millimetres per month), measured at Pauillac meteorological station (kilometre point 50 downstream from Bordeaux city), were provided by the Méteo France Centre of Mérignac. First, the data were deseasonalised. The deseasonalisation was performed by subtracting from the original time-series the corresponding seasonal index values. Then, an annual mean was calculated for the period 1978–2009 (32 years). The two parameters were gathered in Matrix 2 (32 years × 2 parameters).
Environmental and Zooplankton Data
Environmental data were provided by both the “Blayais” nuclear power plant and the French Coastal Monitoring Network SOMLIT (Service d’Observation en Milieu LITtoral, http://somlit.epoc.u-bordeaux1.fr) monitoring programs. SOMLIT gathers data from eight marine stations that monitor some key environmental parameters (see Goberville et al. 2010; Savoye et al. 2012): sea temperature, salinity, dissolved dioxygen, nitrate and nitrite, suspended matter, chlorophyll a concentrations, current speed, river discharge and some biological parameters including five zooplankton species (Table 1). The parameters were monitored from March 1978 to November 2009. Samples were collected nine times a year at a fixed station (E), located 52 km from Bordeaux city. Sampling was carried out 1 m below the surface and 1 m above the bottom at 3-h intervals during a tidal cycle (high and low tide, flood and ebb tide). Only data from high and low tide surface samples were averaged and used because some parameters (e.g., the nutrients and chlorophyll a concentrations) were only measured at these times. Thus, we considered an average tidal moment reflecting a complete tidal cycle, integrating both marine and freshwater influences. An annual average was computed for each physicochemical variable and species for the period 1978–2009 after data were deseasonalised using the method described above. Matrix 3 (32 years × 8 variables) contains a total of eight physical and chemical parameters and Matrix 4 (32 years × 5 species) a total of five species.
Fish Data
From 1979 to 1988, fish sampling was carried out twice a month. Samples were collected 1 m below the surface and 1 m above the bottom. Twelve species were monitored (Table 1). Only one transect, composed of five sites was monitored (Transect 1; Fig. 1) during this period. After 1988, another transect was added (Transect 2; Fig. 1), each transect being composed of three sampling sites. For each sampling, the volume measurement of water filtered was determined with flow meters (General Oceanics 2030 R) to determine fish species densities per cubic meter. Therefore, this study used pooled bottom and surface data (in order to catch the whole species diversity) that were then averaged per transect and per month. We used data from Transect 1 till 1988 and means of data from Transects 1 and 2 data after this year. Matrix 5 (31 years × 12 species) consisted of an annual mean for each of the 12 species for the period 1979–2009. We only selected fish species with an occurrence frequency over 20 % (results not shown). An annual average was calculated for each species for the period 1979–2009 after data were deseasonalised with the same method described previously.
Data Analyses
Analysis 1: Principal Components Analyses on Geolocalised Data (Annual SST and SLP)
Two standardised principal components analyses (PCAs), described in details in Beaugrand et al. (2002), were performed on SST and SLP data matrices (Matrix 1a) at the scale of the North Atlantic (40° N, 65° N, 80° W and 20° E) to interpret both spatial and temporal changes in SST and in SLP. Eigenvectors were normalised to represent the spatial correlation with the corresponding principal components. Normalised eigenvectors were mapped to identify areas most correlated with the first two principal components that summarise long-term changes in annual SST or SLP.
Prior to the analysis, annual means were calculated after transforming monthly data as anomalies by subtracting the long-term average of the corresponding month and geographical cell. The first principal components are called hereafter: PC1 SST, PC2 SST, PC3 SST, PC1 SLP, PC2 SLP and PC3 SLP.
Analysis 2: PCAs on Local Estuarine Data
Three standardised PCAs were performed on each data matrix: (1) environmental data matrix (Matrix 3); (2) zooplankton Matrix (Matrix 4) and (3) fish Matrix (Matrix 5). The PCAs were performed on a correlation matrix to (1) identify the main long-term changes that took place in both abiotic and biotic data (PCs time series analysis), (2) reveal the most important variables that are representative of each long-term change (i.e., each principal component) and (3) examine the correlations between components and both regional and global meteo-oceanic and hydro-climatic variables.
To facilitate reading, our results are mostly presented as bar plots of normalised principal components (component scores were normalised by subtracting the average from each value and then dividing by the standard deviation) of each analysis against time, the principal component data being first smoothed by applying a simple moving average of order 1. The first principal components are called: PC1 Env., PC2 Env. (PCA on Matrix 3), PC1 Zooplankton, PC2 Zooplankton (PCA on Matrix 4), PC1 Fish and PC2 Fish (PCA on Matrix 5).
Matrix 2 only contained two local meteorological variables and were therefore only examined graphically.
