Abstract
We examined the rhizosphere structure of 14 seagrass meadows (seven mixed, three Enhalus acoroides, two Zostera japonica, one Thalassia hemprichii, and one Halophila ovalis) in the Philippines and Vietnam and tested their effect on sediment redox potential by comparing the redox potential in vegetated vs unvegetated sediments. The effect of seagrass photosynthesis on sediment redox potential was tested in an E. acoroides meadow during a short-term (2-day) clipping experiment. In all the meadows, the centroidal depth (i.e., depth comprising 50%) of seagrass belowground biomass was within the top 15 cm sediment layer. Redox potentials in vegetated sediments tended to be higher than those in adjacent unvegetated ones; sediment redox potential anomaly ranged from −61 to 133 mV across the meadows. The centroidal depths of positive redox potential anomaly and seagrass root biomass were significantly correlated across the meadows investigated (type II regression analysis, slope = 0.90, lower confidence limit [CL] = 0.42 upper CL = 1.82, R 2 = 0.59, p < 0.01). Experimental removal of E. acoroides leaves resulted in a decrease in rhizosphere redox potential by 20 mV, further confirming the positive effect of seagrass roots and rhizomes on sediment redox potential and, thus, the general conditions for microbial processes in the coastal zone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Seagrasses modify the environment where their meadows develop and, thus, act as engineering species (sensu Jones et al. 1997). Seagrass canopies reduce water velocity and turbulence (Koch et al. 2006), prevent sediment resuspension (Gacia and Duarte 2001; Terrados and Duarte 1999), and actively trap suspended particles (Agawin and Duarte 2002; Hendriks et al. 2007), thus increasing the particulate input to the underlying sediments (Gacia et al. 2002). These inputs greatly stimulate bacterial activity that is enhanced in seagrass sediments (Duarte et al. 2005). High bacterial activity in sediments leads to the development of anoxic conditions and metabolic pathways that yield products toxic to seagrasses, particularly sulfides (Terrados et al. 1999). Hence, enhanced organic inputs to seagrass sediments, which are particularly high in lush meadows like those found in SE Asia, may be detrimental to seagrass growth and even lead to their decline (e.g., Robble et al. 1991; Calleja et al. 2007; Díaz-Almela et al. 2008).
Seagrasses, however, are able to directly affect the metabolic conditions in the sediments through the release of oxygen by their roots (Borum et al. 2006), i.e., photosynthetically produced during the day and passively diffused from the water column through the plant tissues to the sediment at night (Borum et al. 2006). Oxygen released by seagrass roots may prevent the development of anoxic conditions and exposure of the seagrass rhizospheres to toxic metabolites. Indeed, shading experiments have demonstrated that Thalassia testudinum activity increases sediment redox potential in colonized sediments(Enríquez et al. 2001), and seasonal redox anomalies in a Mediterranean Cymodocea nodosa meadow have been shown to correlate with the seasonal development of the rhizosphere (Marbà and Duarte 2001). The capacity of seagrasses to affect the sediment redox conditions may vary greatly across the seagrass flora because the leaf size (Duarte 1991) and the complexity of the rhizosphere structure (Duarte et al. 1998) differ greatly across seagrass species. This suggests that the capacity of seagrass meadows to affect sediment conditions may be species dependent and may result from interspecific variation during meadow development (e.g., Barrón et al. 2004; Marbà and Duarte 2001).
The effects of seagrasses on the sediments colonized may be more variable in regions with a diverse flora, such as the SE Asian coastal areas, where highly diverse mixed meadows and monospecific meadows of some species develop. The release of significant amounts of oxygen by roots of SE Asia seagrass species has been demonstrated for Cymodocea rotundata (Pedersen et al. 1998), but the effect of these species on sediment redox potential has not been tested yet. In this study, we examine the size and depth distribution of the roots and rhizomes of SE Asian seagrass species and their effect on sediment redox potential. We achieved this goal by examining sediment profiles of root and rhizome biomass across 14 seagrass meadows (seven mixed, three Enhalus acoroides, two Zostera japonica, one Thalassia hemprichii, and one Halophila ovalis) in The Philippines and Vietnam and comparing the vertical distribution of replicate rhizosphere redox potential with profiles of redox potential in adjacent bare sediments. In addition, we experimentally tested the role of seagrass photosynthesis in affecting sediment redox potentials by examining the short-term changes in sediment redox potential in sediments colonized by E. acoroides following clipping of the shoots.
