Abstract
A dramatic increase in prevalence of the recently discovered bopyrid isopod parasite, Orthione griffenis, likely introduced in the 1980s from Asia to the Pacific coast of North America, coincided with the 2002 collapse of a population of its burrowing mud shrimp host, Upogebia pugettensis, in Willapa Bay, Washington that had been stable since monitoring began in 1988. An examination of whether O. griffenis infections were sufficient to cause this decline and other recently noted U. pugettensis population collapses in Pacific Coast estuaries was conducted. O. griffenis prevalence was the highest in large reproductive-sized female shrimp and caused an estimated average 68% loss of U. pugettensis reproduction in Yaquina Bay, Oregon over a 5-year period. O. griffenis prevalence fluctuated from year to year, but trends were similar in all estuaries sampled. Uninfected shrimp transplanted back into locations from which they had disappeared acquired the parasite, suggesting that O. griffenis is extremely effective at finding its host even in estuaries with very low host density. Since both U. pugettensis and O. griffenis have pelagic larval stages, their population dynamics are also influenced by coastal nearshore oceanography and estuarine recruitment success. Coastwide lack of estuarine recruitment appears to coincide with declines in density of a co-occurring thalassinid shrimp, Neotrypaea californiensis, but cannot alone explain U. pugettensis population collapses. Although patterns observed to date could be explained by the presence of either a native or introduced parasitic castrator, assumptions of a resilient co-evolved host–parasite relationship do not apply for introduced species, so continued efforts to follow the spatial extent and consequences of the O. griffenis–U. pugettensis host–parasite relationship are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Upogebia pugettensis is a common species of thalassinid shrimp that inhabits intertidal mudflats in estuaries along the Pacific Northwest coast of North America. This species builds permanent y-shaped burrows (up to 1 m deep) in the sediment and filters large quantities of estuarine phytoplankton (Swinbanks and Luternauer 1987; Dumbauld et al. 1996; Griffen et al. 2004). U. pugettensis and a co-occurring thalassinid shrimp (Neotrypaea californiensis) are considered to be ecosystem engineers, substantially influencing the estuarine benthic community and carbon and nitrogen cycling in areas they inhabit (Bird 1982; Posey 1986; Dumbauld et al. 2001; Ferraro and Cole 2004; Griffen et al. 2004; D'Andrea and DeWitt 2009). Beds of both species once covered thousands of hectares (Hornig et al. 1989; Dewitt et al. 2004; Griffen et al. 2004) and thus included thousands of tons of shrimp biomass. Since 1998, however, most U. pugettensis populations examined between California and British Columbia have declined, and this shrimp appears to be at least locally extinct in Bodega Bay, Tomales Bay, Bolinas Lagoon, San Francisco Bay, and Elkhorn Slough, California where it was previously abundant (J.W. Chapman, B.R. Dumbauld, G. Itani, and J. C. Markham, unpublished manuscript).
A population of U. pugettensis in Willapa Bay, Washington that has been monitored since 1988 (Dumbauld et al. 1996; Dumbauld et al. 2004) declined precipitously at the same time that prevalence of a recently described parasite, the bopyrid isopod, Orthione griffenis Markham 2004, increased. Bopyrid isopods are parasitic castrators that effectively block reproductive capability of their crustacean hosts (Tucker 1930; Anderson 1977; O'Brien and Van Wyk 1985; Munoz and George-Nascimento 1999; Astete-Espinoza and Caceres-Martinez 2000; Calado et al. 2005). Thalassinid shrimp around the world are parasitized by bopyrid isopods (Markham 1986, 2001), but prevalence in natural populations is usually low (<5%). A native bopyrid, Phyllodurus abdominalis Stimpson 1857, parasitizes U. pugettensis (Markham 1977), but it has only rarely been observed in recent surveys. Likely introduced to North America from Asia before the late 1980s (though AMK and MET maintain that there is insufficient evidence to state this and confirmation of the source awaits further genetic analysis), O. griffenis is now widespread and infects U. pugettensis at high prevalence along the Pacific Coast of North America (Smith et al. 2008; Griffen 2009; Williams and An 2009; J.W. Chapman, B.R. Dumbauld, G. Itani, and J.C. Markham, unpublished manuscript). Relatively high prevalence of other bopyrid species has sometimes been observed, e.g., Progebiophilus bruscai on Upogebia dawsoni (Leija-Tristan and Salazar-Vallejo 1991), Parapenaeon consolidatum on Metapenaeopsis dalei (Choi et al. 2004), Entophilus omnitectus on Callianassa aquabaensis (Markham and Dworschak 2005), and Argeia pugettensis on Lissocrangon stylirostris (Marrin Jarrin and Shanks 2008); however, the origin of these parasites has not been questioned and population dynamics of parasite and host have not been monitored over time (but see Perez 1923 for a generalist cryptoniscid isopod Hemioniscus balani, a parasitic castrator of multiple species of barnacle hosts).
Parasites, including bopyrid isopods, are ubiquitous and their significant role in natural communities is becoming increasingly clear (Windsor 1998; Hudson et al. 2006; Dobson et al. 2008; Kuris et al. 2008). Laboratory and field studies have indicated that they can reduce host population densities and growth rates and alter community structure under specific conditions (Stiven 1964; Keymer 1981; Scott and Anderson 1984; Scott 1987; Crofton 1971; Anderson and May 1978; Hudson et al. 1998; Dobson and Hudson 1992). Mathematical models of parasite–host population dynamics assume that parasites are density-dependent sources of mortality or reproductive impairment and identify conditions where macroparasites and pathogens have the potential to regulate populations (Anderson and May 1979). While introduced parasites and diseases have been associated with species declines and extinctions (Warner 1969; Wilcove et al. 1998; Edgerton et al. 2004), most of the evidence for exotic species being the sole cause of extinctions is circumstantial (Gurevitch and Padilla 2004) and most research on parasite–host relations has been conducted in terrestrial environments.
Here, we evaluated data collected over a 20-year period on the bopyrid isopod parasite O. griffenis and its host U. pugettensis in one estuary and four additional years of data in three other estuaries along the Pacific Coast of North America. We used these data to address five questions that explore the host–parasite relationship for this isopod, but particularly to discern whether the association we observed between the appearance of O. griffenis and the population declines and local extinctions of U. pugettensis populations in these estuaries were causal:
-
1.
Is O. griffenis merely a parasitic castrator or can this parasite also cause host mortality? Conversely, did some other factor cause U. pugettensis to die or disappear from locations where it was once abundant?
-
2.
Does O. griffenis cause important loss of host fecundity at the population level?
-
3.
Both U. pugettensis and O. griffenis have pelagic larval stages that disperse in the coastal ocean and contribute to larger West Coast metapopulations. Do different estuarine populations respond individually or is there evidence of synchronicity in host and/or parasite recruitment and population dynamics?
-
4.
Parasite transmission is usually density dependent, allowing hosts to escape their parasites at low abundance. Is this the case for O. griffenis?
-
5.
O. griffenis was likely introduced to the US West Coast from Asia and thus did not co-evolve with U. pugettensis. Could this affect host–parasite population dynamics and host persistence?
Addressing these questions at the temporal and spatial scales necessary to test and ultimately resolve parasite host population dynamics in an open marine system is difficult. We were able to examine the maximum overall effects of O. griffenis on U. pugettensis population dynamics by comparing variations in U. pugettensis density and O. griffenis prevalence over an extended period in three estuaries and by comparing U. pugettensis reproduction and recruitment with later population changes in these estuaries. Finally, we estimated U. pugettensis reproductive losses to O. griffenis castration at the population level in order to gauge its potential magnitude.