Analysis 3: Examination of the Relationships Between Principal Components and Other Related Variables
We combined results from PCAs into a single matrix, adding important variables for interpreting changes. The matrix 32 years × 20 variables had the following variables: PC1 SST, PC2 SST, PC3 SST, PC1 SLP, PC2 SLP, PC3 SLP, PC1 Env., PC2 Env., PC1 Zooplankton, PC2 Zooplankton, PC1 Fish, PC2 Fish, PC3 Fish, local air temperature, local precipitation, NAO, AMO, NHT and EAP. We calculated the correlation matrix and then converted the correlation matrix C into a distance matrix D by applying the following equation:
The hierarchical flexible agglomerative clustering method (Lance and Williams 1967) was then applied onto the distance matrix 20 × 20 variables to identify relationships between variables. Lance and Williams (1967) proposed this general model that encompasses most of the agglomerative clustering methods. By fixing the values of the four parameters α j, α m, β and γ (see Lance and Williams 1967; Legendre and Legendre 1998), one can go from the single to the complete linkage. Here, α j was fixed to 0.625, α m to 0.625, β to–0.25 and γ to 0 so that the method was close to the unweighted centroid clustering (also called unweighted pair-group centroid method, Legendre and Legendre 1998). We then examined graphically all variables that the cluster analysis above identified as relevant and examined their long-term trends. The variables were divided in three categories according to their type of long-term changes, their nature and scale.
Analysis 4: Identification of Major Discontinuities during the Period 1978–2009
Our analysis was restricted to the only variables that were identified as important in the previous analysis and we calculated the distance matrix 32 × 32 years using the Chord distance (Legendre and Legendre 1998). Then, the use of hierarchical flexible agglomerative clustering method detailed above allowed us to identify homogeneous time periods and the main discontinuities.
All the analyses were performed with MATLAB.
Results
Analysis 1: Principal Components Analyses on Geolocalised Data (Annual SST and SLP)
The first standardised PCA revealed an increase in annual SST anomalies with an acceleration phase circa 1993 (Fig. 2). The increase in the first principal component (PC1; 28.3 % of the total variance) reflected an increase in annual SST anomalies (i.e., positive correlation with the first PC or positive values of the first normalised eigenvector) over the East Atlantic basin and regions located around the UK (Fig. 2). The first PC was positively correlated to NHT anomalies (r = 0.97; p ACF < 0.05; Table 2) and the AMO index (r = 0.95, p ACF < 0.05). The second PC (9.2 % of the total variance) reflected variability in annual SST anomalies positively related to the state of the NAO (r = 0.64, p ACF < 0.05). Values of the second PC were inversely correlated to annual SST anomalies over the Subarctic Gyre and positively correlated over areas located to the north of the Subtropical Gyre in the western part of the Atlantic basin. Interestingly, the first PC of annual SST anomalies was positively related to the state of the EAP (r = 0.72, p ACF < 0.05). A gradual change was observed circa 1997, which was in part explained by the EAP index.
The second standardised PCA performed on annual SLP anomalies revealed that the year-to-year variability of the first PC (18.6 % of the total variance, Fig. 3) was highly correlated positively to the state of the NAO index (r = 0.77, p ACF < 0.05). Although this result was expected, the mapping of the first normalised eigenvector that identified the main centre of actions (low-pressure cell centred over Iceland and high-pressure cell centred over the Azores), it indicated that interannual fluctuations in annual SLP can be modulated by the state of the NAO in areas close to the Gironde estuary.
Analysis 2: PCAs on Local Estuarine Data (Matrices 3, 4 and 5)
A first standardised PCA was performed on the matrix 32 years × 8 environmental parameters (Matrix 3). The first PC explained 43.8 % of the total variance. Examination of the first normalised eigenvector (not shown) indicated that variables such as local temperature and salinity were negatively related to the first PC and that variables such as dissolved dioxygen concentrations, current speed and river discharge were positively related to the first PC. The second PC explained 23.3 % of the total variance. Examination of the second eigenvector indicated that the second PC was mainly related positively to chlorophyll a and nitrate and nitrite concentrations.
A second standardised PCA was performed on the matrix 32 years × 5 zooplankton species (Matrix 4). The first PC explained 35.7 % of the total variance. Examination of the first normalised eigenvector (not shown) showed that the first PC was positively correlated to A. bifilosa and M. slabberi densities. The second PC explained 23.6 % of the total variance. Examination of the second normalised eigenvector indicated that the second PC was negatively correlated to N. integer densities.
A third standardised PCA was performed on the matrix 32 years × fish species (Matrix 5). The first PC explained 21.7 % of the total variance. Examination of the first normalised eigenvector (not shown) showed that the first PC was positively correlated to twaite shad (Alosa fallax) and three-spined stickleback (Gasterosteus aculeatus) densities. The second PC explained 18.2 % of the total variance. Examination of the normalised eigenvector (not shown) revealed that the second PC was positively related to the European flounder (Platichthys flesus) densities and negatively related to the European sprat (Sprattus sprattus) densities.
Analysis 3: Examination of the Relationships Between Principal Components and Other Related Variables
The cluster analysis on variables showed there were two groups (Fig. 4a). Group 1 mainly consisted of variables that were correlated (positively or negatively) and the second group (Group 2) of variables that were poorly correlated. We used Group 1 and four parameters (Env. PC1, Zooplankton PC1, local air temperature and Fish PC2) quite highly correlated from the second group (Group 2) to perform a cluster analysis to identify homogeneous time periods and temporal discontinuities (Fig. 4b). The analysis revealed two abrupt shifts: the first occurred between 1987 and 1988 and the second between 1996 and 1997 (and perhaps a third less marked shift, between 2001 and 2002).
Then we examined graphically all variables in the first group. First, we looked at long-term changes in hydro-climatological and meteo-oceanic indices (Fig. 5). This analysis showed a clear gradual shift around 1996. The first shift (1987–1988; only detectible on local air temperature) was much harder to detect probably due to the second being largest.
