Abstract
The study aimed to apply gamma radiation as a cell pretreatment method for lipid extraction and for obtaining astaxanthin from residual biomass of Haematococcus pluvialis. Factor 1 of the two-factor experimental design was represented by cell pretreatment methods: biomass with chloroform:methanol under ultrasound (BCMU), biomass with chloroform:methanol under γ radiation (BCMR), dry biomass under γ radiation (DBR), and control (without pretreatment). Factor 2 considered the vegetative and cystic phases. Cultivation was performed in a mixotrophic system, and biomass was collected in both phases, centrifuged, lyophilized, and submitted to cell pretreatment and lipid extraction. Lipid content and FAMEs were evaluated comparing pretreatment methods and life cycle phases. Total lipid content was higher with the BCMR method in the vegetative (18% DW) and cystic (14% DW) phases. Gamma radiation combined with organic solvent was more efficient for increasing lipid yield, and DBR had a lipid yield similar to BCMU. FAME content differed between phases and pretreatments for most fatty acids, mainly C16:0, C16:1, C18:1n9c, C18:2n6c, C18:2n6t, and C18:3n3. The predominance of saturated or low unsaturated fatty acids makes H. pluvialis, in both phases, suitable for biofuel production. The preservation of astaxanthin from residual cystic biomass was observed when submitted to the DBR method, with a concentration similar to the raw biomass (1.5% DW). Gamma radiation in dry biomass has an antioxidant effect. Therefore, the lipid extraction method preceded by gamma irradiation was efficient for vegetative and cystic cells of H. pluvialis and contributed to the preservation of astaxanthin from residual cystic biomass.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The microalga Haematococcus pluvialis has attracted great interest over the years due to its cellular content, being commonly cultivated for the extraction of the carotenoid astaxanthin [1]. This carotenoid is widely used in the pharmaceutical, nutraceutical, cosmetics, and food industries, as it has antioxidant, anti-inflammatory, antitumor, antidiabetic, and immunomodulatory properties. Moreover, it is also used in aquaculture, both for pigmentation and to improve the immune response and zootechnical performance of shrimp and fish [2,3,4,5].
In addition to astaxanthin, which represents about 4% of the cellular content in the cystic phase, this microalga also has a high concentration of lipids, varying according to the cultivation mode and the life cycle phase (vegetative or cystic) [6]. The increase in lipid accumulation occurs during the formation of aplanospores, simultaneously with the production of astaxanthin, and is induced by stress factors such as nutrient deprivation in the culture medium [7]. The high neutral lipid content and the adequate fatty acid profile make this species a potential source for nutraceuticals, aquafeed, and biofuel [8].
In the current scenario, biofuels produced from microalgae are considered a promising alternative for the production of sustainable energy, given the need to reduce the use of fossil fuels, as well as carbon emissions [9]. Compared with terrestrial cultures, microalgae have higher biomass productivity, rapid growth, and low need for water renewal in their cultivation. Furthermore, they require the use of smaller areas for production, and cultivation does not require the use of contaminants (pesticides or herbicides), only sunlight, water, and nutrients [10, 11]. However, having the ability to produce large amounts of lipids and having an ideal fatty acid profile for biofuel production depends on the species, cultivation conditions, season, and stage of the microalgae’s life cycle [12, 13].
Moreover, the viability of producing lipids from microalgae also depends on the extraction method used, which should provide good yield and cost-effectiveness, in addition to maintaining the integrity of the lipids, being essential for the production process and to ensure oil quality [7]. On a commercial scale, the most used extraction method, which is considered fast and with low lipid degradation, is solvent extraction. In this method, the solvents used must be of low toxicity, pure, immiscible in water, and selective [14]. Also, in order to facilitate the contact between the lipids and the solvent and to promote high yield, time reduction and low cost, biomass pretreatment methods, which can be physical (mechanical or thermal) and chemical, are necessary [15].
The determination of the cell pretreatment and lipid extraction methods to be used must consider the structure of the microalgal cell wall. H. pluvialis has a cell wall formed by cellulose and algaenan, present in the vegetative phase like a no acetolysis-resistant material, while in the cystic phase (aplanospores), it consists of a three-layered algaenan sheath and two polysaccharide layers, having a thicker and highly resistant structure against mechanical and chemical actions [16, 17]. Thus, the pretreatment of microalgae biomass in extraction processes with gamma radiation (γ), the most energetic form of electromagnetic radiation, has the potential to improve the yield and quality of the biomolecules obtained, as it acts in the modification of the structures of the polysaccharides that are present in the cell wall [18].
γ radiation is an effective technique for biomass pretreatment, and when combined with others (e.g., chemical and physical), an increase in the efficiency of the entire process is observed [19]. Gamma rays act on the degradation of polysaccharides, such as cellulose, lignin, alginate, carrageenan, and laminarin, which can cause rupture or depolymerization of the cell wall structure [18]. Irradiation is considered to be a promising alternative for biomass pretreatment [18, 19]. The advantages of this process include high energy efficiency, less energy requirement, easy handling, selectivity, mild temperature, short reaction time, low environmental impact, low capital investment, and few hazardous processes [20, 21].