Materials and Methods
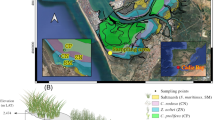
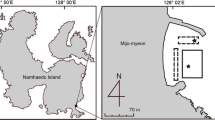
The study was conducted in 14 seagrass meadows distributed across three tropical (Bolinao area, NW Philippines; Palawan Island, the Philippines; Nha Trang, Vietnam) and a temperate (Ha Long Bay, Vietnam) SE Asian regions (Fig. 1). Most of the tropical seagrass meadows in the Bolinao area (16° N 119° E, Panganisan, The Philippines), Ulugan Bay (10° N 118° E, Palawan Island, The Philippines), and Nha Trang (12° N 109° E, Central Vietnam) are mixed, composed of E. acoroides, T. hemprichii, C. rotundata, Cymodocea serrulata, Halodule uninervis, and Syringodium isoetifolium (Table 1). There were four monospecific meadows of either E. acoroides or T. hemprichii (Table 1). In contrast, the meadows studied in Ha Long Bay (20° N 106° E, North Vietnam) are all monospecific meadows of either Z. japonica or H. ovalis (Table 1). The meadows in the Bolinao area are distributed along a siltation gradient from the heavily silted mouth of Alaminos River to the pristine reef lagoon of Santiago Island (Gacia et al. 2003). The meadow located in Santa Barbara, Bolinao, received the highest sediment deposition rate (Table 2) and had the most organic rich sediments (Table 2), whereas total sediment and organic matter deposition rates were lowest at Silaqui (Table 2). Ulugan Bay received water and sediment inputs from ten river watersheds (Padilla, personal communication). Seagrass meadows fringing the mangrove forests that bordered Ulugan Bay received high siltation rates (Table 2), particularly at Umalagan where they grow under turbid water conditions. Two of the meadows studied in the Nha Trang area (My Giang 1 and 2) were exposed to high silt and organic deposition rates from a quarry and activities in a nearby port (Table 2). A third study site in the Nha Trang area (Bay Tien) was continually exposed to intense boat traffic and sewage flow and received a very high sediment and organic loading (Table 2). In contrast, the Dam Gia site is a pristine bay with the lowest sediment deposition rates recorded in the Nha Trang area (Table 2). The meadows studied in Ha Long Bay area are located north of Cat Ba Island, inside a relatively small undisturbed bay with turbid waters (Secchi depth between 0.5 and 1.75 m, Table 2). The meadows are surrounded by mangrove forests (Tu Thi Lan Huong et al. 2003) with a tidal amplitude ranging between 25 and 375 cm annually. Unfortunately, data on sediment deposition rates in the seagrass meadows at Cat Ba Island are not available. The sediment type at the study sites ranged from coarse carbonate to silt (Table 2; Terrados et al. 1998; Lars Kamp-Nielsen, unpublished data). The study was performed during the dry season, when water transparency and photosynthetic seagrass activity are highest (Agawin et al. 2001).
At each meadow, ten shoots of each species present were harvested, the number of seagrass species was counted, and shoot density of extant species was measured in five to 75 (depending on heterogeneity of species distribution) replicates of 50 × 50 cm quadrats for E. acoroides or 30 × 30 cm quadrats for the rest of the species. In addition, seagrass shoot density was quantified in three replicate 20 cm diameter cores. Two and three sediment cores were collected in bare and vegetated sand, respectively, at each site using 50 cm long × 7 cm diameter polyvinyl chloride (PVC) cores, longitudinally perforated with 1.2 cm diameter holes at 2 cm intervals. The holes along the PVC cores were covered with duct tape during sampling to avoid leakage of pore water and sediment and oxygen intrusion. The samples collected were used to estimate vertical profile of sediment redox potential within seagrass rhizospheres and adjacent bare sediments, shoot density, aboveground biomass, and root and rhizome biomass depth distribution.
