Abstract
Objective
Lymphoscintigraphy and sentinel node biopsy are used for the detection of axillary lymph node metastasis in breast cancer patients. However, currently there is no standardized technique. For the detection of axillary lymph node metastasis by lymphoscintigraphy and sentinel node biopsy, in patients with breast cancer, we compared the results of subareolar injections administered on the day of surgery (1-day protocol) with injections administered on the day before surgery (2-day protocol).
Materials and methods
This study included 412 breast cancer patients who underwent surgery between 2001 and 2004. For the 1-day protocol (1 h before surgery) 0.8 ml of Tc-99m Tin-Colloid (37 MBq) was injected in 203 in the subareolar region on the morning of the surgery. For the 2-day protocol (16 h before surgery) 0.8 ml of Tc-99m Tin-Colloid (185 MBq) was injected in 209 patients on the afternoon before surgery. Lymphoscintigraphy was performed in the supine position and sentinel node identification was performed by hand-held gamma probe during surgery.
Results
Among 203 patients with the 1-day protocol, 185 cases (91.1%) were identified by sentinel node lymphoscintigraphy, and 182 cases (89.7%) were identified by gamma probe. Among the 209 patients, in the 2-day protocol, 189 cases (90.4%) had the sentinel node identified by lymphoscintigraphy, and 182 cases (87.1%) by the gamma probe. There was no significant difference in the identification rate of the sentinel node between the 1-day and 2-day protocols by lymphoscintigraphy and the gamma probe (p > 0.05, p > 0.05).
Conclusions
The results of the identification of the sentinel node by subareolar injection according to 1-day or 2-day protocol, in breast cancer patients, showed no significant differences. Because the 2-day protocol allows for an adequate amount of time to perform the lymphoscintigraphy, it is a more useful protocol for the identification of sentinel nodes in patients with breast cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Axillary LN metastasis is the most important factor to consider in the patient prognosis and staging of breast cancer. In addition, after surgery the LN status will influence the decision as to whether chemotherapy is necessary and the determination of the optimal treatment plan. The goal of patient treatment is to prevent disease recurrence and metastasis by local control of the cancer. When axillary lymph node metastasis is suspected, lymphadectomy of level I, II, III axillary lymph nodes is performed to confirm metastasis [1]. When all of the metastatic lymph nodes are removed, the patient survival increases [2]. However, lymphadectomy may miss metastasis in about 60% of patients [3]. In addition, lymph edema develops in about 39% of patients [4].
Recently, the sentinel lymph node method has been introduced as a non-invasive technique to confirm the presence of axillary lymph node metastasis. The sentinel lymph node is the first lymph node to receive the lymphatic vessel influx automatically from the primary tumor. It is the lymph node initially affected by a metastasis. When the sentinel lymph node by biopsy is negative there is no axillary LN metastasis [5]. Consequently, the decision as to whether to perform a lymphadectomy or a simple mastectomy depends on confirmation of the status of sentinel lymph node metastasis.
Currently, sentinel lymph node identification is performed by lymphoscintigraphy after injection of radiopharmaceuticals before surgery, with a gamma probe used during surgery to identify the sentinel lymph node. However, it can be difficult to coordinate the lymphoscintigraphy with the surgery. Therefore, in this study we compared the 1-day protocol (injection the morning of surgery) and the 2-day protocol (injection the day before surgery) and investigated whether there was a difference between the two protocols in the identification rate of the sentinel lymph node by the gamma probe and lymphoscintigraphy.
Methods
Patients
Totally, 412 patients with breast surgery from 2001 to 2004 and confirmed breast cancer were recruited. The number of patients injected at the morning of surgery (1 day protocol) was 203, and the number of patients injected at the day before surgery (2 day protocol) was 209. The mean age of the patients was 47.5 (range 24–76 years). The mean age of the patients with the 1-day protocol was 47.6 (range, 24–75 years). The mean age of the patients with the 2-day protocol was 47.9 (range, 25–76 years). There were 55 patients with ductal carcinoma in situ, 329 patients with invasive ductal carcinoma, 11 patients with invasive lobular carcinoma, 2 patients with medullary carcinoma, and 15 patients with mucinous carcinoma. Among the 203 patients with the 1-day protocol, 160 had invasive ductal carcinoma (78.8%). Among the 209 patients with the 2-day protocol, 169 had invasive ductal carcinoma (80.9%). There were 25 patients with tumor in the breast center including nipple, 87 patients in upper inner quadrant, 227 patients in upper outer quadrant, 21 patients in upper inner quadrant, and 52 patients in upper outer quadrant. Among the 203 patients with the 1-day protocol, 114 had tumor in the upper outer quadrant (56.2%). Among the 209 patients with the 2-day protocol, 113 had tumor in the upper outer quadrant (54.1%) (Table 1). All patients received a sentinel lymph node biopsy and lymphoscintigraphy.
