Abstract
Background
Central lymph node metastasis (LNM) in papillary thyroid carcinoma (PTC) is common. But the association between primary tumor characteristics and specific features of metastatic lymph nodes in PTC has not been fully identified. Determining risk factors for LNM may help surgeons determine rational extent of lymph node dissection.
Methods
Data from 432 patients who underwent thyroidectomy with cervical lymph node dissection for PTC were retrospectively analyzed. The relationships between LNM to central compartment or lateral compartment and clinicopathologic factors were analyzed. Cox regression model was used to determine the risk factors for recurrence-free survival (RFS).
Results
Central lymph node metastasis (CLNM) and lateral lymph node metastasis (LLNM) were found in 216 (50.0%) and 65 (15.0%) patients, respectively. In the multivariate analysis for CLNM, patients < 45 years of age (OR 2.037, 95% CI 1.388–2.988, P < 0.001), extrathyroidal invasion (OR: 2.144, 95% CI 0.824–5.457, P = 0.011), vascular invasion (OR 13.817, 95% CI 1.694–112.693, P = 0.014), LLNM (OR 2.851, 95% CI 1.196–6.797, P = 0.014) and TNM Stage III–IV (OR 465.307, 95% CI 113.903–1900.826, P < 0.001) were independent predictors for high prevalence of CLNM. In the multivariate analysis for LLNM, tumor size more than 1cm (OR 3.474, 95% CI 1.728–6.985, P < 0.001) and CLNM (OR 5.532, 95% CI 2.679–11.425, P < 0.001) were independent predictors for high prevalence of LLNM. Moreover, tumor with T3–T4 stage, extrathyroidal invasion and CLNM were the significant factors related to the RFS.
Conclusion
For patients with pre-operative risk factors of LNM, an accurate preoperative evaluation of central compartment or lateral compartment is needed to find suspicious lymph nodes. And prophylactic lymph node dissection should be performed in patients with high risk of CLNM. Moreover, we suggest performing close follow-up for patients with high risk of RFS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past few decades, the incidence of thyroid cancer (TC), specially papillary thyroid cancer (PTC), is rising at the fastest rate of all malignancies [1, 2]. PTC is generally indolent but very frequently metastasizes to the regional lymph nodes, including central and lateral compartments. The incidence of lymph node metastases (LNM) in cases of PTC has been reported to range from 30 to 80% [3, 4, 5]. Some studies have reported that LNM is a poor prognostic factor [6, 7, 8].
For patients with central lymph node metastasis (CLNM) or lateral lymph node metastases (LLNM) detected by preoperative ultrasonography (US) or physical examination, therapeutic lymph node dissection is advocated [9]. However, there is no consensus on the indications for prophylactic lymph node dissection. The American Thyroid Association (ATA) suggest that prophylactic central neck dissection (CND) should be performed in PTC patients with T3 or T4 stage [10], while the National Comprehensive Cancer Network (NCCN) guideline indicates prophylactic CND for patients aged less than15 or more than 45 years, with tumors more than 4 cm in diameter or with extrathyroidal extension [11]. Besides, the appropriate extent of prophylactic lymph node dissection remains controversial. Radical surgery, such as lateral neck dissection (LND), may injury the spinal accessory nerve or the cervical plexus, which lead to clinically important postoperative morbidities (shoulder dysfunction, neck numbness, and neuropathic pain) [12, 13]. Therefore, assessing high-risk patients with LNM is essential to perform individualized treatment and to avoid the unnecessary prophylactic lymph node dissection.
To find the optimal management of cervical lymph node (LN) in PTC patients, we investigated the risk factors for LNM in the central compartment and lateral compartment by using a large series of patients with surgically proven PTC. Besides, the correlation between LNM and recurrence-free survival (RFS) was also investigated.