The energy efficiency and cost-effectiveness of cell disruption and lipid extraction are the main challenges in the commercialization of microalgae biofuels, as the costs and energy of this process exceed the current price of crude oil. As a result, microalgae biofuels become less attractive compared to fossil fuels, leading to the need for further research in these areas [15]. In addition, feasibility in microalgae biofuel production can be achieved by using high-value-added coproducts from waste biomass after oil extraction, applying the concept of biorefinery [8, 22]. Residual biomass can be used to generate energy or other liquid and gaseous fuels [23], as well as in the food, nutraceutical, and pharmaceutical industry, as it has a wide variety of biomolecules, such as proteins, carbohydrates, vitamins, pigments, and antioxidants, which can guarantee the sustainability of biofuel production from microalgae [24, 25].
Therefore, further research in these areas focused on techniques that improve lipid extraction and biomass conversion into commercial chemicals and energy are essential to achieve a sustainable economy and increase the viability of microalgae-based biofuels. Thus, the aim of the present study was to apply gamma radiation as a method of cell pretreatment for lipid extraction and to obtain astaxanthin in the residual cystic biomass of the microalga H. pluvialis.
Materials and Methods
Experimental Design
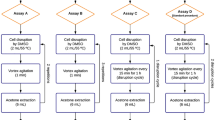
A bifactorial design was developed, with factor 1 being represented by the cell pretreatment methods, biomass with chloroform:methanol under ultrasound (BCMU), biomass with chloroform:methanol under γ radiation (BCMR), dry biomass under γ radiation (DBR), and control (without pretreatment), while factor 2 considered the two phases of the microalgae life cycle: vegetative and cystic. The interactions between the factors represented 8 combinations, in triplicate, with a total of 24 experimental runs.
Microalgal Strain and Cultivation
H. pluvialis was obtained from the Live Food Production Laboratory, at the Department of Fisheries and Aquaculture of the Federal Rural University of Pernambuco. This microalga was grown in fresh water that was previously treated with sodium hypochlorite at 3 ppm for 24 h (with aeration), filtered (40 µm) and autoclaved (120 °C for 30 min), and then enriched with the modified Provasoli culture medium [26] at pH 7.0, presenting in mg L−1: 15 C3H7Na2O6P, 1.98 C2H3NaO2, 2.0 yeast extract, 105 NaNO3, 0.075 ZnCl2, 0.0015 CoCl2.2H2O, 3.0 H3BO3, 24.9 EDTA-Na2.2H2O, 0.15 FeCl3.6H2O, 10.6 Fe(NH4)2(SO4)2.6 H2O, and 0.6 MnCl2.4H2O.
Cultures were developed in mixotrophic and semi-continuous modes, with microalgae being inoculated at an initial density of 5 × 104 cells mL−1, starting from 40 mL tubes to 20 L flasks. The cultures were maintained with constant aeration, temperature of 22 ± 1 °C, full photoperiod, and light intensity of 40 µmol photons m−2 s−1. Microalgal growth was monitored daily, using an optical microscope (magnification of 400 ×) with the aid of a Neubauer chamber. Upon reaching the stationary phase of growth, the vegetative biomass was harvested. For the production of cystic biomass, on the 12th day of cultivation (stationary phase), light intensity was increased (100 µmol photons m−2 s−1) to induce astaxanthin synthesis.
Biomass Harvesting and Drying
Cultures were harvested in the vegetative (zoospores) and cystic phase (aplanospores) by centrifugation (KC5 KINDLY, China) at 3500 × g for 15 min (22 °C). The wet biomass was stored at − 80 °C (SANYO MDF U33V) for 24 h and then freeze-dried at high vacuum and low temperature (ALPHA 1–4 LD PLUS) for 48 h [27]. Finally, the biomass was weighed in an analytical balance to determine dry biomass.
Cell Pretreatment
Cell pretreatment with ultrasound (USC-1400A UNIQUE, Brazil) was carried out in 0.7 g of lyophilized biomass homogenized with 14 mL of chloroform:methanol solvent ratio of 1:1 (v/v) at a frequency of 40 kHz and power of 135 W (two cycles of 15 min).
Gamma irradiation was performed at the Department of Nuclear Energy of the Federal University of Pernambuco, using a cobalt-60 gamma-irradiator (Gammacell 220 Excel MDS Nordion) operated at a dose rate of 1.87 kGy/h. The applied dose level was 5.0 kGy, and two methods were applied: radiation in dry biomass (DBR) and radiation in biomass with chloroform:methanol (1:1 v/v) (BCMR).
Lipid Extraction
Lipid extractions were performed adapting the methodology developed by Bligh and Dyer [28], after cell pretreatment, for biomass in the vegetative and cystic phases. After pretreatment of the dried biomass with radiation (DBR), chloroform:methanol (1:1 v/v) was added and vortexed for 2 min. Then, all samples were submitted to the other stages of lipid extraction. Initially, they were centrifuged at 11,200 × g for 15 min; the supernatant was collected; 5.25 mL of Milli-Q water was added, vortexed for 2 min, and centrifuged at 11,200 × g for 5 min. The mixtures were transferred into a separatory funnel and stayed for 1 h. The lipid fraction was collected in previously weighed glass tubes, and the solvent was evaporated using a laminar flow hood. After that, the tubes were weighed for the calculation of lipid yield (% DW dry weight) and then stored at − 80 °C protected from light.
Fatty Acid Methyl Ester (FAME) Transesterification and Analysis
Transesterification was performed in aliquot of the lipids (1 mL) using a solution of 0.5 mL of potassium hydroxide in methanol (0.5 mol L−1) and vortex-mixed for 2 min. After that, FAMEs were extracted with 2 mL n-hexane, vortex-mixed for 2 min, and centrifuged at 2000 × g for 6 min [29]. Finally, the supernatant was filtered with a 0.22 µm PTFE filter and stored at − 20 °C overnight.