Lymphoscintigraphy
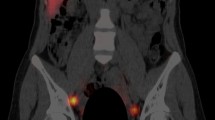
For the 1-day protocol, 99mTc-Tin colloid 37 MBq (total dosage 0.8 ml) on the morning of surgery (1 h before the operation) was injected in the subareolar area. Planar images of the chest were acquired at 5, 15, 30, and 60 min in the sitting position. Lymphoscintigraphy was performed with a two-head gamma camera with a low energy high resolution collimator (Vertex™, ADAC, Milpitas, CA, USA) with a 256 × 256 pixel matrix. The energy range was 20% at 140 keV. For the 2-day protocol, 99mTc-Tin Colloid 185 MBq (total dosage 0.8 ml) was injected the day before surgery (16 h before the operation) in the subareolar area. After injection planar images were acquired at 5, 15, 30, and 60 min by the same method. The lymphoscintigraphy was interpreted by two nuclear medicine physicians who were unaware of the clinical information and prior radiological findings. The patients were divided into those with clear local increase in the vicinity of the breast within 60 min and those without increase until 60 min (Fig. 1).
(a) The 1-day protocol. Anterior chest image shows subareolar injection site in left breast and focal uptake in sentinel node in left axilla (arrow). (b) The 2-day protocol. Anterior chest image shows subareolar injection site in right breast and focal uptake in sentinel node in right axilla (arrow)
Sentinel lymph node biopsy
Primary tumor removal and axillary lymph node dissection were performed in all patients. After the rough location of the sentinel lymph node was confirmed by the images of the lymphoscintigraphy, the areas with high-radiation doses were identified with the gamma probe. If the location of the sentinel lymph node was not confirmed by lymphoscintigraphy, the areas nearest to the injection site were identified among the areas with high-radiation doses with the gamma probe. The tissue biopsy samples were stained with Hematoxylin-Eosin for identification of metastasis to the sentinel lymph node and axillary lymph node.
Statistics
We defined the identification rate as the ability to identify a sentinel lymph node successfully, the false-negative rate (FNR) as the number of patients with a non-metastatic sentinel lymph node but one or more metastatic axillary nodes divided by the total number of patients with metastatic sentinel node or metastatic axillary nodes, and the negative predictive value (NPV) as the number of the true negative sentinel lymph nodes divided by the sum of the true and false-negative sentinel lymph nodes. Cross-tabulation analysis using the Fisher’s exact test for a statistical association between categorical variables was performed. We considered a p value below 0.05 as statistically significant.
Results
Sentinel lymph node identification by lymphoscintigraphy
The number of patients with sentinel lymph nodes identified by lymphoscintigraphy was 185 out of 203 patients with the 1-day protocol, 91.1%. For the 209 patients with the 2-day protocol, the number of patients with a sentinel lymph node identified by lymphoscintigraphy was 189, 90.4%. There was no significant difference between the 1-day and 2-day protocols for the identification of the sentinel lymph nodes by lymphoscintigraphy (p > 0.05) (Table 2).
Sentinel lymph node identification by the gamma probe
The number of patients whose sentinel lymph node was identified by the gamma probe was 182 out of 203 patients with the 1-day protocol, 89.7%. For the 209 patients with the 2-day protocol, the number of patients whose sentinel lymph node was identified by the gamma probe was 182, 87.1%. There was no significant difference between the cases that had the 1-day protocol or 2-day protocol in the identification of the sentinel lymph nodes by the gamma probe (p > 0.05) (Table 2).
