Abstract
Purpose
Anti-thyroglobulin antibodies (TgAb) can be used as a surrogate tumor marker in the follow-up of papillary thyroid carcinoma (PTC). We try to determine if the change in TgAb levels in the first post-operative year is a good predictor of persistence/recurrence risk in TgAb-positive PTC patients.
Methods/patients
105 patients with PTC who underwent thyroidectomy between 1988 and 2014 were enrolled. We calculated the percentage of change in TgAb levels with the first measurement at 1–2 months after surgery and the second one at 12–14 months.
Results
TgAb negativization was observed in 29 patients (27.6%), a decrease of more than 50% was observed in 57 patients (54.3%), less than 50% in 12 patients (11.4%) and in 7 patients (6.7%) the TgAb level had increased. The percentage of persistence/recurrence was 0, 8.8, 16.7 and 71.4% in each group, respectively (p < 0.001). In the multivariate analysis, only the percentage of change in TgAb showed a significant association with the risk of persistence/recurrence, regardless of other factors such as age, size and TNM stages.
Conclusions
Changes in TgAb levels in the first year after surgery can predict the risk of persistence/recurrence of TgAb-positive PTC patients. Patients who achieved negativization of TgAb presented an excellent prognosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Serum thyroglobulin (Tg) is a useful biochemical marker in the post-operative surveillance of differentiated thyroid carcinoma (DTC), since it is only produced in the thyroid tissue [1, 2]. In the last decades, interest in anti-thyroglobulin antibodies (TgAb) have increased, from being a simple marker of thyroid autoimmunity to being necessary for the interpretation of Tg, since they interfere with its measurement [1,2,3]. The presence of TgAb commonly causes falsely low serum Tg measurements; therefore, a reliable detection of TgAb is necessary before evaluating the validity of a Tg measurement. In these cases, the current American Thyroid Association (ATA) guidelines recommend that serum TgAb should be measured in conjunction with serum Tg assay by an immunometric method [4].
The prevalence of TgAb is higher in women and increases with age. A higher frequency has also been observed following iodine supplementation in iodine-deficient areas (as well as autoimmune thyroid diseases such as Hashimoto’s thyroiditis) [5].
In the last years, the prevalence of TgAb has increased in patients with DTC, and the presence of these antibodies is approximately twofold higher in patients with DTC than that of the general population [6,7,8,9,10,11,12,13]. Some studies suggest that the presence of TgAb may be a risk factor for malignancy in patients with thyroid nodules [14, 15]. The prevalence of these antibodies is significantly higher in papillary thyroid cancer (PTC) than in follicular thyroid cancer [7]. In addition, the presence of TgAb shortly after thyroidectomy may be associated with an increased risk of persistence/recurrence of the disease and the higher the level of TgAb the higher the risk [9, 10, 16,17,18,19,20]. It should be noted that TgAb may rise transiently post-operatively as an apparent immune reaction to the surgery itself and may also rise after radioiodine (RAI) ablation therapy [11].
Since the concentration of TgAb depends on the amount of Tg-secreting thyroid tissue [21], monitoring the levels of these antibodies may be a surrogate marker in the follow-up of DTC in patients with TgAb. Destruction of thyroid tissue should considerably reduce or even eliminate the levels of these antibodies, since it should remove the antigenic stimulus that drives their production. Recent studies suggest that patients in whom TgAb concentration decreases more than 50% after surgery have very low risk of persistence/recurrence during the follow-up; in addition, the risk increases if the levels do not fall bellow 50% and it may be even higher if the TgAb concentration increases [22,23,24,25,26]. Following total thyroidectomy and RAI remnant ablation, TgAb levels usually disappear over a median of 3 years in patients without evidence of persistent disease [21].
The aim of this study is to assess whether the change in TgAb levels in the first post-operative year is a good predictor of the risk of persistence/recurrence in TgAb-positive PTC patients.
The secondary aims are to observe when the negativization of TgAb occurs in patients who achieve remission of the disease and if the higher levels are associated with a worse prognosis.
Materials and methods
We analyzed retrospectively the data of 105 patients with PTC and a positive serum TgAb titer at the first post-operative assay (1–2 months after surgery, and always before RAI therapy), who underwent a total thyroidectomy in our hospital between January 1988 and June 2014. Patients with other types of thyroid carcinoma and those in whom TgAb levels were not available were excluded.
99 patients (94.3%) received complementary treatment with 50–115 mCi RAI, following the recommendations of the ATA DTC management guidelines. One patient was treated with chemotherapy (0.9%) and other with radiotherapy (0.9%) in follow-up. TNM classification and staging were based on the American Joint Committee on Cancer Staging Manual, seventh edition 2010 [27].