FAMEs were analyzed by gas chromatography using a GC/FID (flame ionization detector), GC-2010Plus, equipped with AOC-20i autoinjector (Shimadzu, Kyoto, Japan). GC analysis was performed in a ZB-5HT capillary column with an initial column temperature of 150 °C (3 min) and a heating rate of 4 °C/min up to 280 °C and then kept at 280 °C for 15 min. The injector and detector temperatures were 250 °C and 300 °C, respectively. Helium was used as the carrier gas (30 mL/min), and the injection volume was 1 μL with a split ratio of 1:100. Chromatographic peaks were identified by comparing retention times with standard certificate material (Supelco FAME Mix C8–C24, Bellefonte, PA, USA). Three replicates of each FAME analysis were performed.
Astaxanthin Extraction, Analysis, and Quantification
The determination of astaxanthin followed the methodology of Dong et al. [30], in which 5 mg of residual cystic biomass from lipid extraction with different pretreatment methods and 5 mg of lyophilized crude biomass (control) were treated with 1 mL of HCl (4 M) and placed in an oven at 70 °C for 2 min and then cooled and centrifuged at 5000 × g for 5 min. The HCl-treated sample was washed twice with distilled water, centrifuged at 5000 × g for 5 min, resuspended in 1 mL of 90% acetone, and subjected to an ultrasonic bath on ice for 10 min. Subsequently, the sample was centrifuged again at 3500 × g for 5 min, and the obtained extract was stored in amber vials for quantification in UPLC-MS.
Chromatography was performed in an Acquity H-Class (Waters™, USA) Ultra Performance Liquid Chromatograph (UPLC), using a 2.1 × 100 mm HSS T3 column and a particle size of 1.8 µm. The mobile phases used consisted of acetonitrile solution containing 0.1% formic acid (eluent A), methanolic solution containing 0.1% formic acid (eluent B), and ethyl acetate solution containing 0.1% formic acid (eluent C), which were pumped at a flow rate of 0.37 mL min−1; elution was performed in isocratic mode (10% A/50% B/40% C) held for 5 min. Ten microliters of sample was injected, and the column and autoinjector temperatures were maintained at 40 and 10 °C, respectively. The UPLC system was coupled to a SQ Detector 2 single quadrupole mass spectrometer (Waters™, USA). The capillary voltage was 1.5 kV, the cone voltage was 50 V, and the desolvation temperature was 350 °C, with a source gas flow of 650 L h−1. Data acquisition was performed in selected ion recording (SIR) mode, seeking the mass of astaxanthin (597.35 Da), in positive ionization. The acquisition of chromatograms and mass spectra was performed using MassLynx™ software (Waters™, USA).
A calibration curve was used with an astaxanthin commercial standard with 97% purity (SML0982 Sigma, St. Louis, MS, USA) to quantify samples. The entire procedure was performed in triplicate and protected from light.
Statistical Analysis
All data are presented as mean ± standard deviation (n = 3). A two-way ANOVA was used to determine the effect of algal phase and cell pretreatment and their interaction on lipid yield and FAMEs, after confirming normality (Shapiro–Wilk’s test) and homoscedasticity (Bartlett’s test). The values for some FAMEs were transformed using Box–Cox transformation to correct for non-normality and heterogeneous variances. Tukey’s test was used when differences between factors and treatments were detected by ANOVA (p < 0.05). Astaxanthin yield data did not obtain normal distribution and were submitted to the Kruskal–Wallis test (p < 0.05). Statistical analyses were performed using the R Core Team [31].
Results and Discussion
Lipid Content According to Cell Pretreatment and Life Cycle Phases
The total lipid content varied significantly (p < 0.05) according to the phase of the life cycle of H. pluvialis, with an average yield of 15 ± 0.03% DW for the vegetative phase (zoospores) and 10 ± 0.02% DW for the cystic phase (aplanospores). It also differed significantly (p < 0.05) based on the applied extraction method, where BCMR resulted in higher lipid yield, 18.4 ± 1.1% for vegetative, and 13.6 ± 0.6% for cystic phase (Fig. 1). There was no significant difference (p > 0.05) in the interaction between phases and cell pretreatments.
Lipid yield using different cell pretreatment methods in the two phases of the life cycle of H. pluvialis (mean values between pretreatments in the same phase with different letters differ significantly by Tukey’s test. Control, without pretreatment; BCMU, biomass with chloroform:methanol under ultrasound; BCMR, biomass with chloroform:methanol under γ radiation; DBR, dry biomass under γ radiation)
In the vegetative phase, the lipid content can reach up to 25%, varying according to the culture conditions: temperature, pH, light intensity, culture medium, and culture system [7, 32]. In the present study, the average lipid yield of 15% can be attributed to culture conditions, such as low light intensity (40 µmol photons m−2 s−1) and no CO2 insertion, differing from the results of higher lipid content found in the literature, in which the luminosity varies between 100 and 300 µmol photons m−2 s−1 [32]. When considering the extraction methods used, these were suitable for the morphological characteristics of the species, especially regarding the composition of the cell wall [33]. In this phase, zoospores have a cell wall formed by cellulose — biosynthesized during the formation of the primary wall — and by residues of algaenan, a biopolymer formed by dicarboxylic acids, alcohols, and fatty acids (C22–C26), resistant to various chemical and enzymatic treatments [34]. These characteristics reveal the need for physical cellular pretreatment followed by the use of chemical agents, such as those used in this study.