Materials and methods
Patients
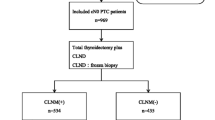
This retrospective study was approved by the Institutional Review Board of Changzhou First People’s Hospital. All participants gave written informed consent for their clinical records to be used in this study. A total of 487 patients with pathologically proven thyroid carcinoma who underwent thyroidectomy from January 2012 to April 2018 at the Changzhou First People’s Hospital were retrospectively reviewed from our department prospective surgical database. Patients were excluded from the study if they have any of the following factors: (1) had another malignancy before thyroidectomy; (2) reoperation; (3) non-PTCs (medullary/follicular/anaplastic) or mixed- type PTC; (4) distant metastasis at diagnosis; (5) had upper mediastinal node metastasis detected before thyroidectomy; (6) underwent non-curative surgery. In total, 432 patients were finally included and evaluated.
Surgical procedures
During the first clinical visit, body mass index (BMI) (kg/m2) was calculated via weight (kg) divided by height (m) squared. Based on the standardized guidelines set by the World Health Organization, enrolled PTC patients were categorized as normal (BMI < 25 kg/m2) and overweight (25 ≤ BMI kg/m2). We used the following protocol for surgical procedures (Fig. 1). All patients underwent ultrasonography (US), fine-needle aspiration (FNA) or Computed tomography (CT) to evaluate their primary lesions and lymph node metastasis. For clinically positive LLNM or intraoperative suspected LLNM, total thyroidectomy (TT) with CND and therapeutic ipsilateral LND was undertaken. LND was not performed in cN0 PTC patients (absence of any pre- or intra-operative evidence of lymph node disease), while prophylactic CND was performed in all cN0 PTC patients. Unilateral thyroid lobectomy and CND were performed for unilateral PTC patients while TT and CND were performed for bilateral PTC patients. LND was performed in the usual fashion from at least level II to level V, sparing the internal jugular vein, spinal accessory nerve, and sternocleidomastoid muscle [14]. CND included prelaryngeal, pretracheal and both the right and left paratracheal nodal basins [15]. All lymph nodes specimens were separated by the surgeon according to neck levels, and were sent to the department of pathology for paraffin fixation and histological analysis.
Histopathologic examination of surgical specimens
Two or more experienced pathologists microscopically reviewed and cross-checked all pathology specimens. Primary tumor characteristics were assessed, including histologic subtype, primary tumor size (measured as the longest diameter of the largest lesion), extrathyroidal invasion (defined as neoplastic infiltration beyond the thyroid fibrous capsule), multifocality (defined as two or more PTC lesions in a single lobe), vascular invasion, regional LNM and BRAF mutation. Micro-PTC was defined as PTC ≤ 10 mm in its maximum diameter while Macro-PTC was PTC > 10 mm in its maximum diameter. Tumor-node-metastasis (TNM) staging was based on the American Joint Committee on Cancer, 7th edition [16]. Recurrence was defined as new evidence of pathologically proven recurrence in the thyroid bed, soft tissue, cervical LN, or other organs on cytology from aspiration biopsy or reoperation.
Postoperative complications
Fiberoptic laryngoscopy was performed preoperatively and postoperatively in all patients to evaluate the mobility of vocal cord. Transient vocal cord palsy was defined as decreased or absent vocal cord mobility resolving within 6 months of surgery. If vocal cord palsy lasted for more than 6 months, it was considered permanent which confirmed by laryngoscopy. Serum calcium and phosphorus concentrations were measured in all patients after surgery. Transient hypocalcemia was defined as an ionized calcium level < 2.10 mmol/L during the hospital stay and the calcium level recovered normal within 6 months. Permanent hypoparathyroidism was diagnosed in patients still requiring calcium supplementation more than 6 months after surgery. If the patient has postoperative incision bleeding that affected breathing, emergency surgery was performed.
Postoperative management and follow-up
Postoperative suppressive levothyroxine (LT4) treatment was administered to all patients. Thyroid-stimulating hormone (TSH) suppression therapy (serum TSH level below 0.5 mIU/L) with LT4 with or without radioactive iodine (RAI) ablation was used for patients underwent total thyroidectomy. Physical examinations, ultrasonography of the neck and serum thyroglobulin (Tg) with Tg antibodies were used for all patients every 6 months for 2 years, and annually thereafter. Further imaging examinations or histological confirmation were used when the level of Tg and/or Tg antibodies significantly elevated. Follow-up data were obtained by outpatient consultations or telephone contact.