Immediately after sampling, sediment redox potential (Eh) profiles were measured at 2 cm depth intervals on the 7-cm diameter PVC cores collected by inserting a Crison Pt electrode, connected to a portable pH meter (Crison model 507), horizontally into the sediments. The electrode was regularly calibrated with a redox standard solution (Crison 468 mv at 25°C) and cleaned and polished to prevent accumulation of Pt-oxides. Redox measurements were referred to the standard hydrogen electrode (207 mV) as described by APHA (1992). The redox potential anomaly across seagrass rhizospheres was calculated as the difference between the redox potential of vegetated sediments and that of bare ones. Positive Eh anomaly across sediment profiles was calculated using the values of Eh anomaly >0.
After measuring the redox potential, the sediment cores containing seagrass rhizospheres were sliced at 2 cm thick intervals; the sediment debris in each slice were removed, whereas live roots and rhizomes were dried for 24 h at 60°C and weighed, and sediment profiles of root and rhizome biomass were estimated.
Aboveground biomass was estimated as the product of average shoot density and average shoot mass, measured after overnight desiccation at 60°C, of each species present in the meadow. Root and rhizome biomass of seagrass rhizospheres was calculated as the sum of their biomass across sediment depth divided by the area.
The depth distribution of seagrass root biomass, belowground biomass, the total sediment redox potential anomaly, and positive sediment redox potential anomaly were summarized by fitting the model Y = 1 − ß d (Gale and Grigal 1987), where Y is the fractional cumulative (i.e., fraction between 0 and 1) root biomass, belowground biomass, and total and positive redox potential anomaly, respectively, comprised between the sediment surface to depth d (cm) and ß a “fitted parameter” (Gale and Grigal 1987). The equation was fitted using nonparametric regression analysis (Wilkinson 1989). The centroidal depth of root biomass, belowground biomass, and redox potential anomaly was calculated using the model, with the sediment depth comprising 50% of the root biomass, belowground biomass, or total and positive redox potential anomaly. We also calculated, from the fitted equation, the depth comprising 95% of the root biomass belowground biomass and total and positive redox potential anomaly.
We experimentally tested the short-term response of the rhizosphere redox conditions in an E. acoroides stand at Dau Gia Bay (Vietnam), the largest seagrass stand in SE Asia (Duarte 1991), to the removal of photosynthetic tissues of the seagrass. We clipped all E. acoroides shoots within a 1-m2 plot. Two days after clipping and between 10:00 a.m. and 10:30 a.m., we collected two sediment cores within the clipped plot, three sediment cores in bare sediment, and three sediment cores in vegetated sediments with undisturbed plants using 50 cm long × 7 cm diameter PVC cores, longitudinally perforated at 2 cm intervals with 1.2 cm diameter holes as described above. Sediment redox potential in the sampled cores was measured following the procedures previously described. Sediment redox potential anomalies for intact and clipped vegetation were calculated as described above.
We assessed the statistical significance of the variability observed in seagrass belowground biomass structure across sites and across species with one-way analysis of variance (ANOVA). The significance of differences between sites or species was tested using Tukey’s post hoc test. The significance of the variability in vertical profiles of seagrass belowground biomass across sites was tested using two-way ANOVA.
Results
The seagrass meadows studied ranged from monospecific meadows of a small seagrass species (e.g., H. ovalis) to mixed meadows containing up to six seagrass species (Table 1), with total seagrass biomass ranging two orders of magnitude from 17.9 g DW m−2 of H. ovalis meadow at Cat Ba 1 to 1756 g DW m−2 of a mixed meadow at My Gian 2 (Table 1). On average, root and rhizome biomass was 19-fold greater than leaf biomass (Table 1). Root and rhizome biomass significantly varied across sites (one-way ANOVA root biomass, p < 0.05; one-way ANOVA rhizome biomass, p < 0.05), the meadows at Dau Gia Bay, My Giang 1 and 2, and Silaqui supporting significantly larger root biomass than the rest (Tukey test, p < 0.05). Root and rhizome biomass also varied significantly across species (one-way ANOVA root biomass, p < 0.05; one-way ANOVA rhizome biomass, p < 0.05) and mixed and E. acoroides meadows tending to support the largest rhizospheres. Seagrass roots and rhizomes extended to variable depths into the sediments (Figs. 2 and 3). Belowground biomass significantly varied across sediment depth (two-way ANOVA depth × site, p < 0.0005), with rhizome biomass exhibiting a significantly (Wilcoxon test, p < 0.01) greater vertical variability into the sediment (range of coefficient of variance (CV) vertical rhizome biomass, 94–387%) than root biomass (range of CV vertical root biomass, 58–286%; Fig. 2). Half of the seagrass belowground biomass comprised the top 15 cm of the sediment (Figs. 2 and 3, Table 3), whereas 95% of the belowground biomass comprised the top 56 cm (Figs. 2 and 3, Table 3). The shallowest rhizosphere development was observed in meadows of the small H. ovalis and Z. japonica species (Fig. 3, Table 3), of which 95% of the root and rhizome biomass comprised the top 15 cm of the sediment. In contrast, the deepest rhizosphere penetration was that of a T. hemprichii meadow in By Tien (Fig. 2, Table 3). Overall, the rhizospheres of monospecific meadows of T. hemprichii and E. acoroides and the mixed meadow at My Gian 2 significantly penetrated deeper into the sediments than those of monospecific beds of smaller species and the rest of the mixed meadows (Tukey test, p < 0.05).