A group of (more local) variables more clearly revealed the first shift that occurred at the end of the 1980s, circa 1987 (Fig. 6) with synchronous switches in the sign of the first principal component scores of physical chemistry, the first principal component scores of zooplankton and the second principal component scores of fish.
The major change observed on climate data (Fig. 5) circa 1996 did not correspond to a shift in the local ecosystem parameters. Interestingly an abrupt change of the ecosystem state was observed at the beginning of the 2000s. This later change was therefore perceptible as a switch in the sign of the second principal component scores of physical chemistry, the second principal component scores of SST, local precipitations and the first principal component scores of fish (Fig. 7). This result has to be however nuanced regarding to the HAC results. Even if we graphically suggested a late shift in the Fish PC1, this PC seemed indeed linked to large-scale climatic variables, showing a less marked response (i.e., a possible shift at the end of the 1990s, see Fig. 7).
Discussion
As expected, large-scale meteo-oceanic indices did influence long-term spatial changes in annual SST and SLP. Long-term changes in annual SST that affect the Gironde estuary (first PC SST) are positively and highly correlated to the AMO and NHT anomalies. Interestingly, the state of the EAP explains a large proportion (52 % of the total variance) of the variance of long-term changes in annual SST. Although this index does not explain a significant part of the variance in the ecosystem state of the estuary, its effects on annual SST suggest that the EAP may be an important climatic proxy when bioclimatological investigations are conducted in the region. This pattern of atmospheric variability explains the long-term changes in the abundance of several species over Europe. For instance, this index is inversely correlated to the abundance of tiger moth Arctia caja in the UK (Conrad et al. 2003). The examination of the spatial changes of the influence of the NAO on annual SST suggests that this atmospheric oscillation is unlikely to influence the Gironde estuary through its effects on annual SST. This result is also in agreement with correlation maps already calculated in the North Atlantic sector (Dickson and Turrell 2000; Beaugrand and Reid 2003). However, it is possible that the NAO has a moderate or sporadic impact on the ecosystem state of the estuary through its effects on annual SLPs (Fig. 3), which influence both the atmospheric circulation and precipitation (Hurrell and Deser 2009). Our results are also in agreement with Goberville et al. (2010) who did not find any significant correlation between the state of the NAO and the state of the coastal systems along French coasts from the English Channel to the Mediterranean Sea. Beaugrand et al. (2000) found a significant correlation between the plankton community and the NAO in the English Channel, yet our investigation found this influence to be not statistically significant in the Bay of Biscay.
Our analyses showed that the warming of the Gironde estuary paralleled the rise in global temperatures with two pronounced accelerating phases circa 1987 and 1996 (Fig. 5). Investigating changes in SST in the large marine ecosystems, Belkin (2009) showed that many oceanic regions experienced a particularly intense warming after 1987 and 1995 that may constitute manifestations of an interannual variability. Reid and Beaugrand (2012) found a pronounced increase in SST at the end of the 1990s over all continental shelves of the oceans but the East Pacific. Although the first shift in sea temperatures coincided with a change in the Gironde ecosystem state (Fig. 6), the second shift was not followed by another abrupt ecosystem shift (Fig. 4 versus 5). Instead, we found an ecosystem shift after 2001 (Fig. 7), which possibly reflects a response to a substantial rise in ocean heat content observed at a global scale (Levitus et al. 2009).
The 1987 shift influenced both abiotic (water temperature, salinity, dissolved oxygen, SPM, river discharge and current speed) and biotic parameters such as zooplankton (e.g., A. bifilosa and M. slabberi) and fish (e.g., S. sprattus and Platichthyis flesus) (Fig. 6). This shift had severe consequences on the physical and chemical properties of the estuary. A strong warming was observed, together with the acceleration of the salinity rise (David et al. 2005). This process reflecting an increased intrusion of marine waters, was first termed ‘marinisation’ by (David et al. 2007a, b). The phenomenon of marinisation was associated to a decrease in river discharge and river runoff, originating from the Garonne river (not significant in the Dordogne river, results not shown) also likely to have been reinforced by the reduction in precipitation that occurred after ∼2000 (see Fig. 7). The phenomenon of marinisation has probably been exacerbated by the increasing demand in irrigation that has in turn reduced turbidity (ΔSPM = −0.31 g l−1) with a decrease in the number of days of presence of the maximum turbidity zone (MTZ) in the middle part of the estuary (David et al. 2005; Coupry et al. 2008). The increase in both temperature and salinity may have favoured M. slabberi and A. bifilosa. These species are probably the best indicator of the change that occurred at the end of the 1980s. These results are in agreement with the observations of David et al. (2005) who showed that high abundances of A. bifilosa were positively correlated with high salinities and temperatures. A. bifilosa longitudinal distribution along the estuary could be strongly driven by salinity and water temperature, the species having optimal haline and thermal values centered on 16 and 20 °C, respectively (Chaalali et al. 2013). Its increasing densities observed in the median part of the Gironde estuary (sampling site E, Fig. 1) could possibly reflect the spatial shift observed in A. bifilosa distribution as the species was preferentially found in more downstream estuarine parts 30 years ago (Chaalali et al. 2013). In the context of water warming and marinisation, upstream regions of the estuary would more correspond to the environmental envelop of A. bifilosa. This could also constitute a response of A. bifilosa to the successful establishment of A. tonsa (Aravena et al. 2009). Concerning M. slabberi, it is known to be a marine species living in coastal areas and using estuaries for juvenile development (especially in summer; Mouny et al. 2000). Its increase in density could therefore also reflect the marinisation process observed in the Gironde (Chaalali et al. 2013; David et al. 2005).