In the cystic phase, the lipid content of aplanospores was lower than that of vegetative cells (Fig. 1) and had lower concentrations than in other culture systems, where they reach up to 37% DW [6]. The cell wall of the aplanospores is composed of a trilaminar algaenan sheath, below which is found a secondary layer composed largely of cellulose and mannose in a homogeneous arrangement, in addition to the tertiary layer with heterogeneously arranged cellulose and mannose [17]. This cell wall is thicker (2–3 ×) than vegetative cells and highly resistant to mechanical and chemical attacks, thus decreasing the bioavailability of accumulated compounds [35].
These characteristics lead to the need to use more aggressive methods in the pretreatment of cells, such as γ rays, which are the most energetic form of electromagnetic radiation and, therefore, have a greater capacity to penetrate the biomass than other types of radiation [19]. Gamma radiation acts in two ways: directly by interacting with biological molecules, promoting excitation, injury, and splitting of the polymeric structure or indirectly producing free radicals in cells; thus, it can damage or modify cell components, depending on the level of irradiation [20]. The solvent irradiation method (BCMR), in both phases of the microalgae life cycle, was more effective than the dry biomass irradiation (DBR) (Fig. 1). This was due to the high formation of free radicals that occurs in the liquid, consequently generating a greater indirect effect [36].
Gamma radiation is an effective technique for the pretreatment of lignocellulosic biomass, modifying the structure, reducing the degree of polymerization of cellulose, and disrupting the cell wall [37]. The use of radiation can be combined with other physical or chemical methods, allowing to reduce the radiation dose used [19, 38]. In the present study, by using a dose of 5.0 kGy with solvents, a greater efficiency in disrupting the cell wall of vegetative and cystic cells of H. pluvialis was found. According to Cheng et al. [39], doses of 0.25–4.0 kGy produce mutant H. pluvialis cells with greater biomass production, while 5.0 kGy prevents microalgae growth, as it causes irreversible damage to cells. Comparatively, in other studies, doses of 1.2 and 5.0 kGy were sufficient for the disruption and depolymerization of the cell wall structure of other biomasses [40, 41].
Gamma irradiation alone is an effective technique for biomass pretreatment, and when the irradiation process is combined with other ones (chemical, physical), there is an increase in the efficiency of the entire process [37]. Thus, the use of γ radiation without solvent (DBR) resulted in lipid yield similar to ultrasound with solvent (BCMU), because of the fact that, in dry conditions, there is a decrease in the efficiency of radiation [20].
Ultrasonic vibration has a direct proportion to power, providing physical effects such as the rise of osmoses and diffusion of solutes, in addition to turbulence between surfaces and damage to the cell wall [42]. Ultrasonic treatment must be allied to the characteristics of the cell wall of the species and biomass conditions, and a relatively high amount of energy is usually required due to the attenuation of power within the medium and due to high cell concentration [43]. For example, the maximum lipid yield for the wet Nannochloropsis and Chlorella paste is reached with powers of 400 and 1000 W, respectively [44, 45]. When ultrasound is applied after biomass harvesting and drying, lower powers can be used, such as 100 to 200 W. In fact, these values can be used for H. pluvialis powder, as in this study, where a power of 135 W was used [46, 47]. In addition, a combination with high temperatures and use of solvents results in more intense effects [43]. Dong et al. [30] using ultrasound combined with acetone, after pretreatment with HCl (4 M) at 70 °C, obtained a lipid yield of 33.3 ± 1.1% for H. pluvialis in the cystic phase, higher than that achieved with hexane, isopropanol, soybean oil, and chloroform:methanol.
Therefore, considering the morphological characteristics of aplanospores, higher radiation doses would certainly provide greater wall rupture, enabling greater lipid yield. However, it is worth noting that there is a need to verify the vulnerability of the compounds.
Fatty Acid Methyl Esters
The fatty acid profile of H. pluvialis was similar for the vegetative and cystic phases, the main ones found being palmitic (C16:0), palmitoleic (C16:1), oleic (C18:1n9c), linoleic (C18:2n6c), linolelaidic (C18:2n6t), and α-linolenic (C18:3n3), corroborating with the findings of Bilbao et al. [48]. Regarding the fatty acid content, there was a significant difference between the vegetative and cystic phases for all FAMEs, except for C18:2n6c (Table 1). As regards to cell pretreatment methods, only myristic (C14:0), palmitoleic (C16:1), α-linolenic (C18:3n3), erucic (C22:1n9), and lignoceric (C24:0) did not show significant difference (Table 1).
The level of saturated fatty acids (SFA) was lower than unsaturated (UFA) in all pretreatments and in the two phases, and both presented a significant difference between cell pretreatments, phases, and interaction of factors. The highest content of SFA was observed in the cystic phase and in the absence of cell pretreatment (control), followed by BCMR and BCMU (Table 1). The predominance of SFAs is a prerequisite to maintain the properties of biofuels, due to the lower risk of polymerization and greater stability when compared to UFAs [49]. Therefore, the high lipid yield combined with the higher SFA content makes cystic H. pluvialis under BCMR suitable for biofuel production.
