Abstract
This research was carried out to study the levels of bacterial wilt resistance and genetic diversity of (near)pentaploid sexual hybrids between S. commersonii (2n = 2x = 24, 1EBN) and cultivated S. tuberosum. Following artificial inoculations with Ralstonia solanacearum, wilting degree was estimated on a scale from 0 to 4, and seven genotypes of 26 (27%) displaying a S. commersonii like behavior were identified. Latent bacterial colonizations were detected in roots of symptomless S. commersonii and hybrids, whereas no bacterial populations were detected within stems. This suggests that the movement and/or growth of the bacterium in the aerial part were strongly inhibited. A molecular study with AFLP markers clustered hybrids into nine groups and provided evidence that resistant hybrids were slightly more similar to cultivated S. tuberosum than to the wild parent. This is important in view of the re-establishment of the cultivated genetic background through backcrosses. Hybrids displayed good fertility and are being used for further breeding efforts.
Resumen
Esta investigación fue realizada para estudiar los niveles de resistencia a la marchitez bacteriana y la diversidad genética de híbridos sexuales (casi) pentaploides entre S. commersoni y S. tuberosum cultivado. Después de las inoculaciones artificiales con Ralstonia solanacearum, la marchitez fue estimada en una escala de 0 a 4 y se identificaron siete genotipos de 26 (27%) que se comportaron como S. commersoni. Se detectaron colonizaciones bacterianas latentes en raíces que no presentaban síntomas de S. commersoni e híbridos mientras que no se detectó población bacteriana dentro de los tallos. Esto sugiere que el movimiento y/o crecimiento de la bacteria en la parte aérea fue fuertemente inhibido. Un estudio molecular con marcadores AFLP agrupó los híbridos en nueve grupos y dio la evidencia de que los híbridos fueron ligeramente más similares al S. tuberosum cultivado que sus progenitores silvestres. Esto es importante en vista del restablecimiento de los antecedentes genéticos de la especie cultivada por medio de retro cruzamientos. Los híbridos mostraron buena fertilidad y están siendo usados para mejoramiento.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacterial wilt caused by Ralstonia solanacearum is a major disease affecting many crops in tropical, sub-tropical and some temperate areas worldwide (Hayward 1994). This vascular bacterium invades roots, and causes partial or complete wilting of the host plants as a consequence of colonization and multiplication. Traditionally, R. solanacearum has been grouped into five races based on host range. Recently, molecular studies provided evidence that it may be regarded as a species complex with four phylotypes related to the geographical origin of the strain (Fegan and Prior 2004). Among economically important hosts, the potato (Solanum tuberosum, 2n = 4x = 48, 4EBN) ranks first in terms of importance worldwide. Only Race 1 and Race 3 cause severe crop failure in potato, the former in tropical and sub-tropical areas, the latter in cooler regions. Recently, the disease has been detected in Western Europe, where international plant quarantine regulations have been toughened to restrict its spread (Zimnoch-Guzowska et al. 2006). Since chemical control is not effective, other preventive methods are difficult to apply, and the level of resistance in cultivated varieties is low, the development of genotypes resistant to R. solanacearum has become a major challenge for potato breeders. A number of tuber-bearing solanums possessing resistance to R. solanacearum has been identified and used in breeding programs. Among them, S. commersonii (2n = 2x = 24, 1EBN) cannot be directly crossed with S. tuberosum due the endosperm balance number incompatibility system (Johnston et al. 1980). To transfer its R. solanacearum resistance to the cultivated potato, breeders have extensively used somatic hybridization (Laferriere et al. 1999; Kim-Lee et al. 2005). Ploidy manipulations through sexual hybridization represent a possible alternative to overcome sexual barriers. Indeed, we doubled the chromosome number of S. commersonii and crossed a 4x clone of this species with 2x genotypes to generate a triploid progeny (Carputo et al. 1997). Resulting triploids that produced 2n eggs were used in 3x × 4x crosses with S. tuberosum and generated an offspring with a (near)pentaploid chromosome number.