FNR and NPV of the sentinel lymph node biopsy
Among the 182 patients with the sentinel lymph node identified by the gamma probe with the 1-day protocol, the number of cases with negative sentinel lymph nodes was 143. Among them, there were nine false-negative cases with positive non-sentinel lymph nodes. The FNR divided by 48 patients with positive axillary lymph nodes was 18.8%. The number of true negative cases was 134, and the NPV divided by 143 cases with negative sentinel lymph nodes was 93.7%. Among 182 patients with the sentinel lymph node identified by the gamma probe with the 2-day protocol, the number of cases with negative sentinel lymph nodes was 151. Among them, there were seven false-negative cases with positive non-sentinel lymph nodes. The FNR divided by 38 patients with positive axillary lymph nodes was 18.4%. The number of true negative cases was 144, and the NPV divided by 151 cases with negative sentinel lymph nodes was 95.3%. There was no significant difference between the 1-day protocol and 2-day protocol in the FNR or NPV of the sentinel lymph node biopsy (p > 0.05) (Table 3).
Discussion
The sentinel lymph node is the initial site of metastasis from the primary tumor to the lymph nodes. The concept of the sentinel lymph node was introduced initially by Cabanas [6] in 1977. Morton et al [7] in 1992 suggested that metastasis to the sentinel lymph node accurately reflected lymph node metastasis in melanoma patients. Krag et al. [8] in 1993 reported that a sentinel lymph node biopsy had an 82% biopsy success rate and 100% accuracy with 99mTc-sulfur colloid in breast cancer. After this, the research on sentinel lymph node identification with radiopharmaceuticals and dyes advanced rapidly.
Giuliano et al [9] reported a 94% detection rate and 100% accuracy with dye. Krag et al. [10] reported a 91% detection rate and 97% accuracy with radiopharmaceuticals. With the high identification rate and accuracy, the clinical possibilities for sentinel lymph node detection of lymph node metastasis were encouraging. However, the usefulness of lymphoscintigraphy prior to surgery was unclear. We used the subareolar injection method in this study, because it identified the sentinel lymph nodes more quickly than the peritumoral method. Although the peritumoral method is now more widely performed the subareolar, one may replace it as a simpler detection method of sentinel lymph nodes [11–14].
Because of time constraints the coordination of lymphoscintigraphy with surgery may be difficult to achieve [15–18]. The results of this study showed that there was a high-identification rate of sentinel lymph nodes, 91.1% by lymphoscintigraphy and 89.7% by gamma probe with the 1 day protocol. For the 2-day protocol the sentinel lymph node identification rate was 90.4% by lymphoscintigraphy and 87.1% by gamma probe. There was no significant difference in the identification rate between the two methods (p > 0.05). However, the number of patients with sentinel nodes identified by lymphoscintigraphy was larger in both the 1-and 2-day protocols than by the gamma probe. The technique of sentinel node identification by gamma probe had need of more experience than sentinel node identification by lymphoscintigraphy. In our study, the result of the sentinel node identification by gamma probe was influenced by the absence of the regular technician to use the gamma probe.
The FNR and the NPV of the sentinel lymph node biopsy with the 1-day protocol were 18.8 and 93.7%, respectively. The FNR and the NPV of the sentinel node biopsy for the 2-day protocol were 18.4 and 95.3%, respectively. There was no significant difference between the 1- and 2-day protocols for the FNR or the NPV of the sentinel lymph node biopsy (p > 0.05). However, the FNR of this study was higher than in previous studies [19–21]. The radiopharmaceutical used in this study was Tc-99m Tin-Colloid, which is the most commonly used commercially available radiopharmaceutical in Korea. Tc-99m Tin-Colloid moved very slowly because of its large particle size (100–1000 nm), and this induced the high FNR.
However, the 2-day protocol has several advantages; the patients may undergo the lymphoscintigraphy in a quiet state and may avoid the NPO. Nuclear medicine physicians may have enough time to perform delayed images and to interpret them. Since the results of this study showed that taking enough time to identify the sentinel nodes after injection of radiopharmaceuticals and the identification of sentinel lymph nodes by lymphoscintigraphy helped identify the sentinel lymph node by the gamma probe, injection of the radiopharmaceutical the day before surgery may provide the optimal time frame in which to carry out this procedure.
References
Van der Wall E. The sentinel node in breast cancer: implications for adjuvant treatment? Eur J Nucl Med. 1999;26(Suppl):s17–9.
Morrow M. Role of axillary dissection in breast cancer management. Ann Surg Oncol. 1996;3:233–4.
Cascinelli N, Greco M, Bufalino R, Clemente C, Galluzzo D, Delle Donne V, et al. Prognosis of breast cancer with axillary lymph node metastases after surgical treatment only. Eur J Cancer Clin Oncol. 1987;23:795–9.