Sediment vertical profiles of root (open circles) and rhizome (filled squares) biomass distribution at the mixed seagrass, Enhalus acoroides and Thalassia hemprichii, meadows of SE Asia investigated. Error bars indicate the standard error of the mean. n = 3 for all profiles with error bars but Lucero (n = 4), n = 2 for profiles without error bars
Sediment vertical profiles of root biomass distribution (upper panels) and sediment vertical profiles of redox potential (mV) in bare (filled circles) and vegetated (open squares) sediments (lower panels) at the SE Asian seagrass meadows of Halophila ovalis and Zostera japonica. Error bars of root biomass profiles indicate the standard error of the mean (n = 3), and error bars of redox potential indicate the standard deviation (n = 2 bare sediments, n = 3 vegetated sediments)
Sediment redox potentials ranged from positive values to negative ones, both within and across meadows, with a tendency for redox potential to decline with depth (Figs. 3 and 4). The sediment redox potential showed a variable vertical profile both in bare and vegetated sediments. Redox potentials under vegetated sediments tended to be higher than those in adjacent unvegetated sediments (Figs. 3 and 4, Table 4), although an inverse pattern was observed in three out of the 11 meadows studied. The redox anomaly associated with vegetated sediments (i.e., the average difference between redox potential in vegetated and unvegetated sediments) ranged from a negative value (i.e., average redox potential in unvegetated sediments exceeding that of vegetated ones) of −60.6 mV to a maximum of 129.3 mV but was, on average, positive across the meadows examined (Table 4). The average redox potential anomaly was positive for all E. acoroides and H. ovalis meadows (Table 4) but did not show a consistent trend for mixed meadows and monospecific beds of Z. japonica (Table 4). The centroidal depth of the positive redox anomaly was relatively shallow, ranging between 0 and 15 cm across the meadows studied (Table 3), and was significantly correlated and similar in magnitude to the centroidal depth of the root biomass across meadows (type II regression analysis, slope = 0.90, lower CL = 0.42, upper CL = 1.82, R 2 = 0.59, p < 0.01; Fig. 5).
Sediment vertical profiles of redox potential (mV) in bare (filled circles) and vegetated (open squares) sediments at the mixed seagrass, Enhalus acoroides and Thalassia hemprichii, meadows of SE Asia investigated. Error bars indicate the standard deviation (n = 2 bare sediments, n = 3 vegetated sediments)
The experimental removal of E. acoroides leaves led to a major change in the vertical redox potential profile within 2 days, with an overall reduction in redox potential throughout the rhizosphere, shifting from positive to negative values between 8 and 18 cm into the sediment (Fig. 6). As a result of the removal of the photosynthetic tissues, the mean redox potential anomaly was reduced to half, from +133 to +69 mV (Fig. 6), and the vertical profile resembled more closely that of the bare sediments, particularly in the top 20 cm layer of the rhizosphere, than that of the undisturbed vegetated sediments (Fig. 6).