The aims of the present research were to study the level of resistance to R. solanacearum race 3, biovar 2 (r3 bv2) of (near)pentaploid S. commersonii–S. tuberosum sexual hybrids and to assess their level of genetic diversity.
Materials and Methods
Plant Material
Twenty-six hybrids (hereafter coded MCPH) produced from 3x × 4x crosses between triploid hybrid MCA1 and Blondy were used in this study. Their somatic chromosome number, ranging from 54 to 67, was previously reported (Carputo 2003). All clones were maintained and multiplied in tissue culture on MS salts (Murashige and Skoog 1962), sucrose 10 g/L and agar 8 g/L.
Disease Resistance Assay
Ralstonia solanacearum (SA 93) r3 bv2, isolated from potato and kindly provided by Dr Nico J.J. Mienie from ARC—Roode Plaat, Pretoria (South Africa), was used for resistance assay. Strain SA 93 was maintained in a sterile distilled water (SDW) stock culture and grown for 24 h at 28°C on YDC medium (Denny and Hayward 2001) when needed. Bacterial suspensions in SDW containing 5 × 107 cfu mL−1 were used for plant inoculations. We tested MCPH hybrids, S. commersonii, and Blondy. Included in the assay were also 2x UP88-P5 and 3x MCA1, parental genotypes of the triploid and (near)pentaploid generation, respectively. One-month-old vitro plants of each hybrid were transplanted into 60 mm diameter pots containing sterile soil in a temperature-controlled greenhouse. When a six to eight leaflet stage was reached, nine to 12 uniform plants for each genotype were inoculated pouring 1 mL of suspension into 1 cm deep holes previously made in the soil (Montanelli et al. 1995). Three control plants were treated with SDW. Test plants were randomly distributed and grown in a climatic cell at 80% RU, with 27°C–14 h day and 21°C–10 h night. The whole experiment was performed twice.
The wilting symptoms of each plant were individually recorded at 6, 9, 12, 15, 17, and 20 days after inoculation as percentage of wilted leaves. Resistance to R. solanacearum was quantified by a wilting degree (WD) in leaves ranging from 0 to 4 (Fock et al. 2001): 0 = no wilted leaves, 1 = up to 25% wilted, 2 = 26% to 50% wilted, 3 = 51% to 75% wilted, 4 = more than 75% wilted. For each genotype the average WD at 6, 9, 12, 15, 17 and 20 days after inoculation was calculated. To select superior hybrids for further breeding, we considered resistant genotypes that at 20 days after inoculation showed an average WD ≤ 1.
To estimate the population of R. solanacearum in roots and stems, three plants each of Blondy, S. commersonii and resistant hybrids MCPH27 and MCPH31 were randomly uprooted and accurately washed to remove all soil particles at 12 days after inoculation (6 days for Blondy). After plants were defoliated, roots and stems of each plant were separated. The crown area of about 6–8 mm was eliminated. Roots and stems of each plant were individually surface treated by soaking into NaClO 0.5% for 3 min and then washed three times in SDW. Roots and stems were blotted dry, weighed, put in a mortar with nine parts (weight/volume) of SDW and extensively homogenized. After 20 min the homogenates were serially diluted and 100 μL of each dilution were spread on SMSA plates (Elphinstone et al. 1996) in triplicate. Plates were incubated at 28°C and the colonies enumerated after 3 days. The lower limit for bacterial recovery was about 1.0 × 102 cfu/g fresh medium.