After the 1987 shift, some fish species that use the system as a nursery ground experienced a significant change in their abundance in response to both salinity and temperature increases. This was particularly noticeable for the European sprat (S. sprattus), which increased its density (Pasquaud, personal communication). In contrast, the European flounder (P. flesus) diminished (results not shown). Pasquaud et al. (2012) pointed out that the increase in salinity had a positive effect on the majority of small marine pelagic fish densities, probably offering more suitable conditions, especially for Engraulis encrasicolus and S. sprattus. Opposite trends were also observed, especially for catadromous species (i.e., the smelt, Pronier and Rochard 1998), that are known to be sensitive to climate changes and are even expected to disappear from most basins located in their southern distribution limit (Lassalle and Rochard 2009). The example of the decrease in density of European flounder in the Bay of Biscay continental shelf seems to support our results (Cabral and Ohmert 2001).
The 1987 abrupt shift detected in the Gironde estuary has also been observed in other temperate northern ecosystems. For example, Reid and Edwards (2001) detected an abrupt ecosystem shift in the North Sea circa 1988. These authors found substantial biological changes ranging from phytoplankton (e.g., Phytoplankton Colour Index) to zooplankton (e.g., Calanus finmarchicus and Calanus helgolandicus) and fish (e.g., Trachurus trachurus). These alterations were also identified in the benthic community structure of the southern North Sea (Kroncke et al. 1998). Weijerman et al. (2005) analysing an important amount of both biological and physical data found a pronounced modification in the ecosystem after 1987 in the North Sea. Subsequent analyses revealed that food web interactions were altered after the shift (Kirby and Beaugrand 2009). Other ecosystems exhibited an abrupt shift at about the same time. In the Baltic Sea, concomitant changes were also observed from phytoplankton to zooplankton to fish after about 1987 (Alheit et al. 2005). For instance, a reduction in the abundance of diatoms was accompanied by an increase in the abundance of dinoflagellates as a response to a pronounced/severe warming. The abundance of the key copepods C. finmarchicus and Pseudocalanus spp. collapsed whereas both Temora longicornis and Acartia spp. remained elevated. Similar abrupt ecosystem shifts were described in the Mediterranean Sea (Conversi et al. 2010) and even in some European lakes (Anneville et al. 2002). Recent findings also suggest that similar changes happened in other basins such as the Black Sea (Conversi et al. 2010). Our analysis on global SST allows us to better understand changes that appeared in the Gironde estuary (Fig. 2a), the North and Baltic Seas as a change is detected circa 1987 in all these regions. It is however unlikely that the connection occurred through the NAO as suggested by Conversi et al. (2010) because the effect of the NAO on temperature is not spatially uniform (Dickson and Turrell 2000) (see also Fig. 2b).
A second abrupt ecosystem shift was detected circa 2001 (Fig. 7). However, it did not follow the major climatic shift observed after about 1996, which triggered some abrupt shifts/changes in the regions close to the Gironde estuary. Luczak et al. (2011) found a concomitant change in zooplankton, European anchovy and sardine (Sardina pilchardus) abundances and in the occurrence of the Balearic shearwater (Puffinus mauretanicus). This change that took place after 1995 was observed in a region corresponding to the Bay of Biscay and the Celtic Sea, as well as in areas around the UK. These biological changes were attributed to an increase in SST. Our results show that the responses of biological systems to hydro-climatological changes are inherently nonlinear and complex, with potential lags difficult to identify in a retrospective study. The more recent abrupt shift in the dynamic regime (sensu, Scheffer 2009) of the Gironde estuary might be related to the substantial rise in ocean heat content documented at a global scale (Levitus et al. 2009; Reid and Beaugrand 2012) after the end of the 1990s. Above, we used the terminology ‘dynamic regime’, instead of ‘stable state’ (of the Gironde estuary) since cycles and intrinsic fluctuations are the more frequent functioning way in nature (Scheffer 2009). But this ecosystem change also coincided with a major alteration in the coastal systems seen in both nutrient concentrations and dissolved oxygen and observed along the western part of the French coast. These changes would be driven by an alteration in atmospheric circulation related mainly to zonal wind, which in turn affected regional patterns of precipitation (Goberville et al. 2010). One of the hydrological consequences of this change consisted of a major decrease in the river discharge leading to falling chlorophyll a and nitrate and nitrite concentrations (see Fig. 7) as a result of lower continental inputs and runoff, an effect also suggested by Goberville et al. (2010). Some fish species such as the twaite shad (A. fallax), the three-spined stickleback (G. aculeatus) experienced this abrupt shift. The twaite shad densities regressed at this period (see Fig. 7). This anadromous species using the estuary as a nursery ground and juvenile stages are known to be more sensitive to hypoxic or anoxic conditions (the young shad growth being affected by deteriorated dissolved oxygen conditions, Maes et al. 2008). Moreover recently, the estuary experienced particularly pronounced drought events (Etcheber et al. 2011) associated with reduced precipitation and river discharge (see Fig. 7), leading to higher turbidities and consequently to greater bacterial activity (especially at low tide and during summer) with possible anoxic consequences (Heip et al. 1995). It would also be interesting to further investigate the incidence of potential anoxic and hypoxic events on the estuarine food web, especially on primary producers and zooplankton (Kromkamp et al. 1995; Irigoien and Castel 1997; David et al. 2007a, b). Thus, a reduction in zooplankton production and in the organisms' food collecting abilities was documented (Gasparini et al. 1999; David et al. 2007a, b; Gonzalez-Ortegon et al. 2010). At a higher level, fish suffered sub-lethal consequences resulting from the clogging of their gills or from a decrease in their reproductive success with more abnormal larvae and mortality (Marchand 1993; Marshall and Elliott 1998; Griffin et al. 2009).