In Fig. 2, the contents of the main fatty acids are represented in boxplot, C16:0 (a), C16:1 (b), C18:1n9c (c), C18:2n6c (d), C18:2n6t (e), and C18:3n3 (f), highlighting the differences between cell pretreatment methods and phases. As in other species of Chlorophyceae, in H. pluvialis, saturated palmitic (C16:0) and monounsaturated oleic (C18:1n9c) fatty acids are predominant in both the vegetative and cystic phases [50]. BCMR and BCMU pretreatments in the cystic phase reached higher palmitic acid content (Fig. 2a), while the oleic acid content was higher for the cystic phase in all cell pretreatments applied (Fig. 2c). As for polyunsaturated fatty acids (PUFAs), linoleic (Fig. 2d), linoelaidic (Fig. 2e), and linolenic (Fig. 2f) acids were predominant; these have a maximum of three unsaturations and an intermediate chain length, with a maximum of 18 carbons. This predominance of saturated or low unsaturated fatty acids, with medium carbon length, makes this microalga a potential to produce biodiesel that remains as liquid at low temperatures with a high energy content [7, 49]. Furthermore, microalgal biomass can also be used as feedstock for bioethanol or biomethane production after refining of value-added molecules, like astaxanthin [8].
Astaxanthin in Residual Cystic Biomass
Considering the residual cystic biomass after lipid extraction, conservation of the astaxanthin was observed when using the pretreatment method with gamma radiation in dry biomass (DBR), obtaining an astaxanthin concentration similar to raw biomass — not subjected to cell pretreatment and lipid extraction (control), of approximately 78 mg L−1 and 1.55% DW (Fig. 3). This method obtained a lipid yield equivalent to the method in which ultrasound was used and lower than the one using gamma radiation in biomass with solvent (Fig. 1). However, the carotenoid concentration in its residual biomass was higher (Fig. 3). The use of gamma radiation in dry biomass has an antioxidant effect [51], preserving astaxanthin from potential degradation, while the gamma radiation methods in biomass with solvent and ultrasound caused the degradation of this carotenoid.
Astaxanthin concentration in residual cystic biomass of H. pluvialis after cell pretreatment and lipid extraction (mean values with distinct superscripts are significantly different by Kruskal–Wallis test. Control, raw biomass; BCMU, residual biomass after ultrasound with chloroform:methanol; BCMR, residual biomass after γ radiation with chloroform:methanol; DBR, residual biomass after γ radiation on dry biomass)
The action of γ rays in dilute biomass causes high formation of free radicals, which contributed to a higher lipid yield, as well as to the greater performance of astaxanthin as an electron donor, neutralizing free radicals, due to its antioxidant property [2]. Furthermore, this carotenoid is a highly unsaturated molecule that easily decomposes when exposed to heat, light, and oxygen, whose reactions can be caused by the formation of free radicals [52]. Its exposure to ultraviolet light, for example, can result both in cis–trans isomerization and cause the destruction of this molecule under more energetic conditions, such as when exposed to a wavelength smaller than 300 nm and diluted in lipophilic solvents [2]. Gamma radiation has a very short wavelength and is very energetic, so when this method of cell pretreatment is combined with the use of chloroform:methanol in the biomass, it is possible to observe greater degradation of astaxanthin.
The ultrasound method caused the degradation of astaxanthin through the propagation of ultrasonic waves, which involves formation, growth, and collapse of microscopic bubbles, generating high temperatures and mechanical action between the biomass and solvent interfaces [53]. In this technique, the astaxanthin content decreases as the potency and treatment time increase, being able to reduce by 25.1% (100 W), 25.5% (300 W), and 29.4% (600 W) after 6 min [52]. In the present study, there was an 87% reduction after lipid extraction at the power of 135 W for 30 min and subsequent extraction of astaxanthin at the same power for 10 min.
Therefore, using the DBR method, it was possible to extract lipids (~ 8.6%) in the cystic phase and preserve astaxanthin from the residual biomass, demonstrating the importance of using γ radiation for these purposes. In H. pluvialis, astaxanthin is the most economically valuable bioproduct, due to its biological properties, being widely used in the pharmaceutical, nutraceutical, cosmetics, and food industries, in addition to aquaculture [1,2,3]. Thus, promoting the use of residual cystic biomass (from lipid extraction) to obtain the carotenoid offers greater economic viability to the biofuel production process.