Molecular Analysis
AFLP analysis was performed using the method described by Vos et al. (1995) and the commercially available AFLP kit and protocol (Gibco-BRL AFLP analysis System I, Life Technologies, Gaithersburg, MD, USA), which employs EcoRI and MseI as restriction enzymes. Five primer combinations were used (Eco-AGG/Mse-CAA; Eco-ACC/Mse-CAT; Eco-ACT/Mse-CAC; Eco-ACT/Mse-CAT; Eco-AGG/Mse-CAG). A fluorometric method was used for detecting AFLP fragments. EcoRI primers were labelled with 6-carboxy-fluorescein (6-FAM), whereas MseI primers were unlabelled. AFLP fragments were electrophoretically separated on 6% denaturing polyacrylamide gels and the fluorescence detected with a Typhoon 9210 scanner (Amersham). AFLP images were then analyzed with ImageQuant TL software, v2003.02 (Amersham Biosciences 2002), and by visual inspection. For each genotype, polymorphic fragments were recorded as present (1) or absent (0). Genetic similarity (GS) between clones was calculated as Jaccard’s similarity coefficients (Jaccard 1908) and clustering was performed using the unweighted pair-group method (UPGMA). NTSYS-pc software was used for genetic similarity analysis and the resulting clusters were represented in the form of a dendrogram.
Data Analysis
Statistical analyses were performed using SPSS software v. 11.5 (Copyright (c) SPSS Inc., 1989–2004. All rights reserved). The repeated measures ANOVA procedure was performed on the WD values recorded at different points of time to test the null effect hypothesis of both genotype and time after inoculation and the interaction of these sources of variability. Included in this analysis were MCPH hybrids, Blondy and S. commersonii. Principal component analysis (PCA) analyzed six variables, each representing the WD value measured at each point of time. To monitor the inheritance pattern of resistance to R. solanacearum from S. commersonii in our hybrids, the degree of dominance (d/a) was calculated according to the formula (Falconer 1989):
d/a is a numerical value to evaluate the tendency for the mean resistance value of MCPH hybrids to deviate from the mid-parent value. The greater the phenotypic dominance, the greater the deviation of d/a from zero, with negative and positive values meaning dominance of the resistant (S. commersonii) and susceptible (S. tuberosum) parent, respectively. Although this formula generally assumes that parents are highly homozygous, it has been used also in Solanum (Culley et al. 2002).
Results
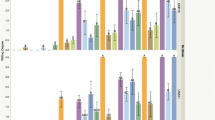
Genotypes, time after inoculation and their interaction were significant sources of variation (Table 1). Hybrids displayed a highly variable response, segregating from resistant to susceptible reactions. Out of 26 hybrids tested, 7 (27%) showed an average WD < 1 and were classified resistant: MCPH0 (2n = 60, WD = 0.0), MCPH6 (2n = 64, WD = 0.5 + 0.2), MCPH27 (2n = 64, WD = 0.2+0.2), MCPH31 (2n = 67, WD = 0.4 + 0.2), MCPH20 (2n = 62, WD = 0.4 + 0.2), MCPH16 (2n = 58, WD = 0.7 + 0.2), MCPH21 (2n = 60, WD = 0.4 + 0.2; data not shown). Figure 1 illustrates the disease progress on S. commersonii, Blondy, and a sample of hybrids over time.
Disease progress on resistant and susceptible S. tuberosum–S. commersonii hybrids caused by Ralstonia solanacearum. Parental genotypes are also included (S. commersonii = CMM; S. tuberosum = Blondy). For each line, the average wilting degree of inoculated plants was calculated at 6, 9, 12, 15, 17 and 20 days after artificial inoculations on a scale of 0 (no wilt) to 4 (76% to 100% leaves wilted). Vertical bars represent standard errors. Results were obtained from two independent experiments
To better visualize the performance of each hybrid, mean values of WD were analysed by PCA and two factors were extracted. Factor 1 explained variability of the WD at 12, 15, 17 and 20 days after inoculation. Factor 2 described variability of the WD at 6 and 9 days after inoculation. Hybrids were plotted against scores of both factor 1 and factor 2 (Fig. 2). Hybrids with a WD < 1 showed a similar disease progress, achieving low to very low scores for factor 1 and average values for factor 2. By contrast, the cultivated parent Blondy and MCPH35 achieved high scores both for factor 2 and for factor 1. Some hybrids (MCPH 29, MCPH 34) displayed average scores for factor 1 and very high scores for factor 2. They were characterized by very early disease symptoms that did not result in plant death at 20 days after inoculation.