Statistical analyses
All statistical analyzes were carried out using the SPSS v 25.0 software (Chicago, IL, USA). The continuous variables were expressed as the means ± standard deviations (SD). The 2 test or Fisher’s exact test was used, as appropriate, for categorical data, whereas continuous variables were compared with Student t tests or the Manne–Whiney U test. Univariate analyzes for the associations between LN metastases and several clinicopathologic factors of the patients were performed using Pearson’s chi-square test or Fisher’s exact test. Binary logistic regression test was used for multivariate analysis of statistically significant variables from the univariate analysis. Univariate analysis of RFS was realized including each risk factor in a Cox regression model. RFS curves were calculated using the Kaplan–Meier method, and the log-rank test was used to evaluate the differences between curves.
Results
Baseline clinicopathological characteristics of PTC patients
The key clinicopathological characteristics were summarized in Table 1. Our study included 105 men (24.3%) and 327 women (75.7%), with a mean ± SD (Standard Deviation) age of 45.1 ± 12.2 years (range 19–80 years). The mean ± SD size of the primary thyroid tumor was 1.22 ± 0.93 cm (range 0.3–7.5 cm). The extrathyroidal invasion and vascular invasion were seen in 66 and 25 patients, respectively. Multifocality was seen in 118 cases.
LLNM were clinically detected in 61 patients before surgery while 19 patients were suspected of LLNM during surgery, and LND was performed in these patients (18.5%).
The remaining 352 patients (81.5%) underwent CND only. The post-operative examination confirmed that: 206 patients (47.7%) had nodes removed without metastases,161 patients (37.3%) had CLNM only, 55 patients (12.7%) had both CLNM and LLNM, and 10 patients (2.3%) had the skip metastases (LLNM without CLNM). In the central compartment, the mean ± SD number of excised and metastatic LNs was 6.14 ± 4.40 (range 1–31) and 2.42 ± 1.53 (range 0–18), respectively. The mean ± SD number of excised and metastatic lymph nodes in the lateral compartment was 18.75 ± 11.57 (range 0–51) and 4.95 ± 4.75 (range 0–22), respectively.
Tumor stages were as follows: T1 in 218 (50.5%); T2 in 138 (31.9%); T3 in 10 (2.3%); and T4 in 66 (15.3%) patients. Nodal stages were as follows: N0 in 206 (47.7%); N1 in 226 (52.3%) patients. Of 64 PTC patients who were performed BRAF mutation analysis, 56 (87.5%) had BRAF mutation positivity.
Predictors of CLNM
Table 2 shows the association between CLNM and several risk factors in PTC patients. In the univariate analysis, the rate of CLNM was significantly higher in male patients and patients < 45 years of age (P = 0.033, P = 0.004, respectively). Moreover, tumor size more than 1cm, presence of extrathyroidal invasion, vascular invasion and LLNM were significantly associated with CLNM (P < 0.001, P = 0.002, P < 0.001, respectively). Besides, tumor with higher T classification and TNM staging were significantly associated with a high prevalence of CLNM (both P < 0.001). In the multivariate analysis, patients < 45 years of age (OR 2.037, 95% CI 1.388–2.988, P < 0.001), presence of extrathyroidal invasion (OR 2.144, 95% CI 0.824–5.457, P = 0.011), presence of vascular invasion (OR 13.817, 95% CI 1.694–112.693, P = 0.014), presence of LLNM (OR 2.851, 95% CI 1.196–6.797, P = 0.014) and TNM Stage III–IV (OR 465.307, 95% CI 113.903–1900.826, P < 0.001) were i ndependent predictors of high prevalence of CLNM.