The outcome of the primary treatment was assessed at 12–14 months after surgery with measurement of TgAb in all patients (including those who had also been evaluated earlier during the first year). We have calculated the percentage of change in the first year with the first measurement (1–2 months after surgery) and that of 12–14 months after surgery.
Subsequent follow-up visits were scheduled every 6–12 months. Each visit included measurement of serum Tg levels, TgAb and a neck ultrasound (US). Patients were defined as having no evidence of disease if they did not show residual cervical tissue detected by US or other imaging studies (computed tomography, magnetic resonance, or fluorodesoxyglucose positron emission tomography) when indicated. Suspicious neck lesions were confirmed by positive findings on fine-needle aspiration cytology (FNAC). Disease status was defined as persistent or recurrent only when lesions were observed by imaging studies (distant metastases) or by US and FNAC (thyroid bed or lymph node disease).
The frequency of persistence/recurrence was observed in four groups: those whose TgAb concentrations had been negativized in the first post-operative year, those who had it below 50% of its initial value, those who had it above 50%, and those whose TgAb concentration had increased.
This study was conducted in compliance with ethical principles based on the Declaration of Helsinki and with the approval of the ethics committee of our center. We used anonymous data for the research.
Laboratory
TgAb levels were measured by radioimmunoassay (RIA) until June 2000 (Brahms, Hennigsdorf/Berlin, Germany) and then using an automated chemiluminescence assay, Immulite 2000 system and Siemens kits (Siemens Healthcare Diagnostics Products Limited, Munich, Germany) after that date.
Statistical analysis
Quantitative variables are reported as mean and standard deviation except for variables that did not follow a normal distribution, such as TgAb values, for which the median and interquartile range were used. Absolute numbers and percentages are used to describe qualitative variables.
The comparison of quantitative variables between the groups was performed with ANOVA test and with Kruskal–Wallis test if the distribution was not normal or the variances were not homogeneous. A Chi square test was performed to compare qualitative variables, and a Fisher’s test was conducted if the expected frequency in any of the assumptions was less than five patients.
A logistic regression model was used to predict which factors were associated with the persistence/recurrence of the disease. For the multivariate analysis, we used the significant factors in the univariate analysis. The level of significance was set at p < 0.05. The results are shown with their 95% confidence limits.
All statistical analyses were performed using the SPSS statistical software (version 21.0; SPSS Institute, Chicago, IL, USA).
Results
Our cohort consisted of 105 patients with TgAb who underwent a total thyroidectomy for PTC. Baseline cohort characteristics are shown in Table 1. During the follow-up [median 8.2 years (range 1.5–26.9)], 12 patients (11.4%) showed persistence/recurrence. In 27.6% of the patients, TgAb negativization was observed in the first year and in 60.9% during the follow-up. A decrease of more than 50% in TgAb was observed in 57 patients (54.3%), 12 patients (11.4%) presented a decrease of less than 50% and in 7 patients (6.7%) TgAb had increased in the first year.
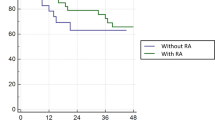
We observed statistically significant differences of persistence/recurrence in the four groups, according to the percentage of change of TgAb in the first year (Table 2). The risk of persistence/recurrence is especially high in the group in which the TgAb increased. Moreover, we did not observe any persistence/recurrence in the group that showed TgAb negativization (Fig. 1). There were also differences in TNM stage between the different groups, with a greater frequency of stage IV in the group in which the TgAb increased.
In this group, age and size were considerably, but not significantly, higher.
We found differences in TgAb levels among the different groups, with a higher median in the group where the decrease was greater than 50%.
As shown in Table 3, we observed a relationship between age, size, TNM stage, percentage of change in TgAb and a worse prognosis of the disease. However, in the multivariate analysis, only the percentage of change in TgAb showed a statistically significant association with the risk of persistence/recurrence. A 1% higher decrease in TgAb levels leads to a 1.6% (95% CI 0.02–3%) decrease in the risk of persistence/recurrence during the follow-up. We did not find differences between TgAb levels and a worse prognosis.
In patients in whom negativization of TgAb was achieved, this occurred in a median of 2 years (interquartile range 1–3 years) and, in two cases, this happened more than 10 years after surgery.
Discussion
In this study, we showed that the percentage of change in TgAb in the first year after thyroidectomy is one of the most important prognostic factors in patients with PTC and TgAb. In addition, we can assert that patients who negativize the TgAb in the first year have a practically null risk of presenting persistence/recurrence of the disease. In our study, patients with increased levels of TgAb presented more advanced stages of the disease, and a trend towards an older age, and larger tumors. However, regardless of these factors, we observed a statistically significant relationship between the percentage of change in TgAb and the risk of persistence/recurrence.