Graphical discrimination of 26 hybrids by PCA performed on wilting degree over time. Extracted factors (1 and 2) comprehensively summarize WD progression. Factor 1 describes disease progress at 12, 15, 17 and 20 days after inoculation, whereas factor 2 describes disease progress at 6 and 9 days after inoculation. Hybrids are plotted in a plane against regression factor scores. Circled clones showed an average WD < 1 and were classified resistant. S. commersonii = CMM, S. tuberosum = Blondy. Results were obtained from two independent experiments
As for the estimation of populations of R. solanacearum within roots and stems of selected genotypes, bacterial populations (expressed as logarithms of cfu/g fresh weight) were below the detection threshold within stems of S. commersonii, MCPH27 and MCPH31. By contrast, high concentrations (log 9.87/g fresh weight) were found in stems of Blondy (Table 2). Bacterial populations recovered from roots were higher than log six per gram fresh weight in all genotypes tested. However, values recorded in S. commersonii, MCPH27 and MCPH31 were statistically lower from those recorded in Blondy.
The degree of dominance in 5x MCPH hybrids and in the 3x progeny they derived from was −0.41 and −0.15 (Table 3). Although this result may be biased due to the genotype of parents used, nonetheless it may suggest that phenotype “resistance to R. solanacearum” derived from S. commersonii is dominant over phenotype “susceptibility to R. solanacearum” present in the diploid clone UP88-P5 and in tetraploid S. tuberosum. No significant correlation was found between resistance to R. solanacearum and the hybrid chromosome number (r = 0.02).
GS values ranged from 0.49 (between MCPH20 and MCPH21) to 0.90 (MPH15 and MCPH24; data not shown). GS values of resistant MCPH0, MCPH6, MCPH16, MCPH20, MCPH21, MCPH27 were more similar to S. tuberosum (0.71, 0.62, 0.72, 0.51, 0.76, 0.79, respectively) than to S. commersonii (0.62, 0.60, 0.64, 0.54, 0.72, 0.72, respectively). As for MCPH31, the opposite was true. The UPGMA dendrogram allowed the clustering into nine main groups (Fig. 3). S. tuberosum cv. Blondy and S. commersonii grouped separately in clusters C and F, respectively. C was the largest cluster, involving eight hybrids (31%). AFLP analysis also allowed the detection of 170 S. commersonii-specific bands. MCPH24 and MCPH20 showed the lowest (8) and largest (98) number of such fragments, respectively. The mean value for resistant hybrids was 44.8, that of susceptible ones was 53.1.
Unweighted pair-group method dendrogram of AFLP data of S. tuberosum–S. commersonii hybrids and their parental genotypes (S. commersonii = CMM, S. tuberosum = TBR). The similarity on the x-axis is based on the Jaccard’s coefficient. Five clusters (A to E) were identified, and the number of S. commersonii introgressed bands of each genotype is reported in parenthesis. Resistant genotypes are underlined
Discussion
In potato, resistance to bacterial wilt by R. solanacearum is deemed very important due to the worldwide importance of this crop and lack of resistant varieties. To transfer the resistance of incongruent S. commersonii through sexual hybridization, we developed a ploidy series involving diploid, triploid, tetraploid and (near)pentaploid sexual hybrids. Results from the present study indicated that a number of S. commersonii–S. tuberosum hybrids show a level of resistance indistinguishable from that of S. commersonii. As far as we know, this is the first report on S. commersonii–S. tuberosum hybrids obtained through sexual hybridization that do not show any symptom after artificial inoculation with R. solanacearum. S. commersonii (+) S. tuberosum hybrids carrying R. solanacearum resistance were reported by Laferriere et al. (1999). Their resistance has been recently transmitted to backcross genotypes (Kim-Lee et al. 2005). To better visualize both the performance of each clone and groupings that could not possibly emerge from the raw data, PCA was employed. Based upon the wilting progress during time, our hybrids were separated into three groups. As wilting symptoms at 6 and 9 days after inoculation of genotypes with WD < 1 and several other hybrids were similar (i.e. absent), a differential level of pre-infection resistance is unlikely to account for the overall differences in the disease progress. These two groups may differ due to either earlier activation of resistance mechanisms or a stronger reaction of the plant to the invasion of the pathogen. Blondy exhibited severe symptoms also at earlier stages, and may be characterized by complete lack of resistance genes as well as of negative-acting host factors that contribute to bacterial growth inhibition. Unfortunately, little is known on the plant–pathogen interaction in potato. In tomato, Wydra and Ber (2007) suggested that xylem cell wall structure play a role as constitutive resistance mechanism, while changes after inoculation contribute to induced resistance on cell wall. In Arabidopsis thaliana, Yang and Ho (1998) demonstrated that the ability of plants to affect either growth or movement of the bacterium is a major factor in disease development.