Conclusions
The present study proposed a new and promising method of biomass pretreatment for lipid extraction from Haematococcus pluvialis using gamma rays. In this perspective, gamma radiation as a cell pretreatment promoted higher lipid yield of H. pluvialis in the vegetative and cystic phases. It is also possible to state that the predominance of saturated or low unsaturated fatty acids makes H. pluvialis, in both phases, suitable for biofuel production. In addition, astaxanthin is obtained from residual cystic biomass subjected to gamma radiation in dry biomass as a cell pretreatment of lipid extraction. Therefore, considering the current challenges regarding the economic feasibility of producing biofuels from microalgae, the extraction of high-value coproducts, such as astaxanthin, combined with high lipid yield and adequate lipid profile makes the process more viable.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Mota GCP, Moraes LBS, Oliveira CYB, Oliveira DWS, Abreu JL, Dantas DMM, Gálvez AO (2022) Astaxanthin from Haematococcus pluvialis: processes, applications, and market. Prep Biochem Biotechnol 52(5):598–609. https://doi.org/10.1080/10826068.2021.1966802
Ding W, Zhao Y, Xu JW, Zhao P, Li T, Ma H, Yu X (2018) Melatonin: a multifunctional molecule that triggers defense responses against high light and nitrogen starvation stress in Haematococcus pluvialis. J Agric Food Chem 66(29):7701–7711. https://doi.org/10.1021/acs.jafc.8b02178
Xie S, Fang W, Wei D, Liu Y, Yin P, Niu J, Tian L (2018) Dietary supplementation of Haematococcus pluvialis improved the immune capacity and low salinity tolerance ability of post-larval white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol 80:452–457. https://doi.org/10.1016/j.fsi.2018.06.039
Ahmed EA, Mohamed HE, Abd El-Salam HS (2022) In vitro antimicrobial activity of astaxanthin crude extract from Haematococcuspluvialis. Egypt J Aquat Biol 26:95–106. https://doi.org/10.21608/EJABF.2022.224854
Alateyah N, Ahmad SMS, Gupta I, Fouzat A, Thaher MI, Das P, Al Moustafa A-E, Ouhtit A (2022) Haematococcus pluvialis microalgae extract inhibits proliferation, invasion, and induces apoptosis in breast cancer cells. Front Nutr 9:882956. https://doi.org/10.3389/fnut.2022.882956
Shah M, Mahfuzur R, Liang Y, Cheng J, Daroch M (2016) Astaxanthin-producing green microalga Haematococcus pluvialis: from single cell to high value commercial products. Front Plant Sci 7:531. https://doi.org/10.3389/fpls.2016.00531
Saha SK, Mchugh E, Hayes J, Moane S, Walsh D, Murray P (2013) Effect of various stress-regulatory factors on biomass and lipid production in microalga Haematococcus pluvialis. Bioresour Technol 128:118–124. https://doi.org/10.1016/j.biortech.2012.10.049
Nishshanka GKSH, Liyanaarachchi VC, Nimarshana PHV, Ariyadasa TU, Chang JS (2022) Haematococcus pluvialis: a potential feedstock for multiple-product biorefining. J Clean Prod 344:131103. https://doi.org/10.1016/j.jclepro.2022.131103
Karpagam R, Jawaharraj K, Gnanam R (2021) Review on integrated biofuel production from microalgal biomass through the outset of transesterification route: a cascade approach for sustainable bioenergy. Sci Total Environ 766:144236. https://doi.org/10.1016/j.scitotenv.2020.144236
Shuba ES, Kifle D (2018) Microalgae to biofuels: promising alternative and renewable energy, review. Renew Sust Energ Rev 81:743–755. https://doi.org/10.1016/j.rser.2017.08.042
Correa DF, Beyer HL, Possingham HP, Fargione JE, Hill JD, Schenk PM (2021) Microalgal biofuel production at national scales: reducing conflicts with agricultural lands and biodiversity within countries. Energy 215:119033. https://doi.org/10.1016/j.energy.2020.119033
Foteinis S, Antoniadis-Gavriil A, Tsoutsos T (2018) Life cycle assessment of algae-to-biodiesel shallow pond production systems in the Mediterranean: influence of species, pond type, by (co)-product valorisation and electricity mix. Biofuel Bioprod Biorefin 4:542–558. https://doi.org/10.1002/bbb.1871
El-Sheekh MM, Gheda SF, El-Sayed AEKB, Abo Shady AM, El-Sheikh ME (2019) Schagerl M (2019) Outdoor cultivation of the green microalga Chlorella vulgaris under stress conditions as a feedstock for biofuel. Environ Sci Pollut Res 26:18520–18532. https://doi.org/10.1007/s11356-019-05108-y
Enamala M, Enamala S, Chavali M, Donepudi J, Yadavalli R, Kolapalli B, Aradhyula T, Velpuri J, Kuppam C (2018) Production of biofuels from microalgae-a review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renew Sustain Energy Rev 94:49–68. https://doi.org/10.1016/j.rser.2018.05.012
Ramluckan K, Moodley K, Bux F (2014) An evaluation of the efficacy of using selected solvents for the extraction of lipids from algal biomass by the Soxhlet extraction method. Fuel 116:103–108. https://doi.org/10.1016/j.fuel.2013.07.118
Onumaegbu C, Mooney J, Alaswad A, Olabi A (2018) Pretreatment methods for production of biofuel from microalgae biomass. Renew Sustain Energy Rev 93:16–26. https://doi.org/10.1016/j.rser.2018.04.015