Methods
Density and Prevalence
U. pugettensis were collected from a mudflat near the Cedar River channel in Willapa Bay, Washington from 1988 through 2002, after which they disappeared, and from a second location near Goose Point from 2003–2009 (see online resource 1 Table S1 and Fig. S1 for locations). Sampling occurred in late summer/early fall (August–November) from 1988 to 2003 and then in June from 2003 to 2009. Sampling began in 2004 at two locations in Tillamook Bay and Yaquina Bay, Oregon (online resource 1 Table S1 and Fig. S1). Quarterly samples of U. pugettensis were collected from Idaho Flats in Yaquina Bay from June 2006 to June 2007. Ten samples were taken at each location with a large corer (40 cm diameter × 60 cm depth). Contents were removed by shovel and sieved for shrimp (3-mm mesh, see Dumbauld et al. 1996 for methodology). Retained shrimp were measured (carapace length (CL) to the nearest 0.5 mm) and sex determined by the presence of anterior pleopods in females and absence in males. The presence of O. griffenis was easily noted by the characteristic bulging carapace over the branchial cavity. Detailed inspections of the branchial chamber for smaller newly settled isopods were made using a dissecting microscope for a subset of the samples taken from Yaquina Bay.
Mortality
An experiment designed to test whether O. griffenis might reduce U. pugettensis survival was conducted in the laboratory. Thirty-two infected and uninfected shrimp were placed in individual plastic centrifuge tubes (50 ml, 2.5 cm diameter) with holes drilled in the top and bottom to allow for water circulation to mimic the burrow environment. The tubes were gravity-fed filtered seawater from elevated header tanks to maintain constant head pressure and flow. Shrimp were measured at the start of the experiment and ranged in size from 15 to 28 mm CL with roughly equal sex ratios of both infected and uninfected shrimp. To evaluate potential interactions of diet and parasitism, shrimp were randomly separated into two treatments: fed (algae added to their respective header tank periodically) or unfed (no algae added). Shrimp molting and mortality were noted daily, and all surviving shrimp were returned to their tubes after recording size of exuvia and of newly molted and dead shrimp.
Reproduction
Since U. pugettensis extrude eggs in the fall (October–November) which hatch in the spring (February–April; Dumbauld et al. 1996), the annual samples taken in June–July missed the reproductive period. We therefore used historical data from Willapa Bay to estimate fecundity in uninfected U. pugettensis (Dumbauld et al. 1996). After 2005, the embryonic stage of eggs on the pleopods was recorded in quarterly data from Yaquina Bay when eggs were present. Eggs were removed from nine ovigerous uninfected female U. pugettensis sampled in Jan 2009 in Yaquina Bay to compare with historical fecundity. A subsample was counted from each brood, and both the subsample and the remaining portion were weighed (dry and wet weight) to estimate fecundity. We estimated the overall effect of O. griffenis prevalence at the population level by integrating castration effects over all female U. pugettensis size classes between 2005 and 2009 (20–32 mm CL by 1-mm increments). We conservatively assumed that O. griffenis castration affects only female shrimp reproduction and therefore estimated the maximum annual U. pugettensis reproductive potential without castration (F) as the sum of average individual fecundities, f i , in size classes i times the number of females, n i , per size class i:
We estimated cumulative reproduction with O. griffenis castration over all female reproductive size classes (F c ) using the relation:
where c i is the proportion of uncastrated females in size class i, based on size-adjusted isopod prevalence (logistic regression model, see “Statistical Analysis” section).
Recruitment
U. pugettensis postlarvae recruit from the ocean to estuaries along the West Coast of the USA from April to July (Dumbauld et al. 1996). Juvenile U. pugettensis were sampled along with older shrimp using the large corer described above from 1988 through 2003 in Willapa Bay; the 0+ age class was distinguished by size because they rapidly grew to 5–13 mm CL by late summer. Beginning in 2004 when large corer sampling shifted to June, separate samples using a 26.5-cm-diameter corer to 10 cm depth were required to sample the newly recruited shrimp (2–5 mm CL) in all three estuaries. Ten of these small cores were taken at each location, contents sieved through 1-mm mesh, and retained shrimp measured under a dissecting microscope (CL to the nearest 0.2 mm).
Survival
A transplant experiment was conducted to determine whether (1) U. pugettensis survived at locations where they were formerly present but had disappeared, (2) recruitment could be a mechanism limiting their recovery, and (3) such isolated shrimp become infected by O. griffenis. Newly recruited shrimp (5–12 mm CL) were collected from Yaquina Bay in September 2007, held and transported in seawater, and transplanted to both the Cedar River location in Willapa Bay from which they had disappeared and the Goose Pt. location where some remained. A second set of these shrimp was transplanted back to the high-density site in Yaquina Bay where they were collected and a second location where they were reported to have been previously abundant but had since disappeared. At all four locations, four shrimp were placed in each of four buckets (19 liters) which had the bottoms removed allowing existing shrimp and burrows to remain in contact with transplanted shrimp. In both estuaries at the locations where adults were present, we added a second treatment consisting of four buckets containing sieved sediment that had been allowed to settle for 24 h. The bottoms of these buckets were intact, preventing contact with and movement below the sediment. Only bottomless buckets were used at the locations where no shrimp were present. Mesh covers were placed over the tops of buckets for the first 24 h until transplanted shrimp had burrowed. All buckets were resampled in 2008, 1 year after placement, and all shrimp found were measured, sexed, and examined for the presence of O. griffenis.
Statistical Analysis
Isopod prevalence was analyzed using a logistic regression model (GLM function, binomial family, and logit link) in the statistical program R (R Development Core Team 2008) with shrimp host size (CL) as a continuous variable and host sex, location, and time entered as categorical factors. Final models were selected using Akaike Information Criteria (AIC; Burnham and Anderson 2002). Annual shrimp recruitment (mean density of recruits at a given location) was analyzed using standard least squares regression with larger juvenile and adult shrimp density as a continuous variable and year and estuary as categorical factors. Recruit density was ln-transformed to normalize data. Mortality experiment results were analyzed using a survival model in R with a censoring indicator to code for time of death and a parametric Weibull model in which hazard increases with time. Isopod presence, host sex and size, and whether the host molted were entered as factors. Ovigery was initially analyzed using a logistic regression model with shrimp host size as a continuous variable, but once the minimum size at maturity was reached, size was not a significant factor so simple contingency tables adequately described the effects of location and time. Results from the transplant experiment were analyzed with two-way ANOVA for U. pugettensis density and O. griffenis prevalence with location and treatment (bucket type) as fixed factors.
Results
Shrimp Density and Bopyrid Isopod Prevalence
U. pugettensis density at the initial sampling location near Cedar River in Willapa Bay was variable, but averaged 118 shrimp m−2 until 2002 when the population suddenly collapsed and no U. pugettensis were found at this location thereafter (Fig. 1). The abrupt decline in U. pugettensis abundance at this location was preceded by a dramatic increase in prevalence of O. griffenis after 1998, reaching over 50% prevalence in 2001 (Fig. 1). O. griffenis only infected larger mud shrimp (>15 mm CL), and prevalence appeared to be higher in female shrimp (Fig. 2). However, when data from the Cedar River location were analyzed with logistic regression, gender did not explain prevalence after controlling for shrimp size and year (online resource 1 Table S2).