Despite a bias induced by the sampling strategy, our results seem to be supported by the study of Taylor et al. (2006) that showed a diminution of the three-spined stickleback associated with an increase in turbidity (linked in our case to the hydrological consequences of this second ecosystem shift).
Our results suggest that despite local anthropogenic influences, the Gironde estuary experienced two abrupt shifts that altered the system, from the physical and chemical environment to plankton and fish. However, dynamical processes, through which climate influence might have propagated through the system, remain difficult to separate. Although mechanisms and potential intermediate pathways leading to these shifts remain elusive, our analyses establish a clear link between the 1987 abrupt ecosystem shift and an alteration in large-scale hydro-climatological forcing, which triggered other shifts at the same time in other European regions, including terrestrial systems (Anneville et al. 2002). The second shift which occurred after 2001 cannot be read as a direct response of the ecosystem to the pronounced climatic shift because it was detected a few years later. Our results suggest that the alteration occurred through regional hydro-climatic channels such as the effect of precipitation on river runoff.
References
Abril, G., H. Etcheber, P. Le Hir, P. Bassoullet, B. Boutier, and M. Frankignoulle. 1999. Oxic/anoxic oscillations and organic carbon mineralization in an estuarine maximum turbidity zone (The Gironde, France). Limnology and Oceanography 44: 1304–1315.
Alheit, J., C. Mollmann, J. Dutz, G. Kornilovs, P. Loewe, V. Mohrholz, and N. Wasmund. 2005. Synchronous ecological regime shifts in the central Baltic and the North Sea in the late 1980s. Ices Journal of Marine Science 62: 1205–1215.
Allen, G. P., P. Castaing, and Klingebi.A. 1972. Study on circulation of water masses at mouth of Gironde. Comptes Rendus Hebdomadaires Des Seances De L Academie Des Sciences Serie D 275:181.
Anneville, O., S. Souissi, F. Ibanez, V. Ginot, J.C. Druart, and N. Angeli. 2002. Temporal mapping of phytoplankton assemblages in Lake Geneva: Annual and interannual changes in their patterns of succession. Limnology and Oceanography 47: 1355–1366.
Aravena, G., F. Villate, I. Uriarte, A. Iriarte, and B. Ibáñez. 2009. Response of Acartia populations to environmental variability and effects of invasive congenerics in the estuary of Bilbao, Bay of Biscay. Estuarine, Coastal and Shelf Science 83: 621–628.
Attrill, M.J., and M. Power. 2002. Climatic influence on a marine fish assemblage. Nature 417: 275–278.
Bamber, R.N., and R.M.H. Seaby. 2004. The effects of power station entrainment passage on three species of marine planktonic crustacean, Acartia tonsa (Copepoda), Crangon crangon (Decapoda) and Homarus gammarus (Decapoda). Marine Environmental Research 57: 281–294.
Barnston, A.G., and R.E. Livezey. 1987. Classification, seasonality and persistence of low-frequency atmospheric circulation patterns. Monthly Weather Review 115: 1083–1126.
Beaugrand, G., M. Edwards, K. Brander, C. Luczak, and F. Ibanez. 2008. Causes and projections of abrupt climate-driven ecosystem shifts in the North Atlantic. Ecology Letters 11: 1157–1168.
Beaugrand, G., F. Ibanez, and P.C. Reid. 2000. Spatial, seasonal and long-term fluctuations of plankton in relation to hydroclimatic features in the English Channel, Celtic Sea and Bay of Biscay. Marine Ecology Progress Series 200: 93–102.
Beaugrand, G., C. Luczak, and M. Edwards. 2009. Rapid biogeographical plankton shifts in the North Atlantic Ocean. Global Change Biology 15: 1790–1803.
Beaugrand, G., and P.C. Reid. 2003. Long-term changes in phytoplankton, zooplankton and salmon related to climate. Global Change Biology 9: 801–817.
Beaugrand, G., P.C. Reid, F. Ibanez, J.A. Lindley, and M. Edwards. 2002. Reorganization of North Atlantic marine copepod biodiversity and climate. Science 296: 1692–1694.
Béguer, M., S. Pasquaud, P. Noël, M. Girardin, and P. Boët. 2008. First description of heavy skeletal deformations in Palaemon shrimp populations of European estuaries: The case of the Gironde (France). Hydrobiologia 607: 225–229.