Several studies have demonstrated the relationship between the persistence of TgAb positivity and worse prognosis in PTC patients [9, 10, 22, 24]. Removal of all thyroid tissue, which eliminates the antigenic stimulus, should lead to a progressive decrease of TgAb and, finally, to its disappearance. The presence of persistently positive TgAb titers is considered as evidence of the existence of functional thyroid cells [21].
Tsushima et al. demonstrated that the lack of a decrease or increase of post-operative TgAb was an independent prognostic factor for disease-free survival with respect to conventional static prognostic factors such as tumor size, extrathyroid extension, and patient age [24]. Therefore, it might be better to use the change rate in the TgAb levels instead of specific TgAb levels to evaluate the disease condition of TgAb-positive PTC patients and to predict patient prognosis. Based on these data, they propose that post-operative changes in TgAb can be used as a surrogate tumor marker.
Our analysis found no prognostic significance of baseline TgAb status. Li et al. found a higher frequency of multifocality, capsular invasion and nodal infiltration in patients with TgAb ≥ 1; however, the influence of high TgAb levels on these factors was not analyzed [28].
The median time of negativization in patients who achieved complete remission was 2 years (interquartile range 1–3 years). Chiovato et al. reported that complete ablation of thyroid tissue by thyroidectomy and treatment with RAI resulted in the disappearance of antithyroid antibodies [21]. They reported that the median disappearance time was 6.3 years for thyroid peroxidase antibody and 3 years for TgAb. It is important to note that in our study, this occurred more than 10 years after treatment in two cases; thereby, the long-term follow-up of these patients is essential.
Our study has some limitations: first, this is a single-center retrospective study with a relatively small number of patients. Second, we used two TgAb assay methods during the entire study period.
In summary, we demonstrated that changes in TgAb in the first year after surgery can be an essential factor in the prognosis of TgAb-positive PTC patients. Based on our data, disappearance of TgAb titers within the first post-operative year seems to be associated with a null risk of persistence/recurrence of the disease.
References
Spencer CA. Editorial: challenges of serum thyroglobulin (Tg) measurement in the presence of Tg autoantibodies. J Clin Endocrinol Metab. 2004;89(8):3702–4.
Demers LM, Spencer CA. Laboratory medicine practice guidelines: laboratory support for the diagnosis and monitoring of thyroid disease. Clin Endocrinol. 2003;58(2):138–40.
Spencer C, Petrovic I, Fatemi S, LoPresti J. Serum thyroglobulin (Tg) monitoring of patients with differentiated thyroid cancer using sensitive (second-generation) immunometric assays can be disrupted by false-negative and false-positive serum thyroglobulin autoantibody misclassifications. J Clin Endocrinol Metab. 2014;99(12):4589–99.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133. doi:10.1089/thy.2015.0020.
Medeiros Neto G. Iodine supplementation and thyroid autoimmunity. Clin Endocrinol. 1994;40(3):435.
Pacini F, Mariotti S, Formica N, Elisei R, Anelli S, Capotorti E, et al. Thyroid autoantibodies in thyroid-cancer incidence and relationship with tumor outcome. Acta Endocrinol. 1988;119(3):373–80.
Kumar A, Shah DH, Shrihari U, Dandekar SR, Vijayan U, Sharma SM. Significance of antithyroglobulin autoantibodies in differentiated thyroid-carcinoma. Thyroid. 1994;4(2):199–202.
Spencer CA, Takeuchi M, Kazarosyan M, Wang CC, Guttler RB, Singer PA, et al. Serum thyroglobulin autoantibodies: prevalence, influence on serum thyroglobulin measurement, and prognostic significance in patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab. 1998;83(4):1121–7.
Rubello D, Casara D, Girelli ME, Piccolo M, Busnardo B. Clinical meaning of circulating antithyroglobulin antibodies in differentiated thyroid cancer. A prospective study. J Nucl Med. 1992;33(8):1478–80.
Chung JK, Park YJ, Kim TY, So Y, Kim SK, Park DJ, et al. Clinical significance of elevated level of serum antithyroglobulin antibody in patients with differentiated thyroid cancer after thyroid ablation. Clin Endocrinol. 2002;57(2):215–21.
Gorges R, Maniecki M, Jcntzen W, Sheu SNY, Mann K, Bockisch A, et al. Development and clinical impact of thyroglobulin antibodies in patients with differentiated thyroid carcinoma during the first 3 years after thyroidectomy. Eur J Endocrinol. 2005;153(1):49–55.
Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988–1994): national health and nutrition examination survey (NHANES III). J Clin Endocrinol Metab. 2002;87(2):489–99.
Qin J, Yu ZQ, Guan HX, Shi LF, Liu YP, Zhao N, et al. High thyroglobulin antibody levels increase the risk of differentiated thyroid carcinoma. Dis Markers. 2015; 2015:648–670.