Size (CL in millimeters) of male (top) and female (bottom) U. pugettensis collected at Cedar River, Willapa Bay before (1988–1997, left) and after O. griffenis became abundant (1998–2002, right). The higher overall prevalence of isopod parasites on female hosts can be explained by their presence on >18 mm CL shrimp which are presumably 2+ animals (presumed age classes divided by dashed lines) and a skewed sex ratio with more large females present regardless of whether the parasite is present
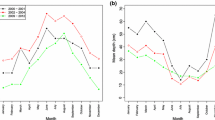
Density of U. pugettensis at sampling locations in Oregon estuaries (Tillamook Bay and Yaquina Bay) was the highest at Idaho Flats in Yaquina Bay (150 shrimp m−2) in 2005, increased at all locations in 2007, and declined thereafter, while density at the Willapa Bay location remained low and continued to decline (Goose Pt.; Fig. 3). O. griffenis prevalence varied from year to year but declined at most locations in 2007 and 2008 (Fig. 4). Patterns were similar at all locations; location was not a significant factor in the logistic regression model while host size and sex were (higher prevalence in females and increasing prevalence with size, Fig. 5, online resource 1 Table S3).
Proportion of U. pugettensis population infested by O. griffenis by size (CL in millimeters) and sex as predicted by logistic regression model (like that shown in online resource 1 Table S3, but averaged across years; \( {\hbox{Logit}} = 0.{\hbox{8846 CL}} - 0.0{\hbox{1337 C}}{{\hbox{L}}^2} - {13}.0{46}0 \) for females and \( {\hbox{Logit}} = 0.{8846} - 0.0{\hbox{1337 C}}{{\hbox{L}}^2} - {13}.{6388} \) for males). Frequency of infected shrimp was coded as 1 and shown as bars for each sex along the top and uninfected shrimp as zeros along the bottom. Note higher isopod prevalence in large female shrimp
Quarterly data collected for O. griffenis in Yaquina Bay suggested that isopod prevalence increases in summer months (\( \bar{x} = {64}\% \) and 73% in 2005 and 2006, respectively) and declines in the winter (\( \bar{x} = {48}\% \) and 49% in 2005 and 2006, respectively). This effect was significant when carapace size was accounted for in the logistic model; however, there was interaction caused by the influence of smaller shrimp, and host size was not significant when a separate analysis was conducted for shrimp >24 mm CL.
Several size classes of mud shrimp were routinely captured. In samples taken during the fall at the Cedar River location (1988 to 2002), only the first three smaller size classes were visually distinguishable and likely represented 0+, 1+, and 2+ age classes (Dumbauld et al. 1996; Fig. 2). In samples taken during early summer at other locations, newly recruited shrimp were rarely encountered and not accurately sampled using the large corer; consequently, these same size ranges likely represented 1+, 2+, and 3+ age classes. If these age class estimates are correct, then O. griffenis infects 1-year-old shrimp only in the latter part of their second summer and isopod prevalence is expected to increase during a shrimp cohort’s second year. Therefore, the large cohort of U. pugettensis which recruited in 2006 may have contributed to the decrease in average prevalence we observed in 2007 and perhaps 2008 before these relatively young shrimp also became infected.
Mortality
Some of the shrimp held in centrifuge tubes in the laboratory survived for the 108-day duration of our mortality experiment, but average survival was 62 days. There was no significant effect of isopod infection, shrimp size, or sex on survival, nor an effect of feeding (starved or fed; online resource 1 Table S4). Almost half of the shrimp molted during the course of the experiment (29 of 64) and molting and survival were positively correlated. The average length of shrimp survival was the greatest for shrimp that molted and were uninfected (86 days) but also higher (65 days) for infected shrimp that molted than for either infected or uninfected shrimp that did not molt (51 and 41 days, respectively; Fig. 6). While there were significant higher order interactions between molting and some of the other factors, AIC values suggest that molting was the most significant factor. When molting was removed from the model, none of the other factors, including infection, were significant (online resource 1 Table S4). Shrimp did not appear to increase in size and average molt increment was not significantly different (t test, p = 0.825) between infected and uninfected shrimp (0.42 and 0.49 mm, respectively).
Reproduction
Only U. pugettensis larger than 20 mm CL extruded eggs, and not all of these females were ovigerous when sampled during fall or early spring. No infected female shrimp sampled after 1998 at the Cedar River site in Willapa Bay were ovigerous. Ovigery in this Willapa Bay host population was significantly reduced from 35% of females in 1997 to only 1% in 2001 (Χ 20.05, 1 = 9.14, p = 0.002) just before the population crashed in 2002. The proportion of ovigerous shrimp sampled in Yaquina Bay fluctuated from 0% to 27% from the few samples collected during the fall and winter months of 2005 to 2009, but these interannual differences were not significant (Χ 20.05, 1 = 4.02, p = 0.4; Fig. 7). We found no difference in the relationship between size (CL) and fecundity (E) for uninfected U. pugettensis from Yaquina Bay sampled in January 2009 compared with that estimated by Dumbauld et al. (1996) before they were infected (E = 0.008 CL4.12), though the data were highly variable and fit a linear relationship as well. At the population level, size-corrected reproduction of uninfected (uncastrated) female U. pugettensis in Yaquina Bay between 2005 and 2009, F c, was estimated to be 32% (c = 0.32) of the expected reproduction without O. griffenis, indicating a reproductive loss (F − F c) of 68% (Fig. 8).
Recruitment
Annual U. pugettensis recruitment covaried in all estuaries with highest recruitment in 2006 and 2007 (Fig. 9). Recruitment was the highest to Yaquina Bay, and new recruits were only observed in Willapa Bay during 2006, the year of highest recruitment in all estuaries. A significant linear relationship between juvenile recruits and adult density was observed each year (Fig. 10; Table 1), possibly resulting from preferential settlement with adults; however, the highest density of adults also occurred in Yaquina Bay so this pattern may just reflect overall recruitment by estuary.
Survival
Newly recruited U. pugettensis transplanted in buckets survived for 1 year and grew from 5 to 10 mm CL when transplanted to 15–25 mm CL at all locations including those where they had previously disappeared. At locations where U. pugettensis populations remained, buckets with bottoms prevented larger shrimp from entering as expected; however, smaller shrimp recruited or moved into these buckets from the surrounding colony in Yaquina Bay, given that an average of 6.8 shrimp per bucket was retrieved and only four shrimp were originally transplanted. The only significant difference was attributable to location with highest density found within the existing U. pugettensis colony in Yaquina Bay (two-factor ANOVA, location F 1, 13 = 33.94, p < 0.001; treatment F 1, 13 = 1.29, p = 0.28, location × treatment F 1, 13 = 3.86, p = 0.07). Transplanted shrimp also became infected with O. griffenis. Moreover, three of four shrimp retrieved (75% prevalence) from the Cedar River location in Willapa Bay were infected though nearby hosts and therefore parasite source populations could not be found in a bay-wide survey (Dumbauld, unpublished data). No significant differences in isopod prevalence were found by treatment or location (two-factor ANOVA, location F 1, 13 = 0.13, p = 0.72; treatment F 1, 13 = 2.13, p = 0.17, location × treatment F 1, 13 = 0.13, p = 0.72).