Belkin, I.M. 2009. Rapid warming of large marine ecosystems. Progress in Oceanography 81: 207–213.
Bopp, L., C. Le Quere, M. Heimann, A.C. Manning, and P. Monfray. 2002. Climate-induced oceanic oxygen fluxes: Implications for the contemporary carbon budget. Global Biogeochemical Cycles 16: 6.1–6.13.
Carpenter, S.R., and W.A. Brock. 2006. Rising variance: A leading indicator of ecological transition. Ecology Letters 9: 308–315.
Cabral, H.N., and B. Ohmert. 2001. Diet of juvenile meagre, Argyrosomus regius within the Tagus estuary. Cahiers de Biologie Maine 42: 289–293.
Chaalali, A., X. Chevillot, G. Beaugrand, V. David, C. Luczak, P. Boët, A. Sottolichio, and B. Sautour (2013). Changes in the distribution of copepods in the Gironde estuary: A warming and marinisation consequence? Estuarine, Coastal and Shelf Science. doi:10.1016/j.ecss.2012.12.004
Cloern, J.E., K.A. Hieb, T. Jacobson, B. Sanso, E. Di Lorenzo, M.T. Stacey, J.L. Largier, W. Meiring, W.T. Peterson, T.M. Powell, M. Winder, and A.D. Jassby. 2010. Biological communities in San Francisco Bay track large-scale climate forcing over the North Pacific. Geophysical Research Letters 37: L21602.
Conrad, K.F., I.P. Woiwod, and J.N. Perry. 2003. East Atlantic teleconnection pattern and the decline of a common arctiid moth. Global Change Biology 9: 125–130.
Conversi, A., S.F. Umani, T. Peluso, J.C. Molinero, A. Santojanni, and M. Edwards. 2010. The Mediterranean Sea regime shift at the end of the 1980s, and intriguing parallelisms with other European basins. PloS One 5: e10633.
Coupry, B., M. Neau, and T. Leurent. 2008. Évaluation des impacts du changement climatique sur l'estuaire de la Gironde et prospective à moyen terme. EAUCEA.
David, V., B. Sautour, and P. Chardy. 2007a. The paradox between the long-term decrease of egg mass size of the calanoid copepod Eurytemora affinis and its long-term constant abundance in a highly turbid estuary (Gironde estuary, France). Journal of Plankton Research 29: 377–389.
David, V., B. Sautour, and P. Chardy. 2007b. Successful colonization of the calanoid copepod Acartia tonsa in the oligo-mesohaline area of the Gironde estuary (SW France)—natural or anthropogenic forcing? Estuarine Coastal and Shelf Science 71: 429–442.
David, V., B. Sautour, P. Chardy, and M. Leconte. 2005. Long-term changes of the zooplankton variability in a turbid environment: The Gironde estuary (France). Estuarine, Coastal and Shelf Science 64: 171–184.
Decastro, M., N. Lorenzo, J.J. Taboada, M. Sarmiento, I. Alvarez, and M. Gomez-Gesteira. 2006. Influence of teleconnection patterns on precipitation variability and on river flow regimes in the Mino River basin (NW Iberian Peninsula). Climate Research 32: 63–73.
Dickson, R.R., and W.R. Turrell. 2000. The NAO: The dominant atmospheric process affecting oceanic variability in home, middle, and distant waters of European Atlantic salmon. In The ocean life of Atlantic salmon. Environmental and biological factors influencing survival, ed. D. Mills. Bodmin: Fishing News Books.
Edwards, M., and A.J. Richardson. 2004. Impact of climate change on marine pelagic phenology and trophic mismatch. Nature 430: 881–884.
Enfield, D.B., A.M. Mestas-Nunez, and P.J. Trimble. 2001. The Atlantic multidecadal oscillation and its relation to rainfall and river flows in the continental US. Geophysical Research Letters 28: 2077–2080.
Etcheber, H., S. Schmidt, A. Sottolichio, E. Maneux, G. Chabaux, J.M. Escalier, H. Wennekes, H. Derriennic, M. Schmeltz, L. Quemener, M. Repecaud, P. Woerther, and P. Castaing. 2011. Monitoring water quality in estuarine environments: Lessons from the MAGEST monitoring program in the Gironde fluvial-estuarine system. Hydrology and Earth System Sciences 15: 831–840.
Gasparini, S., J. Castel, and X. Irigoien. 1999. Impact of suspended particulate matter on egg production of the estuarine copepod, Eurytemora affinis. Journal of Marine Systems 22: 195–205.
Goberville, E., G. Beaugrand, B. Sautour, and P. Treguer. 2010. Climate-driven changes in coastal marine systems of Western Europe. Marine Ecology Progress Series 408: 129–U159.
Goberville, E., G. Beaugrand, B. Sautour, and P. Treguer. 2011. Evaluation of coastal perturbations: A new mathematical procedure to detect changes in the reference state of coastal systems. Ecological Indicators 11: 1290–1300.
Gonzalez-Ortegon, E., M.D. Subida, J.A. Cuesta, A.M. Arias, C. Fernandez-Delgado, and P. Drake. 2010. The impact of extreme turbidity events on the nursery function of a temperate European estuary with regulated freshwater inflow. Estuarine, Coastal and Shelf Science 87: 311–324.