Although plants resistant to R. solanacearum often do not show any symptom, their roots and/or stems may be nevertheless invaded by the bacterium. Assessment of bacterial populations in asymptomatic plants has rarely been done, and genotypes have been usually classified resistant by absence of disease symptoms. In our experiments, bacterial populations were not detected in the stems of symptomless S. commersonii and hybrids tested. Therefore, even though latent colonizations were detected in roots of symptomless plants, S. commersonii and its hybrids can be considered resistant to r3 bv2 of R. solanacearum. This conclusion is based on the results reported by Fock et al. (2001) in S. tuberosum–S. stenotomum somatic hybrids. According to these authors, if bacterial populations within stems remain at a low level, there will be a reduced possibility of further multiplication, and genotypes may be considered resistant. Since no correlation exist between foliage symptoms and tuber infection (Ciampi and Sequeira 1980), inadvertent movement of the pathogen through latently infected tubers is possible. Therefore, research is under way to unravel the details of bacterial wilt resistance mechanisms in S. commersonii and to assess the levels of resistance of hybrids in tubers and under field conditions. Preliminary analyses carried out by both direct isolation and PCR are encouraging, since they did not reveal occurrence of the pathogen in tubers produced by asymptomatic genotypes. The finding of successful root colonization can be an additional problem in the use of these resistant hybrids. Indeed, bacterial transmission from infected roots to other non-infected fields via contaminated irrigation water and field equipment may be possible. A recent paper by Tomlinson et al. (2005) provided evidence that no visible symptoms were observed in potato crops irrigated with water periodically contaminated with R. solanacearum and tests on harvested tubers failed to detect the pathogen. Further studies are necessary to determine how environmental conditions, biological activity, inoculum pressure and other factors may affect populations of the bacterium. Our plant material will be offered to anyone interested in further studies aimed at elucidating these aspects.
Little is known of the genetic control of R. solanacearum resistance in potato (Rowe and Sequeira 1970; Watanabe et al. 1992). Although we performed no in-depth genetic analysis, our study provided some useful information from the genetic and breeding standpoint. Indeed, estimation of the degree of dominance indicated that phenotypic “resistance to R. solanacearum” derived from S. commersonii is dominant over phenotypic “susceptibility to R. solanacearum” typical of S. tuberosum. The AFLP analysis pointed out two aspects. First, out of seven resistant hybrids with WD < 1, six were slightly more similar to cultivated S. tuberosum than to the wild parent. This is important in view of the re-establishment of the cultivated genetic background through backcrosses. Second, hybrids with lower WD were well distributed in the dendrogram, indicating that they may provide not only bacterial wilt resistance/tolerance but also allelic diversity for exploiting heterosis for quantitative traits. Finally, our study highlighted the lack of a significant correlation between hybrid chromosome number and WD. By contrast, in S. tuberosum (+) S. phureja tetraploid hybrids, Fock et al. (2000) found the highest level of resistance in a genotype with a somatic chromosome number higher than 48. The authors hypothesized that extra-chromosomes belonged to the resistant parent, thus giving a gene dosage effect with a higher expression of resistance.