Hagen C, Siegmund S, Braune W (2002) Ultrastructural and chemical changes in the cell wall of Haematococcus pluvialis (Volvocales, Chlorophyta) during aplanospore formation. Eur J Phycol 37(2):217–226. https://doi.org/10.1017/S0967026202003669
Kim B, Lee SY, Narasimhan AL, Kim S, Oh YK (2022) Cell disruption and astaxanthin extraction from Haematococcus pluvialis: recent advances. Bioresour Technol 343:126124. https://doi.org/10.1016/j.biortech.2021.126124
Sankaran R, Cruz RAP, Pakalapati H, Show PL, Ling TC, Chen WH, Tao Y (2020) Recent advances in the pretreatment of microalgal and lignocellulosic biomass: a comprehensive review. Bioresour Technol 298:122476. https://doi.org/10.1016/j.biortech.2019.122476
Hassan SS, Williams GA, Jaiswal AK (2018) Emerging technologies for the pretreatment of lignocellulosic biomass. Bioresour Technol 262:310–318. https://doi.org/10.1016/j.biortech.2018.04.099
Kassim MA, Khalil HPSA, Serri NA, Kassim MHM, Syakir MI, Aprila NS, Dungani R (2016) Irradiation pretreatment of tropical biomass and biofiber for biofuel production. In: Monteiro W (ed) Radiation effects in materials, 1st edn. IntechOpen, Croatia, pp 329–338. https://dx.doi.org/10.5772/62728
Subhash GV, Rajvanshi M, Kumar GRK, Sagaram US, Prasad V, Govindachary S, Dasgupta S (2022) Challenges in microalgal biofuel production: a perspective on techno economic feasibility under biorefinery stratagem. Bioresour Technol 343:126155. https://doi.org/10.1016/j.biortech.2021.126155
Chia SR, Chew KW, Show PL, Yap YJ, Ong HC, Ling TC, Chang JS (2018) Analysis of economic and environmental aspects of microalgae biorefinery for biofuels production: a review. Biotechnol J 13(6):1700618. https://doi.org/10.1002/biot.201700618
Chew KW, Yap JY, Show PL, Suan NH, Juan JC, Ling TC, Chang JS (2017) Microalgae biorefinery: high value products perspectives. Bioresour Technol 229:53–62. https://doi.org/10.1016/j.biortech.2017.01.006
Chen J, Li J, Dong W, Zhang X, Tyagi RD, Drogui P, Surampalli RY (2018) The potential of microalgae in biodiesel production. Renew Sust Energ Rev 90:336–346. https://doi.org/10.1016/j.rser.2018.03.073
Marinho YF, Malafaia CB, de Araújo KS, da Silva TD, dos Santos APF, de Moraes LB, Gálvez AO (2021) Evaluation of the influence of different culture media on growth, life cycle, biochemical composition, and astaxanthin production in Haematococcus pluvialis. Aquac Int 29(2):757–778. https://doi.org/10.1007/s10499-021-00655-z
Silva DLB, de Moraes LBS, Oliveira CYB, da Silva Campos CVF, de Souza BR, Gálvez AO (2022) Influence of culture medium on growth and protein production by Haematococcus pluvialis. Acta Sci Technol 44:e59590–e59590. https://doi.org/10.4025/actascitechnol.v44i1.59590
Bligh E, Dyer W (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
de Araújo KS, Siqueira SM, dos Santos LD, Malafaia CB, Barbosa MO (2019) A preliminary study of the physico-chemical properties and fatty acid profile of five palm oil genotypes cultivated in Northeast of Brazil. J Env Anal Progr 4(4):251–256. https://doi.org/10.24221/jeap.4.4.2019.2632.251-256
Dong S, Huang Y, Zhang R, Wang S, Liu Y (2014) Four different methods comparison for extraction of astaxanthin from green alga Haematococcus pluvialis. Sci World J. https://doi.org/10.1155/2014/694305
R Core Team (2021) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. Available at https://www.R-project.org/. Accessed 13 July 2021
Chekanov K, Lobakova E, Selyakh I, Semenova L, Sidorov R, Solovchenko A (2014) Accumulation of astaxanthin by a new Haematococcus pluvialis strain BM1 from the White Sea coastal rocks (Russia). Mar Drugs 12(8):4504–4520. https://doi.org/10.3390/md12084504
Rizwan M, Mujtaba G, Memon S, Lee K, Rashid N (2018) Exploring the potential of microalgae for new biotechnology applications and beyond: a review. Renew Sustain Energy Rev 92:394–404. https://doi.org/10.1016/j.rser.2018.04.034
Wang SB, Hu Q, Sommerfeld M, Chen F (2004) Cell wall proteomics of the green alga Haematococcus pluvialis (Chlorophyceae). Proteomics 4(3):692–708. https://doi.org/10.1002/pmic.200300634
Praveenkumar R, Lee K, Lee J, Oh YK (2015) Breaking dormancy: an energy-efficient means of recovering astaxanthin from microalgae. Green Chem 17(2):1226–1234. https://doi.org/10.1039/C4GC01413H
Singh R, Singh D, Singh A (2016) Radiation sterilization of tissue allografts: a review. World J Radiol 8(4):55. https://doi.org/10.4329/wjr.v8.i4.355
Li QM, Li XJ, Xiong XY, Hu QL, Tan XH, Wang KQ, Su XJ (2016) Analysis of degradation products and structural characterization of giant reed and Chinese silvergrass pretreated by 60Co-γ irradiation. Ind Crops Prod 83:307–315. https://doi.org/10.4329/wjr.v8.i4.355
Torun M (2017) Radiation pretreatment of biomass. In: Sun Y, Chmielewski A (eds) Applications of ionizing radiation in materials processing, 2nd edn. Institute of Nuclear Chemistry and Technology, Warszawa, pp 447–460