Kissin MW, Querci della Rovere G, Easton D, Westburv G. Risk of lymphedema after the treatment of the breast cancer. Br J Surg. 1986;73:580–4.
Tiourina T, Arends B, Huysmans D, Rutten H, Lemaire B, Muller S. Evaluation of surgical gamma probes for radioguided sentinel node localization. Eur J Nucl Med. 1998;25:1224–31.
Cabanas RM. An approach for the treatment of penile carcinoma. Cancer. 1977;39:456–66.
Morton DL, Wen DR, Wong JH. Technical detail of intraoperative lymphatic mapping for early stage melanoma. Arch Surg. 1992;127:392–9.
Krag DN, Weaver DL, Alex JC. Surgical resection and radiolocalization of the sentinel node in breast cancer using gamma probe. Surg Oncol. 1993;2:335–40.
Giuliano AE, Jones RC, Brennan M, Statman R. Sentinel lymphadenectomy in breast cancer. J Clin Oncol. 1997;15:2345–50.
Krag D, Weaver D, Ashikaga T, Moffat F, Klimberg VS, Shriver C, et al. The sentinel node in breast cancer-a multicenter validation study. N Eng J Med. 1998;339:941–6.
D’Eredita G, Giardina C, Guerrieri AM, Berardi T. A further validation of subareolar injection technique for breast sentinel lymph node biopsy. Ann Surg Oncol. 2006;13:701–7.
Peley G, Sinkovies I, Toth J, Farkas E, Keresztes S, Koves I. Subareolar injection of radioactive colloid for sentinel lymph node identification in breast cancer patients. Am Surg. 2004;70:625–9.
Maza S, Thomas A, Winzer KJ, Huttner C, Blohmer JU, Hauschild M, et al. Subareolar injection of technetium-99m nanocolloid yields reliable data on the axillary lymph node tumour status in breast cancer patients with previous manipulations on the primary tumour: a prospective study of 117 patients. Eur J Nucl Med Mol Imaging. 2004;31:671–5.
D’Eredita G, Ferrarese F, Cecere V, Massa ST, de Carne F, Fabiano G. Subareolar injection may be more accurate than other techniques for sentinel lymph node biopsy in breast cancer. Ann Surg Oncol. 2003;10:942–7.
McCarter MD, Yeung H, Yeh S, Fey J, Borqen PI, Cody HS 3rd. Localization of the sentinel node in breast cancer: identical results with same-day and day-before isotope injection. Ann Surg Oncol. 2001;8:682–6.
Koizumi M, Nomura E, Yamada Y, Takiguchi T, Tanaka K, Yoshimoto M, et al. Sentinel node detection using 99mTc-rhenium sulphide colloid in breast cancer patients: evaluation of 1 day and 2 day protocols, and a dose-finding study. Nucl Med Commun. 2003;24:663–70.
Yeung HW, Cody HS III, Turlakow A, Riedel ER, Fey J, Gonen M, et al. Lymphoscintigraphy and sentinel node localization in breast cancer patients: a comparison between 1-day and 2-day protocols. J Nucl Med. 2001;42:420–3.
Gutman F, Sanson A, Piquenot JM, Hitzel A, Ladonne JM, Dessoqne P, et al. Intra-individual comparison of sentinel lymph node scintigraphy on the day of injection and on the following day in breast cancer. Nucl Med Commun. 2006;27:5–9.
Jaderbord JM, Harrison PB, Kiser JL, Maynard SL. The feasibility and accuracy of the sentinel lymph node biopsy for breast carcinoma. Am Surg. 1999;65:704–5.
Bedrosian I, Reynolds C, Mick R, Callans LS, Grant CS, Donohue JH, et al. Accuracy of sentinel lymph node biopsy in patients with large primary breast tumors. Cancer. 2000;88:2540–5.
Doting MH, Jansen L, Nieweg OE, Piers DA, Tiebosch AT, Koops HS, et al. Lymphatic mapping with intralesional tracer administration in breast carcinoma patients. Cancer. 2000;88:2546–52.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seok, J.W., Kim, I.J., Heo, YJ. et al. Comparison of subareolar injection lymphoscintigraphy with the 1-day and the 2-day protocols for the detection of sentinel lymph nodes in patients with breast cancer. Ann Nucl Med 23, 465–469 (2009). https://doi.org/10.1007/s12149-009-0259-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-009-0259-z