In conclusion, unusual interspecific sexual hybridization resulted in new genetic material potentially valuable for potato breeding. Due to the good fertility of (near)pentaploids (Caruso et al. 2008), there should be no further barrier to the introgression of resistance genes into cultivated varieties. This plant material is also suitable for further studies on the genetics and nature of R. solanacearum resistance of wild S. commersonii. Interestingly, Elphinstone (1994) hypothesized that potato resistance against R. solanacearum is enhanced when genes encoding for both biotic and abiotic stresses are combined together. Since S. commersonii is highly resistant to low temperatures, Erwinia carotovora and viruses, it will be interesting to further investigate this hypothesis in the material developed here.
References
Carputo, D. 2003. Cytological and breeding behavior of pentaploids derived from 3x × 4x crosses in potato. Theoretical and Applied Genetics 106:883–888.
Carputo, D., A. Barone, T. Cardi, A. Sebastiano, L. Frusciante, and S.J. Peloquin. 1997. Endosperm balance number manipulation for direct in vivo germplasm introgression to potato from a sexually isolated relative (Solanum commersonii Dun.). Proceedings of the National Academy of Sciences 94:12013–12017.
Caruso, I., L. Castaldi, G. Caruso, L. Frusciante, and D. Carputo. 2008. Breeding potential of Solanum tuberosum–S. commersonii pentaploid hybrids: fertility studies and tuber evaluation. Euphytica 164:357–363.
Ciampi, L., and L. Sequeira. 1980. Multiplication of Pseudomonas solanacearum in resistant potato plants and the establishment of latent infections. American Potato Journal 57:319–329.
Culley, D.E., B.B. Dean, and C.R. Brown. 2002. Introgression of the low browning trait from the wild Mexican species Solanum hjertingii into cultivated potato (S. tuberosum L.). Euphytica 125:293–303.
Denny, T.P., and A.C. Hayward. 2001. II Gram-negative bacteria, F. Ralstonia. In Laboratory guide for identification of plant pathogenic bacteria, eds. N.W. Schaad, J.B. Jones, and W. Chun, 3rd ed. 151–174. St Paul: APS Press.
Elphinstone, J.G. 1994. Inheritance of resistance to bacterial diseases. In Potato genetics, eds. J.E. Bradshaw and G.R. Mackay, 429–446. Wallingford: CAB International.
Elphinstone, J.G., J. Hennessy, J.K. Wilson, and E. Stead. 1996. Sensitivity of different methods for the detection of Ralstonia solanacearum in potato tuber extracts. Bulletin OEPP/EPPO Bulletin 26:663–678.
Falconer, D.S. 1989. Introduction to quantitative genetics, 3rd Eds. Essex: Longman Scientific and Technical.
Fegan, M., and P. Prior. 2004. How complex is the “Ralstonia solanacearum species complex”. In Bacterial wilt: The disease and Ralstonia solanacearum species complex, eds. C. Allen, P. Prior, and A. C. Hayward, 449–461. St Paul: APS Press.
Fock, I., C. Collonnier, A. Purwito, J. Luisetti, V. Souvannavong, F. Vedel, A. Servaes, A. Ambroise, H. Kodja, G. Ducreux, and D. Sihachakr. 2000. Resistance to bacterial wilt in somatic hybrids between Solanum tuberosum and Solanum phureja. Plant Science 160:165–176.
Fock, I., C. Collonnier, J. Luisetti, A. Purwito, V. Souvannavong, F. Vedel, A. Servaes, A. Ambroise, H. Kodja, G. Ducreux, and D. Sihachakr. 2001. Use of Solanum stenotomum for introduction of resistance to bacterial wilt in somatic hybrids of potato. Plant Physiology & Biochemistry 39:899–908.