Discussion
Prevalence of the bopyrid isopod parasite O. griffenis in U. pugettensis was high in all populations sampled from 2005 to 2009, infecting 17–94% of these shrimp. Historical prevalence of this isopod was low and increased exponentially beginning in 1997 at the Cedar River location in Willapa Bay. This remarkably rapid increase is correlated with the sharp decline and ultimate population collapse of its host, U. pugettensis, at this site and corroborated by direct accounts of the disappearance of this shrimp at many other locations along the Pacific Northwest coast of North America (J.W. Chapman, B.R. Dumbauld, G. Itani, and J. C. Markham, unpublished manuscript). We attempted to answer several questions in order to elucidate the underlying mechanism for these dynamics and further link these events.
Is O. griffenis Simply a Parasitic Castrator or Can It also Cause Host Mortality?
Bopyrid isopods cause blood loss and effectively castrate their decapod crustacean hosts (Kuris 1974; Walker 1977; O'Brien and Van Wyk 1985), but are not expected to cause mortality. Since U. pugettensis is likely a novel host for O. griffenis and some bopyrids are not full castrators, we wanted to verify this relationship for this host/parasite system. Smith et al. (2008) determined from field collections that O. griffenis caused significant weight loss in U. pugettensis but found the relationship to be variable and could not control for other possible factors like molting and feeding history. We found no evidence that infection, shrimp size, or sex affects survival of shrimp over a 108-day period in the laboratory. Furthermore, both infected and uninfected hosts molted during the experiment and although we saw a positive correlation between molting and shrimp survival, there were complex interactions between molting and other factors and no direct influence of infection on survival. This suggests that isopod presence itself is not a direct source of the shrimp’s disappearance from locations where they were historically abundant. Results of our reciprocal field transplant experiment also provided evidence that U. pugettensis could survive at locations such as the Cedar River site in Willapa Bay from which they had disappeared and that there was no significant difference between survival at these sites and locations where they are still present.
Changes in secondary sexual characteristics, particularly feminization of male hosts, are reported for other species of thalassinidean shrimps infected with bopyrids (Tucker 1930; Munoz and George-Nascimento 1999). This could indirectly influence mortality by changing the host’s susceptibility to predation, if, for example, males are more likely to leave their burrows and/or fight to mate with females (Bass and Weis 1999). While Griffen (2009) hypothesizes that this could be an important factor in the O. griffenis–U. pugettensis relationship, we found little evidence of male feminization (ovipores were consistently present on females with modified first pleopods and absent on males without these pleopods; as noted previously by Smith et al. 2008). We did not examine ovary and testis size or development; however, both male and female chela morphometry has been shown to be slightly but significantly affected in infected U. pugettensis (Woods et al. 2006). Tucker (1930) found feminization of male Upogebia pusilla (previously Upogebia littoralis) infected by Gyge branchialis but noted that secondary formation of female pleopods occurred primarily in those shrimp with large parasites, indicating that they were infected at smaller sizes before the onset of sexual maturity. Perhaps male feminization does not occur for U. pugettensis because infection by O. griffenis only occurs at larger host size. Tucker also found that infected males outnumbered females while females outnumbered males in uninfected shrimp. We saw no such trend for U. pugettensis with large female shrimp outnumbering males regardless of infection, presumably due to other sources of sex-specific direct or indirect mortality like the agonistic mating behavior mentioned above.
Does O. griffenis Impact U. pugettensis Fecundity?
We found significant effects of O. griffenis on female shrimp ovigery and fecundity. With the exception of two female shrimp infected with small isopods, no infected ovigerous females were observed in any of our collections; these two females bore reduced clutches of 15 eggs and 1,120 eggs versus an expected fecundity of 7,443 and 6,314 eggs, respectively (based on size–fecundity relationship in Dumbauld et al. 1996). Fecundity of uninfected shrimp collected from the extant Yaquina Bay population was similar to that observed historically in both Willapa Bay and Yaquina Bay populations (Bird 1982; Dumbauld et al. 1996). The high levels of parasite prevalence we observed between 2005 and 2009 led to significant reproductive loss (68%) at the population level in Yaquina Bay
A lack of information on postlarval recruitment relative to previous years’ fecundity complicates estimates of the reproductive costs of O. griffenis to U. pugettensis production at the population level. We therefore explored the potential effects of O. griffenis on population growth rate (dN/dt) as a function of population size N over time t between uninfested and infested populations of U. pugettensis using a logistic model (see online resource 2). Infection and therefore castration decreased population growth. For West Coast groundfish stocks, a precautionary management threshold of 40% unfished biomass is used as a rebuilding target for overfished stocks that could still be potentially very slow to recover (Hutchings 2000; NMFS 1999). We estimate that U. pugettensis populations previously could have reached 40% of unfished biomass in less than 2 years, but may require 7 years to do so under current conditions with egg production losses due to O. griffenis. Logistic growth estimates presume interactions similar to a fishery on reproductive females, yet the effects of O. griffenis on the dynamics of U. pugettensis populations are more complex. Lost reproduction increases the relative importance of all other factors affecting U. pugettensis population dynamics including larval production and survival, and the resilience of these populations is likely compromised by O. griffenis. Since this isopod does not affect U. pugettensis mortality (Fig. 6; online resource 1 Table S4), castrated U. pugettensis females and males remain in the population to compete with the remaining reproductive members and produce new O. griffenis propagules independent of reproduction by the uncastrated U. pugettensis population.
Is Shrimp Recruitment Affected by Lost Fecundity?
Annual U. pugettensis recruitment and O. griffenis prevalence were temporally synchronous among estuaries suggesting that both host and parasite are part of larger West Coast metapopulations which are greatly influenced by dynamics of the coastal ocean where their larval stages develop. We suspect that local U. pugettensis recruitment occurs in most years due to their relatively short 3-week pelagic larval period (Hart 1937; D'Andrea and Dumbauld, unpublished data). One hypothesis is that, in some years (those characterized by delayed spring transition and/or more wind relaxations), shrimp larvae transit the entire coast and larger populations to the south populate those to the north (due to the predominantly northward currents during this period), while in others (early transition and fewer relaxations), larvae are transported south or offshore and do not recruit anywhere in abundance. If true, then U. pugettensis populations in isolated estuaries or estuaries with few source populations to the south (e.g., Willapa Bay) would only receive nonlocal recruits in a few years (e.g., 2006; Fig. 9) and might decline rapidly once their populations became significantly infested and reproduction compromised by O. griffenis. While this physical oceanographic pattern enabled Shanks and Roegner (2007) to predict recruitment patterns for Dungeness crab (Cancer magister) along the Pacific Northwest coast of the USA, their larvae reside in the coastal ocean for several months and can transit broad areas of the continental shelf and settle in nearshore ocean areas as well. U. pugettensis larvae reside in the plankton for a much shorter period, must return to estuaries, and much less is known about the physical oceanographic processes in nearshore areas (<18.5 km from the shore) where their larvae are most abundant (D’Andrea and Dumbauld, unpublished data).