Sediment vertical profiles of a root (open circles) and rhizome (filled squares) biomass and b redox potential in bare sediments (filled circles), in rhizosphere sediments of entire Enhalus acoroides shoots (open squares), and in rhizosphere sediments of clipped E. acoroides shoots (filled triangles) at the end of the clipping experiment conducted at Dau Gia Bay, Vietnam. The duration of the clipping experiment was 2 days. Error bars of root and rhizome biomass indicate the standard error of mean (n = 3), and error bars of redox potential indicate the standard deviation (n = 3 bare and vegetated sediments, n = 2 sediments with clipped vegetation)
Discussion
The results presented show that seagrass rhizospheres can vary greatly in size, from low root and rhizome biomass extending only a few cm into the sediment, to belowground biomass in excess of 1 kg DW m−2 extending deeper than 70 cm into the sediments. The differences reflect the large variation in the species composition of the meadow, ranging from small species such as H. ovalis and Z. japonica, with leaf canopies just a few cm over the sediment surface to large ones, like E. acoroides, with leaf canopies higher than 1 m (Duarte 1991). The seagrass rhizospheres studied extended across sediment depths encompassing most of the range described for the seagrass flora (Duarte et al. 1998; Enríquez et al. 2001; Marbà and Duarte 2001). However, there was considerable variability in rhizosphere structure across meadows of any one species, depending on the stage of meadow development. Indeed, previous studies clearly showed that redox potentials change during the colonization of the sediments by the seagrass rhizosphere (Marbà and Duarte 2001).
Our results showed a general tendency for seagrass rhizospheres to maintain higher redox potentials relative to unvegetated sediments, as observed in earlier studies (e.g., Enríquez et al. 2001; Marbà and Duarte 2001). However, this was not always the case in the meadows examined; some meadows showed little effect on redox potentials. It seems unvegetated sediments represent a poor control for the effects of seagrasses on redox potentials, for in addition to the absence of seagrasses, the organic carbon inputs to unvegetated sediments and the associated microbial metabolism are likely to be lower than those of seagrass sediments (Barrón et al. 2004; Gacia et al. 2002). Hence, the redox anomalies revealed in this study provide conservative estimates of the effects of seagrasses on redox potentials.
The short-term manipulative experiment that was conducted further showed the importance of seagrasses influence on sediment redox conditions. The removal of the photosynthetic tissues of the plants led to a reduction in the positive sediment redox anomaly by half within 2 days, with the redox profile in the top 20 cm of the sediment reflecting the pattern observed in unvegetated sediments. However, removal of E. acoroides canopy did not change the redox potential in deeper rhizosphere when compared to that in the deep rhizosphere of intact plants (Fig. 6). The lack of response of deep rhizosphere redox potential to the removal of photosynthetic tissues is probably due to the short duration of the experiment. Two days of experimental shading of a T. testudinum meadow decreased the redox potential within the top 25 cm, but not deeper, whereas a decrease in redox potential was detected along the entire rhizosphere (38 cm) after 10 days of shading (Enríquez et al. 2001). Hence, our results are consistent with results from previous manipulative experiments that showed a rapid reduction in sediment redox conditions with increased shading (Enríquez et al. 2001; Gacia et al. 2005) and, thus, provide evidence that seagrass photosynthetic activity influences sediment redox potential. These results are consistent with the view that seagrass rhizospheres affect redox potentials through the release of photosynthetic oxygen through the roots (Borum et al. 2006).
Our results show that redox potential in bare sediments adjacent to seagrass beds often range between −150 and −50 mV (Figs. 3 and 4, Table 4), suggesting that sulfate reduction is the main process for degradation of organic matter in these sediments (Boudreau and Jørgensen 2001; Schüring et al. 2000). There is evidence that sediment sulfides and anoxic conditions exacerbate the decline of seagrass meadows (Carlson et al. 1994; Calleja et al. 2007; Robble et al. 1991). Plant activity tends to increase redox potential in seagrass rhizosperes by 30 mV on average, although in some meadows it increased by more than 100 mV (Table 4). Hence, seagrass activity contributes to maintain sediment redox conditions above the Eh threshold for sulfate reduction to be the dominant redox reaction, and thus, it helps to improve the environmental conditions for seagrass survival by preventing to some extent anoxic conditions and exposure of the plants to sulfides (e.g., Terrados et al. 1999).
The results obtained also show that the depth distribution of the rhizospheres is consistent with the depth distribution of seagrass effects on redox potentials, as suggested in the past (Marbà and Duarte 2001). This observation suggests that the effects of seagrass on sediment microbial activity are dependent on the extent of the rhizosphere into the sediment and may affect microbial activity deep down into the sediments (>50 cm), with potentially important consequences for microbial metabolism and carbon cycling (Duarte et al. 2005).