Hayward, A.C. 1994. The hosts of Pseudomonas solanacearum. In Bacterial wilt: The disease and its causative agent, Pseudomonas solanacearum, eds. A.C. Hayward and G.L. Hartman, 9–23. Wallingford: CAB International.
Jaccard, P. 1908. Nouvelles recherches sur la distribution florale. Bulletin Société Vaudoise Science Naturelles 44:223–270.
Johnston, S.A., T.M. den Nijs, S.J. Peloquin, and R.E. Hanneman Jr. 1980. The significance of genic balance to endosperm development in interspecific crosses. Theoretical and Applied Genetics 57:5–9.
Kim-Lee, H., J.S. Moon, Y.J. Hong, M.S. Kim, and H.M. Cho. 2005. Bacterial wilt resistance in the progenies of the fusion hybrids between haploid of potato and Solanum commersonii. American Journal of Potato Research 82:129–137.
Laferriere, L.T., J.P. Helgeson, and C. Allen. 1999. Fertile Solanum tuberosum + S. commersonii somatic hybrids as sources of resistance to bacterial wilt caused by Ralstonia solanacearum. Theoretical and Applied Genetics 98:1272–1278.
Montanelli, C., A. Chiari, T. Chiari, F. Stefanini, and G. Nascari. 1995. Evaluation of resistance to Pseudomonas solanacearum in potato under controlled conditions. Euphytica 81:35–43.
Murashige, T., and F. Skoog. 1962. A revised medium for rapid growth and bioassays with tobacco tissue culture. Plant Physiology 15:473–497.
Rowe, P.R., and L. Sequeira. 1970. Inheritance of resistance to Pseudomonas solanacearum in Solanum phureja. Phytopathology 60:1499–1501.
Tomlinson, D.T., J.G. Elphinstone, M.S. Hanafi, T.M. Schoala et al. 2005. Survival of Ralstonia solanacearum biovar 2 in canal water in Egypt. In Potato in progress: Science meets practice, eds. A.J. Haverkort and P.C. Struik, 228–232. Wageningen: Wageningen Academic Publishers.
Vos, P., R. Hogers, M. Bleeker, M. Reijans, V. van de Lee, M. Hornes, A. Frijters, J. Pot, J. Peleman, M. Kuiper, and M. Zabeau. 1995. AFLP: a new technique for DNA fingerprinting. Nucleic Acid Research 23:4407–4414.
Watanabe, J.A., H. Nashaar, and M. Iwanaga. 1992. Transmission of bacterial wilt resistance by first division restitution (FDR) 2n pollen via 4x × 2x crosses in potatoes. Euphytica 60:21–26.
Wydra, K., and H. Ber. 2007. Immunohistochemical changes in methyl-ester distribution of homogalacturonan and side chain composition of rhamnogalacturonan I as possible components of basal resistance in tomato inoculated with Ralstonia solanacearum. Physiological and Molecular Plant Pathology 70:13–24.
Yang, C.H., and G.D. Ho. 1998. Resistance and susceptibility of Arabidopsis thaliana to bacterial wilt caused by Ralstonia solanacearum. Phytopathology 88:330–334.
Zimnoch-Guzowska, E., E. Lojkowska, and M. Pérombelon. 2006. Resistance to bacterial pathogens. In Genetic improvement of Solanaceous crops, Vol. 1:”Potato”, eds. M.K. Razdan and A.K. Mattoo, 339–395. Enfield: Science Publishers.
Acknowledgments
This work is dedicated to the fondest memory of Stan Peloquin, mentor and great friend for our group. We gratefully acknowledge the technical support of S. Alberino, I. Caruso and R. Garramone. This is contribution no. 191 from the Dept. of Soil, Plant, Environmental and Animal Production Sciences. The experiments comply with the current Italian laws.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carputo, D., Aversano, R., Barone, A. et al. Resistance to Ralstonia solanacearum of Sexual Hybrids Between Solanum commersonii and S. tuberosum . Am. J. Pot Res 86, 196–202 (2009). https://doi.org/10.1007/s12230-009-9072-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-009-9072-4