Griffin, F.J., E.H. Smith, C.A. Vines, and G.N. Cherr. 2009. Impacts of suspended sediments on fertilization, embryonic development, and early larval life stages of the Pacific herring, Clupea pallasi. The Biological Bulletin 216: 175–187.
Halpern, B.S., S. Walbridge, K.A. Selkoe, C.V. Kappel, F. Micheli, C. D'Agrosa, J.F. Bruno, K.S. Casey, C. Ebert, H.E. Fox, R. Fujita, D. Heinemann, H.S. Lenihan, E.M.P. Madin, M.T. Perry, E.R. Selig, M. Spalding, R. Steneck, and R. Watson. 2008. A global map of human impact on marine ecosystems. Science 319: 948–952.
Hatun, H., M.R. Payne, G. Beaugrand, P.C. Reid, A.B. Sando, H. Drange, B. Hansen, J.A. Jacobsen, and D. Bloch. 2009. Large bio-geographical shifts in the north-eastern Atlantic Ocean: From the subpolar gyre, via plankton, to blue whiting and pilot whales. Progress in Oceanography 80: 149–162.
Heip, C. H. R., N. K. Goosen, P. M. J. Herman, J. Kromkamp, J. J. Middelburg, and K. Soetaert. 1995. Production and consumption of biological particles in temperate tidal estuaries, vol 33. In Oceanography and marine biology—an annual review, 1–149. London: U C L Press Ltd
Hurrell, J.W. 1995. Decadal trends in the North Atlantic Oscillation: regional temperatures and precipitations. Science 269: 676–679.
Hurrell, J.W., J.W.Y. Kushnir, and M. Visbeck. 2001. The North Atlantic Oscillation. Science 291: 603–605.
Hurrell, J.W., and C. Deser. 2009. North Atlantic climate variability: The role of the North Atlantic Oscillation. Journal of Marine Systems 78: 28–41.
Imbert, H., S. de Lavergne, F. Gayou, C. Rigaud, and P. Lambert. 2008. Evaluation of relative distance as new descriptor of yellow European eel spatial distribution. Ecology of Freshwater Fish 17: 520–527.
Irigoien, X., and J. Castel. 1997. Light limitation and distribution of chlorophyll pigments in a highly turbid estuary: The Gironde (SW France). Estuarine, Coastal and Shelf Science 44: 507–517.
Kirby, R.R., and G. Beaugrand. 2009. Trophic amplification of climate warming. Proceedings of the Royal Society B 276: 4095–4103.
Knight, J.R., R.J. Allan, C.K. Folland, M. Vellinga, and M.E. Mann. 2005. A signature of persistent natural thermohaline circulation cycles in observed climate. Geophysical Research Letters 32: 125–143.
Kromkamp, J., J. Peene, P. Vanrijswijk, A. Sandee, and N. Goosen. 1995. Nutrients, light and primary production by phytoplankton and microphytobenthos in the eutrophic, turbid Westerschelde estuary (The Netherlands). Hydrobiologia 311: 9–19.
Kroncke, I., J.W. Dippner, H. Heyen, and B. Zeiss. 1998. Long-term changes in macrofaunal communities off Norderney (East Frisia, Germany) in relation to climate variability. Marine Ecology Progress Series 167: 25–36.
Lance, G.N., and W.T. Williams. 1967. A general theory of classificatory sorting strategies. I. Hierarchical systems. The Computer Journal 9: 60–64.
Lassalle, G., and E. Rochard. 2009. Impact of 21st century climate change on diadromous fish spread over Europe, North Africa and the Middle East. Global Change Biology 15: 1072–1089.
Legendre, P., and L. Legendre. 1998. Numerical ecology, 2nd edn. Amsterdam: Elsevier.
Levitus, S., J.I. Antonov, T.P. Boyer, R.A. Locarnini, H.E. Garcia, and A.V. Mishonov. 2009. Global ocean heat content 1955–2008 in light of recently revealed instrumentation problems. Geophysical Research Letters 36: L07608.
Lobry, J., L. Mourand, E. Rochard, and P. Elie. 2003. Structure of the Gironde estuarine fish assemblages: A comparison of European estuaries perspective. Aquatic Living Resources 16: 47–58.
Luczak, C., G. Beaugrand, M. Jaffré, and S. Lenoir. 2011. Climate change impact on Balearic shearwater through a trophic cascade. Biology Letters 7:702–705.
Maes, J., M. Stevens, and J. Breine. 2008. Poor water quality constrains the distribution and movements of twaite shad Alosa fallax fallax (Lacepede, 1803) in the watershed of river Scheldt. Hydrobiologia 602: 129–143.
Marchand, J. 1993. The influence of seasonal salinity and turbidity maximum variations of the nursery function of the Loire estuary (France). Netherlands Journal of Aquatic Ecology 27: 427–436.
Marshall, J., Y. Kushner, D. Battisti, P. Chang, A. Czaja, R. Dickson, J. Hurrell, M. McCartney, R. Saravanan, and M. Visbeck. 2001. North Atlantic climate variability: Phenomena, impacts and mechanisms. International Journal of Climatology 21: 1863–1898.
Marshall, S., and M. Elliott. 1998. Environmental influences on the fish assemblage of the Humber estuary, UK. Estuarine, Coastal and Shelf Science 46: 175–184.