We were surprised by the rapid collapse of the mud shrimp population in Willapa Bay and intrigued by the correlation with rising prevalence of O. griffenis (Fig. 1), yet this may be merely a coincidence. Our results indicate that U. pugettensis recruitment also depends on density of adults, but estuary and adult shrimp densities are autocorrelated and differences between locations within estuaries are not sufficiently examined within a given year to clearly define the spatial scale at which recruitment and apparent aggregation occurs. Further, the concentrated recruitment of juvenile U. pugettensis into high-density areas of mature individuals could maintain patch density at the expense of patch area. Where recruitment is less than mortality, declining U. pugettensis abundances would result in declining patch areas in addition to declining density within patches. Movement of mature U. pugettensis is limited to extensions of their original burrows since these large shrimp are unable to reburrow after removal from their burrows (Chapman, unpublished data), but prereproductive U. pugettensis readily reburrow and anecdotal evidence indicates that such secondary dispersal of 1-year-old U. pugettensis has occurred (Feldman 2001). This adds further complexity to stationary measurements, and discrete U. pugettensis patches that diminish in size around stationary sampling locations, will appear as population collapses like those observed in Willapa Bay. Such declines will appear to occur much more slowly, if at all, however, when patches overlap with the stationary monitoring location. Observations suggest that this situation occurred at our long-term monitoring locations, but we are presently quantifying the pattern by accurately mapping the spatial distribution and abundance of U. pugettensis over time in the remaining Yaquina Bay population.
The recruitment dynamics of a co-occurring shrimp, N. californiensis, which is parasitized by another bopyrid isopod, Ione cornuta Bates 1862, provide an instructive comparison. We sampled N. californiensis using the same large corer at nearby locations in each estuary (see online resource 1 Table S1 and Fig. S1 for locations). I. cornuta, which infects all sizes of shrimp, was only detected four times (<1% prevalence) during 1988 to 2009 at our long-term monitoring location near the Palix River in Willapa Bay (online resource 1 Fig. S2), and its prevalence was below 4% at all sites monitored in Oregon and Washington estuaries (online resource 1 Table S5). Very few I. cornuta were collected (n = 30), and we found no relationship between prevalence and location or host size or sex (logistic regression all p values >0.10). Therefore, I. cornuta is clearly not affecting N. californiensis reproduction at the population level. Nonetheless, density of shrimp at the long-term monitoring location also declined from a maximum of 472 shrimp m−2 in 1995 to 52 shrimp m−2 in 2008 (online resource 1 Fig. S2) and declined at some, but not all, locations in Oregon estuaries from 2005 to 2008 (online resource 1 Fig. S3). While ocean conditions clearly affect larval survival and recruitment of both burrowing shrimp species, reduced natality due to O. griffenis infection would be expected to substantially augment declines in U. pugettensis recruitment. Furthermore, while N. californiensis density has declined, we are unaware of any population extinctions or collapses like those observed for U. pugettensis (J.W. Chapman, B.R. Dumbauld, G. Itani, and J. C. Markham, unpublished manuscript).
Finally, while little is known about O. griffenis recruitment, their larvae (microniscans) rely on a secondary planktonic host (a copepod) that is found in the nearshore ocean and not in the estuary (Dumbauld and Chapman, personal observation). Personal observations of higher abundance of the cryptoniscid stage which returns to the estuary suggest that O. griffenis recruitment occurs throughout the summer. Further stochasticity and a time lag therefore complicate host–parasite population dynamics.
Is Transmission of O. griffenis Density Dependent, Allowing U. pugettensis to Escape Its Effects at Low Host Abundance?
Host population size, parasite population size, and parasite transmission are critical parameters in epidemiological models (Anderson and May 1979; May and Anderson 1979; Fenton et al. 2002; Lafferty and Gerber 2002; Deredec and Courchamp 2003). One essential feature of these models is a density-dependent threshold in host population size below which parasite transmission cannot persist (first recognized by Kermack and McKendrick 1927). While both host and parasite populations can be affected by other phenomena including Allee effects at low population sizes, parasites are only expected to cause local host population extinctions under restricted circumstances (Kuris and Lafferty 1992; O'Keefe and Antonovics 2002; Deredec and Courchamp 2003; de Castro and Bolker 2005). Host extinction is most likely to occur when the parasite infects more than one host species (low specificity) with differential pathology in those hosts. Spillover to other less common hosts which act as reservoirs can also cause local or even global extinctions. This has been notably demonstrated for saturniid moths depleted by gypsy moth tachinid parasitoids and perhaps for the coconut moth and other zygaenid moths (Boettner et al. 2000; Kuris 2003). Extinctions might also occur if transmission is not density dependent (e.g., behaviorally controlled, as in sexually transmitted diseases) or when local spatial structure in density occurs for the host or parasite. Theoretical explorations of these effects have seldom been tested, particularly in marine systems (but see Blower and Roughgarden 1989; Lafferty 1993).
Results of our transplant experiment of uninfected shrimps to Willapa Bay where the population of U. pugettensis had collapsed to near zero and their subsequent infection within a year by O. griffenis at an equivalent prevalence to that in Yaquina Bay, where these shrimp were still relatively abundant and parasite prevalence also high, suggest that O. griffenis transmission was not locally density dependent. Similarly, while we saw interannual differences in O. griffenis prevalence, we found no significant differences in size-corrected prevalence between estuaries where shrimp density was markedly different. This was also true for populations sampled during 2008 from Morro Bay, California to Grays Harbor, Washington (J.W. Chapman, B.R. Dumbauld, G. Itani, and J. C. Markham, unpublished manuscript), suggesting that the density-dependent transmission threshold for O. griffenis is either extremely low or is influenced by factors that differ from those influencing its host. Two ways that this might occur are: (1) O. griffenis’s secondary planktonic host could be widespread and common in all years and (2) surviving infected U. pugettensis could act as temporary reservoir hosts, potentially allowing O. griffenis persistence below U. pugettensis Allee densities at least 1 year after shrimp recruitment declines or ends. Since reduced juvenile U. pugettensis abundances do not affect O. griffenis reproduction and each U. pugettensis female is not likely to produce more than three or four annual broods (Dumbauld et al. 1996), only three or four consecutive years of reproductive failure could be sufficient for local U. pugettensis extinction.
Could the Lack of Co-evolution Between U. pugettensis and O. griffenis Influence Host Parasite Population Dynamics and Host Persistence?
Previous empirical (Perez 1923; Blower and Roughgarden 1989; Torchin et al. 2001) and theoretical (Blower and Roughgarden 1987; Kuris and Lafferty 1992) evidence suggests that both native and introduced marine parasitic castrators can control host population demographics. The parasitic barnacle, Sacculina carcini, appears to limit size and biomass of green crabs, Carcinus maenas, across several native populations in Europe, but where this crab is introduced it is not infected with parasitic castrators and is more abundant than in its native range (Torchin et al 2001). Similarly, anecdotal information suggests that shipments to Chesapeake Bay containing live xanthid crabs (Rhithropanopeus harrisii) infected with the sacculinid barnacle, Loxothylacus panopaei (Van Engel et al. 1965), led to successful barnacle infection of this host along with another abundant xanthid, Eurypanopeus depressus. Both species of crabs soon became less common (Andrews 1980) while a third, formerly rare xanthid, Dyspanopeus sayi (= Neopanope texana sayi), was not susceptible to the barnacle and became the dominant mud crab in Chesapeake Bay (Andrews 1980). With the exception of work by Blower and Roughgarden (1987, 1989), who were able to predict biogeographic patterns of intra- and interspecific prevalence among three northeastern Pacific barnacle species and their cryptoniscid isopod parasite H. balani, our study is the first long-term quantitative description of the dynamics of an introduced marine parasitic castrator and its host.