Predictors of LLNM
Univariate analysis showed that tumor size more than 1cm, presence of extrathyroidal invasion, vascular invasion and CLNM were significantly associated with LLNM (P < 0.001, P = 0.031, P < 0.001, respectively). Moreover, tumor with higher T classification and TNM staging were also significantly associated with a high prevalence of LLNM (both P < 0.001) (Table 3). As a result of multivariate analysis, tumor size more than 1cm (OR 3.474, 95% CI 1.728–6.985, P < 0.001) and CLNM (OR 5.532, 95% CI 2.679–11.425, P < 0.001) were independent predictive factors of LLNM (Table 3).
Postoperative evaluation
As shown in Table 4, 73 of the 352 patients (20.7%) who underwent thyroidectomy and CND developed postoperative complications, which significantly less than those (31.3%) who underwent thyroidectomy combined with CND and LND (P = 0.043). Vocal cord paralysis was observed in 53 patients (15.1%) who underwent thyroidectomy and CND. Of these patients, 2 (0.6%) had permanent vocal cord paralysis. For 80 patients who underwent thyroidectomy combined with CND and LND, 20 (25.0%) developed vocal cord paralysis, and 5 (6.3%) had had permanent vocal cord paralysis. In patients who underwent thyroidectomy and CND, 13 (3.7%) developed postoperative hypocalcemia requiring calcium supplement, and 3 (0.9%) had permanent hypocalcemia requiring calcium supplement for more than 6 months after surgery. While 9 of 80 patients (11.3%) who underwent thyroidectomy combined with CND and LND had postoperative hypocalcemia, and 6 (7.5%) developed permanent hypocalcemia. Three patients (0.9%) who underwent thyroidectomy and CND required emergency surgery due to immediate postoperative bleeding, which significantly less than the patients (5.0%) who underwent thyroidectomy combined with CND and LND. There were no postoperative deaths.
Follow-up and recurrences
Postoperative follow-up ranged from 6 to 89 months (average follow-up period: 42 months). During follow-up, 50 patients (11.6%) developed recurrent disease, including 28 patients (6.5%) had cervical lymph nodes recurrence, 14 patients (3.2%) had thyroid bed recurrence and 8 patients (1.9%) had lung recurrence (Table 1). The rate of recurrence in patients with CLNM was 16.2%, which was significantly higher than 6.9% in patients without CLNM (P = 0.003). The prevalence of LNs recurrence was significantly higher in patients with CLNM than patients without CLNM (9.7%, 3.2%, respectively, P = 0.006). Despite the rate of recurrence in patients with LLNM was higher than patients without LLNM (18.5%, 10.4%, respectively), there was no statistical difference (P = 0.060) (Table 5).
Predictors of RFS
Univariate analyzes in relation to RFS was conducted to determine the single variable which influenced risk of recurrence (Table 6). Tumor with T3–T4 stage, presence of extrathyroidal invasion and CLNM were the significant factors related to the RFS (P < 0.001, P < 0.001, P = 0.012, respectively), while other investigated variables had no influence on RFS. Patients with extrathyroidal invasion had 3.945 times higher risk of recurrence than patients without extrathyroidal invasion (Table 6 and Fig. 2). Patients with CLNM had 3.120 times higher risk of recurrence than patients without CLNM (Table 6 and Fig. 3). Similarly, risk of recurrence was 3.394 times higher among patients with T3–T4 stage (Table 6 and Fig. 4).
Discussion
Several studies have confirmed the metastatic lymph node size, number and ratio may have influence on locoregional recurrence in patients with PTC [17, 18, 19]. However, the association between LNM and primary tumor characteristics has not been fully established. Prophylactic lymph node dissection has the potential to reduce recurrence and survival, but may lead to clinically important postoperative morbidity [12, 13]. Hence, the associations between primary tumor characteristics and LNM were analyzed to improve the individualized treatment and to avoid the unnecessary prophylactic lymph node dissection.