Kim ES, Lim DJ, Baek KH, Lee JM, Kim MK, Kwon HS, et al. Thyroglobulin antibody is associated with increased cancer risk in thyroid nodules. Thyroid. 2010;20(8):885–91.
Grebe SKG. Thyroglobulin autoantibodies, thyroid nodules, and new insights into some old questions. Thyroid. 2010;20(8):841–2.
Phan HTT, Jager PL, van der Wal JE, Sluiter WJ, Plukker JTM, Dierckx R, et al. The follow-up of patients with differentiated thyroid cancer and undetectable thyroglobulin (Tg) and Tg antibodies during ablation. Eur J Endocrinol. 2008;158(1):77–83.
Soyluk O, Boztepe H, Aral F, Alagol F, Ozbey NC. Papillary thyroid carcinoma patients assessed to be at low or intermediary risk after primary treatment are at greater risk of long term recurrence if they are thyroglobulin antibody positive or do not have distinctly low thyroglobulin at initial assessment. Thyroid. 2011;21(12):1301–8.
Kim TY, Kim WB, Kim ES, Ryu JS, Yeo JS, Kim SC, et al. Serum thyroglobulin levels at the time of (131)I remnant ablation just after thyroidectomy are useful for early prediction of clinical recurrence in low-risk patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab. 2005;90(3):1440–5.
Seo JH, Lee SW, Ahn BC, Lee J. Recurrence detection in differentiated thyroid cancer patients with elevated serum level of antithyroglobulin antibody: special emphasis on using 18F-FDG PET/CT. Clin Endocrinol. 2010;72(4):558–63.
Durante C, Tognini S, Montesano T, Orlandi F, Torlontano M, Puxeddu E, et al. Clinical aggressiveness and long-term outcome in patients with papillary thyroid cancer and circulating anti-thyroglobulin autoantibodies. Thyroid. 2014;24(7):1139–45.
Chiovato L, Latrofa F, Braverman LE, Pacini F, Capezzone M, Masserini L, et al. Disappearance of humoral thyroid autoimmunity after complete removal of thyroid antigens. Ann Intern Med. 2003;139(5):346–51.
Kim WG, Yoon JH, Kim WB, Kim TY, Kim EY, Kim JM, et al. Change of serum antithyroglobulin antibody levels is useful for prediction of clinical recurrence in thyroglobulin-negative patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab. 2008;93(12):4683–9.
Spencer C, Fatemi S. Thyroglobulin antibody (TgAb) methods—strengths, pitfalls and clinical utility for monitoring TgAb-positive patients with differentiated thyroid cancer. Best Pract Res Clin Endocrinol Metab. 2013;27(5):701–12.
Tsushima Y, Miyauchi A, Ito Y, Kudo T, Masuoka H, Yabuta T, et al. Prognostic significance of changes in serum thyroglobulin antibody levels of pre- and post-total thyroidectomy in thyroglobulin antibody-positive papillary thyroid carcinoma patients. Endocr J. 2013;60(7):871–6.
Yamada O, Miyauchi A, Ito Y, Nakayama A, Yabuta T, Masuoka H, et al. Changes in serum thyroglobulin antibody levels as a dynamic prognostic factor for early-phase recurrence of thyroglobulin antibody-positive papillary thyroid carcinoma after total thyroidectomy. Endocr J. 2014;61(10):961–5.
Hsieh CJ, Wang PW. Sequential changes of serum antithyroglobulin antibody levels are a good predictor of disease activity in thyroglobulin-negative patients with papillary thyroid carcinoma. Thyroid. 2014;24(3):488–93.
Edge SB, Compton CC. The American joint committee on cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17(6):1471–4.
Li C, Yu W, Fan J, Li G, Tao X, Feng Y, et al. Thyroid functional parameters and correlative autoantibodies as prognostic factors for differentiated thyroid cancers. Oncotarget. 2016;7(31):49930–8.
Acknowledgements
We would like to thank our center for the technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights statement
The research involving human participants are conducted in a way which respects the dignity, rights, and welfare of participants.
Ethical approval
This study was conducted in compliance with ethical principles based on the Declaration of Helsinki and with the approval of the ethics committee of our center. We used anonymous data for the research.
Rights and permissions
About this article
Cite this article
Ernaga-Lorea, A., Hernández-Morhain, M.C., Anda-Apiñániz, E. et al. Prognostic value of change in anti-thyroglobulin antibodies after thyroidectomy in patients with papillary thyroid carcinoma. Clin Transl Oncol 20, 740–744 (2018). https://doi.org/10.1007/s12094-017-1782-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-017-1782-3