Specialist predators and parasites coexist because they have not driven their prey to extinction over evolutionary time. Some introduced species may rapidly evolve or alter the evolution of other species in recipient communities (Vermeij 1996; Huey et al. 2005), but most introductions are not successful and result in rapid extinction due in part to Allee effects (e.g., Carlton and Geller 1993; Williams 1996; Taylor and Hastings 2005). The success of introduced species that do become established is often attributed to their escape from natural (co-evolved) enemies (Keane and Crawley 2002; Shea and Chesson 2002; Mitchell and Power 2003; Torchin et al. 2003). Similarly, the success of introduced predators and parasites can also be due to their escape from co-evolved relationships with their hosts and prey. Species invasions can provide insight into these co-evolved ecological relationships (Sax et al. 2007). In particular, invasions are consistent with “neoclassical biological control theory” which suggests that introductions can have maximum impacts on naïve prey when a reservoir of alternative hosts or prey is established and maintained (Hokkanen and Pimentel 1989; Kuris 2003). The pattern of local U. pugettensis population collapses we have observed can be explained for either a native or introduced parasitic castrator, but if O. griffenis was introduced and U. pugettensis was a naïve host as we suspect, then assumptions of a co-evolved host–parasite relationship would not apply and broader impacts of O. griffenis on U. pugettensis populations should continue to be examined.
Summary
The bopyrid parasite O. griffenis did not cause direct mortality of its host U. pugettensis, but significantly reduced fecundity at the estuarine population scale in all estuaries sampled. U. pugettensis recruitment and therefore population dynamics must also be affected. Because both host and parasite have pelagic larval stages, we have yet to resolve the complex dynamics of this relationship at the larger marine metapopulation scale. Infectious diseases and parasites are increasingly viewed as important forces controlling community dynamics. While available evidence suggests that they are rarely the sole cause of species extinctions (de Castro and Bolker 2005; Smith et al. 2006), marine parasitic castrators like O. griffenis can reduce host fitness to zero without increasing mortality and the castrated hosts compete with uninfected hosts while increasing their potential for new infestations on broader scales (Kuris and Lafferty 1992; Lafferty and Kuris 2002; McCallum et al. 2004; Kuris et al. 2008). Parasites can contribute to local host extinctions when they force host population instability (Boots and Sasaki 2003), when transmission is not density dependent, or when there is an abundant host reservoir (Boettner et al. 2000). Some of these factors appear to be at work in the nonco-evolved U. pugettensis–O. griffenis relationship. Due to the important role of these shrimp in West Coast estuaries (MacGinitie 1935; Dewitt et al. 2004; Dumbauld et al. 2004; Griffen et al. 2004; D'Andrea and DeWitt 2009), further investigation to understand recent declines and conserve remaining U. pugettensis populations is warranted.
References
Anderson, G. 1977. The effects of parasitism on energy flow through laboratory shrimp populations. Marine Biology 42: 239–251.
Anderson, R.M., and R.M. May. 1978. Regulation and stability of host–parasite population interactions. 1. Regulatory processes. The Journal of Animal Ecology 47: 219–247.
Anderson, R.M., and R.M. May. 1979. Population biology of infectious diseases. 1. Nature 280: 361–367.
Andrews, J.D. 1980. A review of introductions of exotic oysters and biological planning for new importations. Marine Fisheries Review 42: 1–11.
Astete-Espinoza, L.P., and C.W. Caceres-Martinez. 2000. Efecto del parasitismo del isopodo boprido Ionella agassizi (Isopoda: Epicaridea) (Bornnier 1900) sobre la fisiologia nutricional del nape Neotrypaea uncinata (M. Edwards, 1837) (Decapoda: Thalassinidea). Revista chilena de historia natural 73: 243–252.
Bass, C.S., and J.S. Weis. 1999. Behavioral changes in the grass shrimp, Palaemonetes pugio (Holthuis), induced by the parasitic isopod, Probopyrus pandalicola (Packard). Journal of Experimental Marine Biology and Ecology 241: 223–233.
Bird, E. M. 1982. Population dynamics of thalassinidean shrimps and community effects through sediment modification. Ph.D. dissertation, University of Maryland, College Park, Maryland, p.150
Blower, S., and J. Roughgarden. 1987. Population-dynamics and parasitic castration—a mathematical-model. The American Naturalist 129: 730–754.
Blower, S., and J. Roughgarden. 1989. Population-dynamics and parasitic castration—test of a model. The American Naturalist 134: 848–858.
Boettner, G.H., J.S. Elkinton, and C.J. Boettner. 2000. Effects of a biological control introduction on three nontarget native species of saturniid moths. Conservation Biology 14: 1798–1806.
Boots, M., and A. Sasaki. 2003. Parasite evolution and extinctions. Ecological Letters 6: 176–182.
Burnham, K.P., and D.E. Anderson. 2002. Model selection and multimodel inference: A practical information-theoretic approach, 2nd ed. New York: Springer.
Calado, R., C. Bartilotti, and L. Narciso. 2005. Short report on the effect of a parasitic isopod on the reproductive performance of a shrimp. Journal of Experimental Marine Biology and Ecology 321: 13–18.
Carlton, J.T., and J.B. Geller. 1993. Ecological roulette: The global transport of nonindigenous marine organisms. Science 261: 78–82.
Choi, J.H., G. Jamieson, K.H. Han, and S.Y. Hong. 2004. Parapenaeon consolidatum (Isopoda: Bopyridae) and the relative growth and reproduction of Metapenaeopsis dalei (Decapoda: Penaeidae) in South Korea. Journal of Shellfish Research 23: 237–242.
Crofton, H.D. 1971. Model of host–parasite relationships. Parasitology 63: 343–364.
D'Andrea, A.F., and T.H. DeWitt. 2009. Geochemical ecosystem engineering by the mud shrimp Upogebia pugettensis (Crustacea: Thalassinidae) in Yaquina Bay, Oregon: Density-dependent effects on organic matter remineralization and nutrient cycling. Limnology and Oceanography 54: 1911–1932.
de Castro, F., and B. Bolker. 2005. Mechanisms of disease-induced extinction. Ecological Letters 8: 117–126.
Deredec, A., and F. Courchamp. 2003. Extinction thresholds in host–parasite dynamics. Annales Zoologici Fennici 40: 115–130.
Dewitt, T. H., A. F. D'Andrea, C. A. Brown, B. D. Griffen, and P. M. Eldridge. 2004. Impact of burrowing shrimp populations on nitrogen cycling and water quality in western North American temperate estuaries. In Proceedings of the symposium on “Ecology of large bioturbators in tidal flats and shallow sublittoral sediments-from individual behavior to their role as ecosystem engineers”, 107–118. Nagasaki, Japan: Nagasaki University.
Dobson, A.P., and P.J. Hudson. 1992. Regulation and stability of a free-living host–parasite system—Trichostrongylus-tenuis in red grouse. 2. Population-models. The Journal of Animal Ecology 61: 487–498.
Dobson, A., K.D. Lafferty, A.M. Kuris, R.F. Hechinger, and W. Jetz. 2008. Homage to Linnaeus: How many parasites? How many hosts? Proceedings of the National Academy of Sciences of the United States of America 105: 11482–11489.
Dumbauld, B.R., D.A. Armstrong, and K.L. Feldman. 1996. Life-history characteristics of two sympatric thalassinidean shrimps, Neotrypaea californiensis and Upogebia pugettensis, with implications for oyster culture. Journal of Crustacean Biology 16: 689–708.