Cheng J, Li K, Yang Z, Zhou J, Cen K (2016) Enhancing the growth rate and astaxanthin yield of Haematococcus pluvialis by nuclear irradiation and high concentration of carbon dioxide stress. Bioresour Technol 204:49–54. https://doi.org/10.1016/j.biortech.2015.12.076
Torun M, Abbasova D, Solpan D, Güven O (2014) Caffeine degradation in water by gamma irradiation, zonation and zonation/gamma irradiation. Nukleonika 59(1):25–35. https://doi.org/10.2478/nuka-2014-0004
Bhat NA, Wani IA, Hamdani AM, Gani A, Masoodi FA (2016) Physicochemical properties of whole wheat flour as affected by gamma irradiation. LWT- Food Sci Technol 71:175–183. https://doi.org/10.1016/j.lwt.2016.03.024
Gao Y, Nagy B, Liu X, Simándi B, Wang Q (2009) Supercritical CO2 extraction of lutein esters from marigold (Tagetes erecta L) enhanced by ultrasound. J Supercrit Fluids 49(3):345–350. https://doi.org/10.1016/j.supflu.2009.02.006
Lee SY, Cho JM, Chang YK, Oh YK (2017) Cell disruption and lipid extraction for microalgal biorefineries: a review. Bioresour Technol 244:1317–1328. https://doi.org/10.1016/j.biortech.2017.06.038
Adam F, Abert-Vian M, Peltier G, Chemat F (2012) “Solvent-free” ultrasound-assisted extraction of lipids from fresh microalgae cells: a green, clean and scalable process. Bioresour Technol 114:457–465. https://doi.org/10.1016/j.biortech.2012.02.096
Natarajan R, Ang W, Chen X, Voigtmann M, Lau R (2014) Lipid releasing characteristics of microalgae species through continuous ultrasonication. Bioresour Technol 158:7–11. https://doi.org/10.1016/j.biortech.2014.01.146
Zou TB, Jia Q, Li HW, Wang CX, Wu HF (2013) Response surface methodology for ultrasound-assisted extraction of astaxanthin from Haematococcus pluvialis. Mar Drugs 11(5):1644–1655. https://doi.org/10.3390/md11051644
Khoo KS, Chew KW, Yew GY, Manickam S, Ooi CW, Show PL (2020) Integrated ultrasound-assisted liquid biphasic flotation for efficient extraction of astaxanthin from Haematococcuspluvialis. Ultrason Sonochem 67:105052. https://doi.org/10.1016/j.ultsonch.2020.105052
Bilbao S, Damiani C, Salvador G, Leonardi P (2016) Haematococcus pluvialis as a source of fatty acids and phytosterols: potential nutritional and biological implications. J Appl Phycol 28(6):3283–3294. https://doi.org/10.1007/s10811-016-0899-z
Nigam PS, Singh A (2011) Production of liquid biofuels from renewable resources. Prog Energy Combust Sci 37(1):52–68. https://doi.org/10.1016/j.pecs.2010.01.003
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54(4):621–639. https://doi.org/10.1111/j.1365-313X.2008.03492.x
Khawory M, Sain A, Rosli M, Ishak M, Noordin M, Wahab H (2020) Effects of gamma radiation treatment on three different medicinal plants: microbial limit test, total phenolic content, in vitro cytotoxicity effect and antioxidant assay. Appl Radiat Isot 157:109013. https://doi.org/10.1016/j.apradiso.2019.109013
Zhao L, Zhao G, Chen F, Wang Z, Wu J, Hu X (2006) Different effects of microwave and ultrasound on the stability of (all-E)-astaxanthin. J Agric Food Chem 54(21):8346–8351. https://doi.org/10.1021/jf061876d
Deora NS, Misra NN, Deswal A, Mishra HN, Cullen PJ, Tiwari BK (2013) Ultrasound for improved crystallisation in food processing. Food Eng Rev 5:36–44. https://doi.org/10.1007/s12393-012-9061-0
Acknowledgements
The authors would like to thank the Foundation for Science and Technology of the State of Pernambuco (FACEPE) for the aid granted to Laenne Moraes [14722.01/16] and the National Council for Scientific and Technological Development (CNPq) for the aid granted to Alfredo Gálvez [PQ 308063/2019-8] and Ranilson Bezerra [PQ 307107/2019-1] and the Northeast Strategic Technologies Center (CETENE) for its availability to carry out the analyses.
Funding
This work was supported by the Foundation for Science and Technology of the State of Pernambuco (FACEPE) (grant number 14722.01/16 to Laenne Moraes) and the National Council for Scientific and Technological Development (CNPq) (grant numbers PQ 308063/2019–8 to Alfredo Gálvez and PQ 307107/2019–1 to Ranilson Bezerra).
Author information
Authors and Affiliations
Contributions
Writing — original draft preparation, Laenne Moraes; conceptualization, Laenne Moraes, Carolina Malafaia, Yllana Marinho, and Ranilson Bezerra; investigation, Laenne Moraes, Géssica Mota, Carolina Malafaia, and Túlio Silva; methodology, Laenne Moraes, Carolina Malafaia, Antônio Oliveira, and Ana Melo; formal analysis, Laenne Moraes, Túlio Silva, and Antônio Oliveira; writing — review and editing, Carolina Malafaia, Yllana Marinho, and Alfredo Gálvez; resources, Ranilson Bezerra, Alfredo Gálvez, and Ana Melo; supervision, Ranilson Bezerra and Danielli Dantas; and project administration, Ranilson Bezerra.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Moraes, L.B.S., Malafaia, C.B., da Silva, T.D. et al. Gamma Radiation as a Pretreatment for Co-extraction of Lipids and Astaxanthin in Haematococcus pluvialis. Bioenerg. Res. 16, 1841–1850 (2023). https://doi.org/10.1007/s12155-022-10531-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-022-10531-0