In summary, our results confirm the role of seagrasses in affecting the redox conditions of coastal sediments and, therefore, the general conditions for microbial processes and metabolic fluxes in the coastal zone. These effects, which result from the photosynthetic activity of the plants and subsequent oxygen transport and release into the rhizosphere, are dependent on the size and extent of the rhizosphere, which are species-specific traits, and suggest that this role of seagrasses is strongly dependent on the species composition of the meadows.
References
Agawin, N.S.R., and C.M. Duarte. 2002. Evidence of direct particle trapping by a tropical seagrass meadow. Estuaries 25: 1205–209.
Agawin, N.S.R., C.M. Duarte, M.D. Fortes, J.S. Uri, and J.E. Vermaat. 2001. Temporal changes in the abundance, leaf growth and photosynthesis of three co-occurring Philippine seagrasses. Journal of Experimental Marine Biology and Ecology. 260: 217–239.
APHA American Public Health Association. 1992. Oxidation-reduction potential measurement in clean water. In Standard methods for the examination of waterand wastewater. 18th edn. APHA, Washington, DC, 260–263.
Barrón, C., N. Marbà, J. Terrados, H. Kennedy, and C.M. Duarte. 2004. Community metabolism and carbon budget along a gradient of seagrass (Cymodocea nodosa) colonization. Limnology and Oceanography. 49: 1642–1651.
Borum, J., K. Sand-Jensen, T. Binzer, O. Pedersen, and T.M. Greve. 2006. Oxygen movement in seagrasses. In Seagrasses: biology, ecology and conservation, ed. A.W.D. Larkum, R.J. Orth, and C.M. Duarte, 225–270. Dordrecht: Springer.
Boudreau, B.P., and B.B. Jørgensen. 2001. The benthic boundary layer. Transport processes and biogeohemistry. New York: Oxford University Press.
Calleja, M., N. Marbà, and C.M. Duarte. 2007. The relationship between seagrass (Posidonia oceanica) decline and porewater sulfide pools in carbonate sediments. Estuarine, Coastal and Shelf Science 73: 583–588.
Carlson Jr., P.R., L.A. Yarbro Jr., and T.R. Barber. 1994. Relationship of sediment sulfide to mortality of Thalassia testudinum in Florida Bay. Bulletin of Marine Science 54: 733–746.
Díaz-Almela, E., N. Marbà, E. Álvarez, R. Santiago, M. Holmer, A. Grau, S. Mirto, R. Danovaro, A. Petrou, M. Argyrou, I. Karakassis, and C.M. Duarte. 2008. Benthic input rates predict seagrass (Posidonia oceanica) fish farm-induced decline. Marine Pollution Bulletin 56: 1332–1342.
Duarte, C.M. 1991. Allometric scaling of seagrass form and productivity. Marine Ecolog Progress Series 77: 289–300.
Duarte, C.M., M. Merino, N.S.R. Agawin, J. Uri, M.D. Fortes, M.E. Gallegos, N. Marbà, and M. Hemminga. 1998. Root production and belowground seagrass biomass. Marine Ecology Progress Series 171: 97–108.
Duarte, C.M., M. Holmer, and N. Marbà. 2005. Plant microbe-interactions in seagrass meadows. In Macro-and Microorganisms in Marine Sediments, ed. E. Kristensen, R. Haese, and J. Kotska, 31-60 Coastal and Estuarine Studies, 60, Washington, American Geophysical Union.
Enríquez, S., N. Marbà, C.M. Duarte, B. van Tussenbroek, and G. Reyes-Zavala. 2001. Effects of seagrass (Thalassia testudinum) on sediment redox conditions. Marine Ecology Progress Series 219: 149–158.
Gacia, E., and C.M. Duarte. 2001. Sediment retention by a Mediterranean Posidonia oceanica meadow: The balance between deposition and resuspension. Estuarine, Coastal and Self Science 52: 505–514.
Gacia, E., C.M. Duarte, and J.J. Middelburg. 2002. Carbon and nutrient deposition in a Mediterranean seagrass (Posidonia oceanica) meadow. Limnology and Oceanography 47: 23–32.