Dumbauld, B.R., K.M. Brooks, and M.H. Posey. 2001. Response of an estuarine benthic community to application of the pesticide carbaryl and cultivation of Pacific oysters (Crassostrea gigas) in Willapa Bay, Washington. Marine Pollution Bulletin 42: 826–844.
Dumbauld, B.R., K. Feldman, and D. Armstrong. 2004. A comparison of the ecology and effects of two species of thalassinidean shrimps on oyster aquaculture operations in the eastern North Pacific. In Proceedings of the symposium on “Ecology of large bioturbators in tidal flats and shallow sublittoral sediments-from individual behavior to their role as ecosystem engineers”, ed. A. Tamaki, 53–61. Nagasaki: Nagasaki University.
Edgerton, B.F., P. Henttonen, J. Jussila, A. Mannonen, P. Paasonen, T. Taughol, I. Edsman, and Souty-Grosset. 2004. Understanding the causes of disease in freshwater crayfish. Conservation Biology 18: 1466–1474.
Feldman, K. 2001. Contrasting patterns of habitat-specific recruitment success in sympatric species of thalassinidean shrimp: Effects of epibenthic bivalve shell with implications for population control in areas with commercial oyster aquaculture. Ph.D. dissertation, University of Washington, Seattle, Washington, p.194
Fenton, A., J.P. Fairbairn, R. Norman, and P.J. Hudson. 2002. Parasite transmission: Reconciling theory and reality. The Journal of Animal Ecology 71: 893–905.
Ferraro, S.P., and F.A. Cole. 2004. Optimal benthic macrofaunal sampling protocol for detecting differences among four habitats in Willapa Bay, Washington, USA. Estuaries 27: 1014–1025.
Griffen, B.D. 2009. Effects of a newly invasive parasite on the burrowing mud shrimp, a widespread ecosystem engineer. Marine Ecology Progress Series 391: 73–83.
Griffen, B.D., T.H. DeWitt, and C. Langdon. 2004. Particle removal rates by the mud shrimp Upogebia pugettensis, its burrow, and a commensal clam: effects on estuarine phytoplankton abundance. Marine Ecology Progress Series 269: 223–236.
Gurevitch, J., and D.K. Padilla. 2004. Are invasive species a major cause of extinctions? Trends in Ecology & Evolution 19: 470–474.
Hart, J.F.L. 1937. Larval and adult stages of British Columbia Anomura. Canadian Journal of Research 15: 179–219.
Hokkanen, H.M.T., and D. Pimentel. 1989. New associations in biological-control—theory and practice. The Canadian Entomologist 121: 829–840.
Hornig, S., S.D. Smith, and A. Sterling. 1989. Species Profiles: Life histories and environmental requirements of coastal fishes and invertebrates (Pacific Northwest): Ghost shrimp and blue mud shrimp. Biological Report, 1–14. Washington, D.C.: Fish and Wildlife Service.
Hudson, P.J., A.P. Dobson, and D. Newborn. 1998. Prevention of population cycles by parasite removal. Science 282: 2256–2258.
Hudson, P.J., M. Cattadori, B. Boag, and A.P. Dobson. 2006. Climate disruption and parasite–host dynamics: Patterns and processes associated with warming and the frequency of extreme climatic events. Journal of Helminthology 80: 175–182.
Huey, R.B., G.W. Gilchrest, and A.P. Hendry. 2005. Using invasive species to study evolution: case studies with Drosophila and salmon. In Species invasions: Insights into ecology, evolution, and biogeography, ed. D.F. Sax, J.J. Stachowicz, and S.D. Gaines, 139–164. Sunderland: Sinauer Associates.
Hutchings, J.A. 2000. Collapse and recovery of marine fishes. Nature 406: 882–885.
Keane, R.M., and M.J. Crawley. 2002. Exotic plant invasions and the enemy release hypothesis. Trends in Ecology & Evolution 17: 164–170.
Kermack, W.O., and A.G. McKendrick. 1927. Contributions to the mathematical theory of epidemics. 1. Proceedings of the Royal Society B-Biological Sciences 263: 75–81.
Keymer, A. 1981. Population-dynamics of Hymenolepis-diminuta in the intermediate host. The Journal of Animal Ecology 50: 941–950.
Kuris, A.M. 1974. Trophic interactions: Similarity of parasitic castrators to parasitoids. The Quarterly Review of Biology 49: 129–148.
Kuris, A.M. 2003. Did biological control cause extinction of the coconut moth, Levuana iridescens, in Fiji? Biological Invasions 5: 133–141.
Kuris, A.M., and K.D. Lafferty. 1992. Modeling crustacean fisheries—effects of parasites on management strategies. Canadian Journal of Fisheries and Aquatic Sciences 49: 327–336.
Kuris, A.M., R.F. Hechinger, J.C. Shaw, K.L. Whitney, L. Aguirre-Macedo, C.A. Boch, A.P. Dobson, E.J. Dunham, B.L. Fredensborg, T.C. Huspeni, J. Lorda, L. Mababa, F.T. Mancini, A.B. Mora, M. Pickering, N.L. Talhouk, M.E. Torchin, and K.D. Lafferty. 2008. Ecosystem energetic implications of parasite and free-living biomass in three estuaries. Nature 454: 515–518.
Lafferty, K.D. 1993. Effects of parasitic castration on growth, reproduction and population-dynamics of the marine snail Cerithidea californica. Marine Ecology Progress Series 96: 229–237.
Lafferty, K.D., and L.R. Gerber. 2002. Good medicine for conservation biology: The intersection of epidemiology and conservation theory. Conservation Biology 16: 593–604.
Lafferty, K.D., and A.M. Kuris. 2002. Trophic strategies, animal diversity and body size. Trends in Ecology & Evolution 17: 507–513.
Leija-Tristan, A., and S.I. Salazar-Vallejo. 1991. Parasitismo de Progebiophilus bruscai (Isopoda: Bopyridae) sobre el camaron Upogebia dawsoni (Thalassinoidea: Upogebiidae), en Baja California Sur, Mexico. Revista de Biología Tropical 39: 1–5.
MacGinitie, G.E. 1935. Ecological aspects of a California marine estuary. American Midland Naturalist 16: 629–765.
Markham, J.C. 1977. The status and systematic position of the bopyrid isopod genus Phyllodurus Stimpson, 1857. Proceedings. Biological Society of Washington 90: 813–819.
Markham, J.C. 1986. Evolution and zoogeography of Isopoda Bopyridae, parasites of Crustacea Decapoda. Crustacean Research 4: 143–164.
Markham, J.C. 2001. A review of the bopyrid isopods parasitic on thalassinidean decapods. In Crustacean issues: Isopod systematics and evolution, ed. B. Kensley and R.C. Brusca, 195–204. Amsterdam: Balkema.
Markham, J.C., and P.C. Dworschak. 2005. A new species of Entophilus Richardson, 1903 (Isopoda: Bopyridae: Entophilinae) from the Gulf of Aqaba, Jordan. Journal of Crustacean Biology 25: 413–419.
Marrin Jarrin, J.R., and A.L. Shanks. 2008. Ecology of a population of Lissocrangon stylirostris (Caridea: Crangonidae), with notes on the occurrence and biology of Its parasite, Argeia pugettensis (Isopoda: Bopyridae). Journal of Crustacean Biology 28: 613–621.
May, R.M., and R.M. Anderson. 1979. Population biology of infectious-diseases. 2. Nature 280: 455–461.