The prevalence of overall LNM in this study was 52.3%, including 37.3% in CLNM, 12.7% in CLNM and LLNM, and 2.3% in the skip metastases, which conforms to incidence of LNM previously reported [3, 4, 5]. In the present study, the LNM pattern according to neck level was similar to that reported previously [20]. Cervical LNM occurs first at the node of the central compartment and then at the node of the lateral compartment, while the skipping metastasis is rare. Considering the fact that reoperation for recurrence of the central compartment may cause significant complications such as recurrent laryngeal nerve damage and persistent hypoparathyroidism, Japanese Society of Thyroid Surgeons (JSTS) recommend to routinely perform the prophylactic CND [21]. Although we did not conduct prospective studies to demonstrate whether conventional prophylactic CND could improve prognosis, we found that CLNM is a poor prognostic factor for PTC patients. Patients with CLNM had 3.12 times higher risk of recurrence than patients without CLNM. In addition, in a subgroup analysis, we found that patients with CLNM are prone to recurrence in cervical LNs. Yuan et al. [22] suggested that risk factors affecting CLNM were less than 45 years old, greater than 2cm of tumor size and bilaterality, while the effect of extrathyroidal invasion and vascular invasion had no significant difference. Koo et al. [23] considered that risk factors affecting CLNM were age, thyroid capsule invasion, greater than 1 cm of tumor size and lymphatic invasion. In this study, we found that patients aged less than 45 years, presence of extrathyroidal invasion, presence of vascular invasion, presence of LLNM, or having TNM stage III–IV, would have a high prevalence of CLNM. A part of these risk factors could be pre-operatively identified with US. For example, extrathyroidal invasion could be detected by pre-operative US with a sensitivity of 78.5–80.1% [24]. Besides, massive extrathyroidal invasion could be also detected intraoperatively. Considering the low sensitivity (51–58.3% only) of neck US for detecting LNM in central compartment [25, 26], we conclude that careful prophylactic CND should be recommended for PTC patients with above risk factors.
Although the second most common compartment for LNM is the lateral neck, none of the leading guidelines recommended prophylactic LND, moreover, the indication for prophylactic LND remained controversial [10, 11, 21]. In our study, we showed that the incidence of LLNM increased with the tumor size and presence of CLNM. On the contrary, the presence of LNM in lateral compartment does not increase the risk of recurrence of PTC in a series of cN0 or cN1 patients. Compared with patients without LLNM, patients with LLNM had higher risk of recurrence in cervical LNs, thyroid bed and lung, but there was no statistical difference, which was consistent with the result of Nobuyuki et al. [27] that patients undergoing prophylactic LND had the same recurrence rate as those who did not. This may attribute to the lymphatic drainage characteristics of central compartment, which was broader and multidirectional than lateral compartment [28]. From the perspective of quality of postoperative life, LND, especially level V lymphadenectomy, may lead to postoperative morbidity associated with the spinal accessory nerve and postoperative dysfunction in the strength of the trapezius muscle secondary to traction, devascularization, or thermal injury. Moreover, patients who underwent LND had a higher prevalence of permanent vocal cord palsy, permanent hypocalcaemia and emergency surgery than those with CND only. Therefore, from the perspective of postoperative recurrence, survival and quality of life, we recommend to perform LND only for patients with LLNM instead of performing prophylactic LND for high risk patients without LLNM.
Various factors and the staging system have been used to predict the prognosis in patients with PTC. Age, tumor size, extrathyroidal invasion, and neck LNM were generally accepted variables that were related to survival and recurrence [29, 30, 31]. In our data, apart from the CLNM, presence of extrathyroidal invasion and tumor with T3–T4 stage were significantly associated with RFS. It was not surprising to identify association between T classification and RFS. The risk of recurrence in patients with extrathyroidal invasion was 3.945 times higher than that in patients without extrathyroidal invasion. Similarly, many authors also found that patients with extrathyroidal invasion seemed to have higher mortality and recurrence rates than those without. Arora t al. [29] presumed that the extrathyroidal invasion itself had a negative effect on survival, while Clain et al. [30] and Noguchi et al. [31] showed that extrathyroidal invasion was associated with extranodal extension in LNM and had an effect on the survival rate. In our study, patients with extrathyroidal invasion were 2.144 times more likely to have CLNM than those without extrathyroidal invasion. The association between extrathyroidal invasion and CLNM may provide a theoretical basis for managing LNs in patients with PTC.