Gacia, E., C.M. Duarte, N. Marbà, J. Terrados, H. Kennedy, M.D. Fortes, and T.N. Huang. 2003. Sediment deposition and production in SE-Asia seagrasses meadows. Estuarine, Coastal and Self Science 56: 1–11.
Gacia, E., H. Kennedy, C.M. Duarte, J. Terrados, N. Marbà, S. Papadimitriou, and M. Fortes. 2005. Light-dependence of the metabolic balance of a highly productive Philippine seagrass community. Journal of Experimental Marine Biology and Ecology 316: 55–67.
Gale, M.R., and D.K. Grigal. 1987. Vertical root distributions of northern tree species in relation to successional status. Canadian Journal of Forest Research 17: 829–834.
Hendriks, I.E., T. Sintes, T. Bouma, and C.M. Duarte. 2007. Experimental assessment and modeling evaluation of the effects of seagrass (P. oceanica) on flow and particle trapping. Marine Ecology Progress Series 356: 163–173.
Jones, C.G., J.H. Lawton, and M. Shachak. 1997. Positive and negative effects of organisms as physical ecosystem engineers. Ecology 78: 1946–1957
Koch, E.W., J.D. Ackerman, J. Verduin, and M. van Keulen. 2006. Fluid Dynamics in Seagrass Ecology—from Molecules to Ecosystems. In Seagrasses: biology, ecology and conservation, ed. A.W.D. Larkum, R.J. Orth, and C.M. Duarte, 193–225. Dordrecht: Springer.
Marbà, N., and C.M. Duarte. 2001. Growth and sediment space ocupation by seagrass (Cymodocea nodosa) roots. Marine Ecology Progress Series 224: 291–298.
Pedersen, O., J. Borum, C.M. Duarte, and M.D. Fortes. 1998. Oxygen dynamics in the rhizosphere of Cymodocea rotundata. Marine Ecology Progress Series 169: 283–288.
Robble, M.B., T.R. Barber, P.R. Carlson, M.J. Durako, J.W. Fouqurean, L.K. Muehlstein, D. Porter, L.A. Yarbro, R.T. Zieman, and J.C. Zieman. 1991. Mass mortality of the tropical seagrass Thalassia testudinum in Florida Bay (USA). Marine Ecology Progress Series 71: 297–299.
Schüring, J., H.D. Schulz, W.R. Fisher, J. Böttcher, and W.H.M. Duijnisveld. 2000. Redox. Fundamentals, processes and applications. Berlin: Springer.
Terrados, J., and C.M. Duarte. 1999. Experimental evidence of reduced particle resuspension within a seagrass (Posidonia oceanica L.) meadow. Journal of Experimental Marine Biology and Ecology 243: 45–53.
Terrados, J., C.M. Duarte, M.D. Fortes, J. Borum, N.S.R. Agawin, S. Bach, U. Thampanya, L. Kamp-Nielsen, W.J. Kenworthy, O. Geertz-Hansen, and J. Vermaat. 1998. Changes in community structure and biomass of seagrass communities along gradients of siltation in SE Asia. Estuarine, Coastal and Shelf Science 46: 757–768.
Terrados, J., C.M. Duarte, L. Kamp-Nielsen, N.R.S. Agawin, E. Gacia, C.D.A. Lacap, M.D. Fortes, J. Borum, M. Lubanski, and T. Greve. 1999. Are seagrass growth and survival constrained by the reducing conditions of the sediment? Aquatic Botany 65: 175–197.
Tu Thi Lan Huong, J.E., J.T. Vermaat, N. Van Tien, C.M. Duarte, J. Borum, and N.H. Tri. 2003. Seasonality and depth zonation of intertidal Halophila ovalis and Zostera japonica in Ha Long Bay (Northern Viet Nam). Aquatic Botany 75: 147–157.
Wilkinson, L. 1989. SYSTAT: the system for statistics. Evanston: SYSTAT, Inc.
Acknowledgements
This work was funded by the European project PREDICT (ERB3514PL972504, INCO Programme). We thank Lars Kamp-Nielsen for providing information on sediment type for the studied sites.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marbà, N., Duarte, C.M., Terrados, J. et al. Effects of Seagrass Rhizospheres on Sediment Redox Conditions in SE Asian Coastal Ecosystems. Estuaries and Coasts 33, 107–117 (2010). https://doi.org/10.1007/s12237-009-9250-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-009-9250-0