Mouny, P., J.-C. Dauvin, and S. Zouhiri. 2000. Benthic boundary layer fauna from the Seine estuary (eastern English Channel, France): spatial distribution and seasonal changes. Journal of the Marine Biological Association of the United Kingdom 80: 959–968.
Nicolas, D., A. Chaalali, H. Drouineau, J. Lobry, A. Uriarte, A. Borja, and P. Boët. 2011. Impact of global warming on European tidal estuaries: Some evidence of northward migration of estuarine fish species. Regional Environmental Change 11: 639–649.
Pasquaud, S., M. Béguer, M. Hjort Larsen, A. Chaalali, E. Cabral, and J. Lobry. 2012. Increase of marine juvenile fish abundances in the middle Gironde estuary related to warmer and more saline waters, due to global changes. Estuarine, Coastal and Shelf Science 104: 46–53.
Pauly, D. 2003. Ecosystem impacts of the world's marine fisheries. Global Change NewsLetter 55: 21–23.
Perry, A.L., P.J. Low, J.R. Ellis, and J.D. Reynolds. 2005. Climate change and distribution shifts in marine fishes. Science 308: 1912–1915.
Pronier, O., and E. Rochard. 1998. Fonctionnement d'une population d'éperlan (Osmerus eperlanus, Osmériformes Osmeridae) située en limite méridionale de son aire de répartition, influence de la température. Bulletin français de la pêche et de la pisciculture 350–351: 479–497.
Raitsos, D.E., G. Beaugrand, D. Georgopoulos, A. Zenetos, A.M. Pancucci-Papadopoulou, A. Theocharis, and E. Papathanassiou. 2010. Global climate change amplifies the entry of tropical species into the Eastern Mediterranean Sea. Limnology and Oceanography 55: 1478–1484.
Reid, P. C., and G. Beaugrand. 2012. Global synchrony of an accelerating rise in sea surface temperature. Journal of the Marine Biological Association of The United Kingdom 92, 1435–1450.
Reid, P. C., and M. Edwards. 2001. Long-term changes in the Pelagos, Benthos and fisheries of the North Sea. In Burning issues of North Sea ecology, 107–115. Stuttgart: E Schweizerbart'sche Verlagsbuchhandlung.
Rochard, E., G. Castelnaud, and M. Lepage. 1990. Sturgeons (Pisces, Acipenseridae)—threats and prospects. Journal of Fish Biology 37: 123–132.
Savoye, N., V. David, F. Morisseau, H. Etcheber, G. Abril, I. Billy, K. Charlier, G. Oggian, H. Derriennic, and B. Sautour. 2012. Origin and composition of particulate organic matter in a macrotidal turbid estuary: The Gironde Estuary, France. Estuarine, Coastal and Shelf Science 108: 16–28.
Sautour, B., and J. Castel. 1995. Comparative spring distribution of zooplankton in 3 macrotidal European estuaries. Hydrobiologia 311: 139–151.
Scavia, D., J.C. Field, D.F. Boesch, R.W. Buddemeier, V. Burkett, D.R. Cayan, M. Fogarty, M.A. Harwell, R.W. Howarth, C. Mason, D.J. Reed, T.C. Royer, A.H. Sallenger, and J.G. Titus. 2002. Climate change impacts on US coastal and marine ecosystems. Estuaries 25: 149–164.
Scheffer, M. 2009. Critical transitions in nature and society. Princeton: Princeton University Press
Taylor, E.B., J.W. Boughman, M. Groenenboom, M. Sniatynski, D. Schluter, and J.L. Gow. 2006. Speciation in reverse: Morphological and genetic evidence of the collapse of a three-spined stickleback (Gasterosteus aculeatus) species pair. Molecular Ecology 15: 343–355.
Weijerman, M., H. Lindeboom, and A.F. Zuur. 2005. Regime shifts in marine ecosystems of the North Sea and Wadden Sea. Marine Ecology Progress Series 298: 21–39.
Woodruff, S.D., R.J. Slutz, R.L. Jenne, and P.M. Steurer. 1987. A comprehensive ocean–atmosphere data set. Bulletin of the American Meteorological Society 68: 1239–1250.
Acknowledgements
We express our thanks to staff of the National Institute for the Sciences of the Universe (INSU), and especially of the Aquitaine Observatory of the Sciences of the Universe (OASU), to the Irstea Bordeaux and the entire SOMLIT team—technicians, researchers, captains and crews—who have contributed to the collection of samples since 1978. Data since 1997 are available at http://somlit.epoc.u-bordeaux1.fr/fr. Special thanks are due to Ms. Gaëlle Pauliac and Mr. Boris Phu for their comments and suggestions and to the anonymous referees for their valuable comments and revision that improved the quality of this paper. We also thank Méteo France for providing us meteorological data. This monitoring program was supported by Electricité de France (EDF), the Institut Français de Recherche pour l'Exploitation de la Mer (IFREMER) and the Centre National de la Recherche Scientifique (CNRS). This study was funded by the Conseil Régional d'Aquitaine.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaalali, A., Beaugrand, G., Boët, P. et al. Climate-Caused Abrupt Shifts in a European Macrotidal Estuary. Estuaries and Coasts 36, 1193–1205 (2013). https://doi.org/10.1007/s12237-013-9628-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-013-9628-x