There are several potential limitations in our study. First, our study cohort was a retrospective study, and all data were collected from a single medical center, features of a nonrandomized would be produced inevitably. Second, we lacked information on other exposures, the possibility of residual confounding variables of measured or unmeasured factors were not ruled out. Third, different surgeons participated in the management of patients, there may have be differences in performing thyroidectomy or lymph node dissection.
In conclusion, our study demonstrated that age, extrathyroidal invasion, vascular invasion, LLNM and TNM stage were the important predictive factors for CLNM in PTC patients, while tumor size and CLNM were the predictive factors for LLNM. Therefore, for patients with pre–operative risk factors of LNM, an accurate preoperative evaluation of central compartment or lateral compartment is needed to find suspicious LNs. And prophylactic CND should be performed in patients with high risk of CLNM. Moreover, CLNM, extrathyroidal invasion and T classification were significantly associated with RFS. We suggest performing close follow-up for patients with high risk of RFS.
References
Lim H, Devesa SS, Sosa JA, et al. rends in thyroid cancer incidence and mortality in the United States, 1974–2013. JAMA. 2017;317(13):1338–48.
Morris LGT, Sikora AG, Tosteson TD, et al. The increasing incidence of thyroid cancer: the influence of access to care. Thyroid. 2013;23(7):885–91.
Mazzaferri EL, Jhiang SM. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med. 1994;97(5):418–28.
Shaha A. Prognostic factors in papillary thyroid carcinoma and implications of large nodal metastasis. Surgery. 2004;135(2):237–9.
Kyu Eun L, Il Yong C, Eunyoung K, et al. Ipsilateral and contralateral central lymph node metastasis in papillary thyroid cancer: patterns and predictive factors of nodal metastasis. Head Neck. 2013;35(5):672–6.
Grogan RH, Kaplan SP, Cao H, et al. A study of recurrence and death from papillary thyroid cancer with 27 years of median follow-up. Surgery. 2013;154(6):1436–47.
Beasley NJ, Lee J, Eski S, et al. Impact of nodal metastases on prognosis in patients with well-differentiated thyroid cancer. Arch Otolaryngol Head Neck Surg. 2002;128(7):825.
Ito Y, Kudo T, Kobayashi K, et al. Prognostic factors for recurrence of papillary thyroid carcinoma in the lymph nodes, lung, and bone: analysis of 5768 patients with average 10-year follow-up. World J Surg. 2012;36(36):1274–8.
Carty SE, Cooper DS, Doherty GM, et al. Consensus statement on the terminology and classification of central neck dissection for thyroid cancer. Thyroid. 2009;19(11):1153–8.
Cooper DS, Doherty GM, Haugen BR, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19(11):1167–214.
Tuttle RM, Ball DW, Byrd D, et al. Thyroid carcinoma. J Natl Compr Canc Netw. 2010;8(11):1228–744.
Inoue H, Nibu K, Saito M, et al. Quality of life after neck dissection. Arch Otolaryngol Head Neck Surg. 2006;132(132):662–6.
Terrell JE, Welsh DE, Bradford CR, et al. Pain, quality of life, and spinal accessory nerve status after neck dissection. Laryngoscope. 2010;110(4):620–6.
Jr SB, Ferris RL, Goldenberg D, et al. American Thyroid Association consensus review and statement regarding the anatomy, terminology, and rationale for lateral neck dissection in differentiated thyroid cancer. Thyroid. 2012;22(5):501.
Group ATASW, Carty SE, Cooper DS, et al. Consensus statement on the terminology and classification of central neck dissection for thyroid cancer. Thyroid, 2009, 19(11):1153–1158.
Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol, 2010, 17(6):1471–1474.
Parameswaran R, Sohail A, Ezzat TM, et al. The number of positive lymph nodes in the central compartment has prognostic impact in papillary thyroid cancer. Langenbecks Arch Surg. 2013;398(3):377–82.