McCallum, H.I., A. Kuris, C.D. Harvell, K.D. Lafferty, G.W. Smith, and J. Porter. 2004. Does terrestrial epidemiology apply to marine systems? Trends in Ecology & Evolution 19: 585–591.
Mitchell, C.E., and A.G. Power. 2003. Release of invasive plants from fungal and viral pathogens. Nature 421: 625–627.
Munoz, G., and M. George-Nascimento. 1999. Efectos reproductivos reciprocos en la simbiosis entre napes (Decapods: Thalassinidea) e isopodos bopiridos (Isopoda: Epicaridea) en Lenga, Chile. Revista Chilena de Historia Natural 72: 49–56.
NMFS. 1999. Our living oceans. Report on the status of U.S. living marine resources, 1999. U.S. Dep. Commer., NOAA Tech. Memo. NMFS-F/SPO-41.
O'Brien, J.J., and P. Van Wyk. 1985. Effects of crustacean parasitic castrators (epicaridean isopods and rhizocephalan barnacles) on growth of crustacean hosts. In Crustacean issues: Factors in adult growth, ed. A.M. Wenner, 191–218. Rotterdam: Balkema.
O'Keefe, K.J., and J. Antonovics. 2002. Playing by different rules: The evolution of virulence in sterilizing pathogens. The American Naturalist 159: 597–605.
Perez, C. 1923. The castration of crustacean decapods parasited by Epicaridea. Comptes Rendus Hebdomadaires Des Seances De L Academie Des Sciences 176: 1934–1936.
Posey, M.H. 1986. Changes in a benthic community associated with dense beds of a burrowing deposit feeder, Callianassa californiensis. Marine Ecology Progress Series 31: 15–22.
R Development Core Team. 2008. R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. http://www.R-project.org
Sax, D.F., J.J. Stachowicz, J.H. Brown, J.F. Bruno, M.N. Dawson, S.D. Gaines, R.K. Grosberg, A. Hastings, R.D. Holt, M.M. Mayfield, M.I. O'Connor, and W.R. Rice. 2007. Ecological and evolutionary insights from species invasions. Trends in Ecology & Evolution 22: 465–471.
Scott, M.E. 1987. Regulation of mouse colony abundance by Heligmosomoides polygyrus (Nematoda). Parasitology 95: 111–129.
Scott, M.E., and R.M. Anderson. 1984. The population-dynamics of Gyrodactylus-bullatarudis (Monogenea) within laboratory populations of the fish host Poecilia-reticulata. Parasitology 89: 159–194.
Shanks, A.L., and G.C. Roegner. 2007. Recruitment limitation in Dungeness crab populations is driven by variation in atmospheric forcing. Ecology 88: 1726–1737.
Shea, K., and P. Chesson. 2002. Community ecology theory as a framework for biological invasions. Trends in Ecology & Evolution 17: 170–176.
Smith, K.F., D.F. Sax, and K.D. Lafferty. 2006. Evidence for the role of infectious disease in species extinction and endangerment. Conservation Biology 20: 1349–1357.
Smith, A.E., J.W. Chapman, and B.R. Dumbauld. 2008. Population structure and energetics of the bopyrid isopod parasite Orthione griffenis in mud shrimp Upogebia pugettensis. Journal of Crustacean Biology 28: 228–233.
Stiven, A.E. 1964. Experimental studies on the host parasite system Hydra and Hydramoeba hydroxena (Entz.). II. The components of a single epidemic. Ecological Monographs 34: 119–142.
Swinbanks, D.D., and J.L. Luternauer. 1987. Burrow distribution of thalassinidean shrimp on a Fraser delta tidal flat, British Columbia. Journal of Paleontology 613: 315–332.
Taylor, C.M., and A. Hastings. 2005. Allee effects in biological invasions. Ecological Letters 8: 895–908.
Torchin, M.E., K.D. Lafferty, and A.M. Kuris. 2001. Release from parasites as natural enemies: Increased performance of a globally introduced marine crab. Biological Invasions 3: 333–345.
Torchin, M.E., K.D. Lafferty, A.P. Dobson, V.J. McKenzie, and A.M. Kuris. 2003. Introduced species and their missing parasites. Nature 421: 628–630.
Tucker, B.W. 1930. On the effects of an epicaridean parasite, Gyge branchialis, on Upogebia littoralis. The Quarterly Journal of Microscopical Science 74: 1–118.
Van Engel, W.A., W.A. Dillon, D. Zwerner, and D. Eldridge. 1965. Loxothylacus panopaei (Cirripedia, Sacculinidae) an introduced parasite on the xanthid crab in Chesapeake Bay, U.S.A. Crustaceana 10: 111–112.
Vermeij, G.J. 1996. An agenda for invasion biology. Biological Conservation 78: 3–9.
Walker, S.P. 1977. Probopyrus pandalicola: Discontinuous ingestion of shrimp hemolymph. Experimental Parasitology 41: 198–205.
Warner, R.E. 1969. The role of introduced diseases in the extinction of the endemic Hawaiian avifauna. Condor 70: 101–120.
Wilcove, D.S., D. Rothstein, J. Dubow, A. Phillips, and E. Losos. 1998. Quantifying threats to imperiled species in the United States. Bioscience 48: 607–615.
Williams, M.R. 1996. Species-area curves: The need to include zeroes. Global Ecology and Biogeography Letters 5: 91–93.
Williams, J.D., and J.M. An. 2009. The cryptogenic parasitic isopod Orthione griffenis Markham, 2004 from the eastern and western Pacific. Integrative and Comparative Biology 49: 114–126.
Windsor, D.A. 1998. Most of the species on earth are parasites. International Journal for Parasitology 28: 1939–1941.
Woods, L., J.W. Chapman, and B.R. Dumbauld. 2006. The effects of a blood-sucking parasite from castration to feminization. REU Summer Reports, 1–11. Newport: Oregon State University, Hatfield Marine Science Center.
Acknowledgments
This research was funded in part by the US Department of Agriculture, Agricultural Research Service, the Washington State Legislature via a grant to JWC to examine biological control of shrimp, a grant to AMK from the USFWS through the Coastal Resources Alliance, funding from NSF for the REU program at Hatfield Marine Science Center, and in-kind field assistance from the LCCSC-OYCC field crews. Historical monitoring was funded by the Washington Department of Fish and Wildlife and several grants to David Armstrong and BRD from the Western Regional Aquaculture Center. We especially thank Lee McCoy and Cara Fritz for their help in the field and with data analysis. Katelyn Cassidy, Roy Hildenbrand, Roxanna Hintzman, Kristine Feldman, Bruce Kauffman, Michael Herrle, Andy Smith, Lauren Woods, Caitlin White, Xeronimo Castaneda, Erin Ashley, Sara Heerhartz, and numerous others assisted with shrimp sampling. Finally, we thank Melanie Frazier, Vladlina Gerteseva, and two anonymous reviewers for their comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 26174 kb)
Rights and permissions
About this article
Cite this article
Dumbauld, B.R., Chapman, J.W., Torchin, M.E. et al. Is the Collapse of Mud Shrimp (Upogebia pugettensis) Populations Along the Pacific Coast of North America Caused by Outbreaks of a Previously Unknown Bopyrid Isopod Parasite (Orthione griffenis)?. Estuaries and Coasts 34, 336–350 (2011). https://doi.org/10.1007/s12237-010-9316-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-010-9316-z