Ji JM, Jong Ho Y, Ji Min H, et al. The prognostic value of the metastatic lymph node ratio and maximal metastatic tumor size in pathological N1a papillary thyroid carcinoma. Eur J Endocrinol. 2013;168(2):219–25.
Wang LY, Palmer FL, Nixon IJ, et al. Central lymph node characteristics predictive of outcome in patients with differentiated thyroid cancer. Thyroid. 2014;24(12):1790–5.
Noguchi S, Noguchi A, Murakami N Papillary carcinoma of the thyroid. I. Developing pattern of metastasis. Cancer,2015,26(5):1053-1060.
Takami H, Ito Y, Okamoto T, et al. Therapeutic strategy for differentiated thyroid carcinoma in japan based on a newly established guideline managed by Japanese Society of Thyroid Surgeons and Japanese Association of Endocrine Surgeons. World J Surg. 2011;35(1):111–21.
Yuan J, Gang Z, Du J, et al. To identify predictors of central lymph node metastasis in patients with clinically node-negative conventional papillary thyroid carcinoma. Int J Endocrinol. 2016;2016:1–6.
Koo BS, Choi EC, Yoon YH, et al. Predictive factors for ipsilateral or contralateral central lymph node metastasis in unilateral papillary thyroid carcinoma. Ann Surg. 2009;249(5):840–4.
Kim H, Kim J-A, Son EJ, et al. Preoperative prediction of the extrathyroidal extension of papillary thyroid carcinoma with ultrasonography versus MRI: a retrospective cohort study. Int J Surg. 2014;12(5):544–8.
Kim E, Park JS, Son KR, et al. Preoperative diagnosis of cervical metastatic lymph nodes in papillary thyroid carcinoma: comparison of ultrasound, computed tomography, and combined ultrasound with computed tomography. Thyroid. 2008;18(4):411.
Guidoccio F, Grosso M, Orsini F, et al. Thyroid ultrasound and other imaging procedures in the pediatric age. Curr Pediatr Rev. 2016;12(4):253–64.
Nobuyuki W, Quan-Yang D, Kiminori S, et al. Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection. Ann Surg. 2003;237(3):399–407.
Mirallié E, Visset J, Sagan C, et al. Localization of cervical node metastasis of papillary thyroid carcinomaa. World J Surg. 1999;23(9):970–4.
Arora N, Turbendian HK, Scognamiglio T, et al. Extrathyroidal extension is not all equal: implications of macroscopic versus microscopic extent in papillary thyroid carcinoma. Surgery. 2008;144(6):942–8.
Clain JB, Sophie S, Laura DR, et al. Extrathyroidal extension predicts extranodal extension in patients with positive lymph nodes: an important association that may affect clinical management. Thyroid. 2014;24(6):951–7.
Noguchi S, Yamashita H, Uchino S, et al. Papillary microcarcinoma. World J Surg. 2008;32(5):747–53.
Acknowledgements
Bao-Qiang Wu took charge of conceiving and designing the study; Xing-Hai Yang were responsible for collecting the data and analyzing and interpreting the data; Jia-Wei Feng took charge of writing the manuscript; Dong-Lin Sun was responsible for providing critical revisions; approving the final version of the manuscript was in charge of Yong Jiang and Zhen Qu. Lei Qin, the english language editor, was responsible for correcting language and grammar issues.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
This manuscript has not been published nor submitted for publication elsewhere. All authors have contributed significantly, and agree with the content of the manuscript. The authors reported no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Ethical approval
This study has been approved by the Institutional Review Board of Changzhou First People’s Hospital ethics committee, and has been performed according to the ethical standards laid down in the 1964 Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Feng, JW., Yang, XH., Wu, BQ. et al. Predictive factors for central lymph node and lateral cervical lymph node metastases in papillary thyroid carcinoma. Clin Transl Oncol 21, 1482–1491 (2019). https://doi.org/10.1007/s12094-019-02076-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02076-0