Abstract
Background
Data on treatment outcome and prognostic factors in patients with metastatic soft tissue sarcoma (STS) are limited in the literature.
Methods
A total of 119 patients with metastatic STS treated between June 2003 and December 2012 were analyzed for treatment outcome and prognostic factors.
Results
Median age was 37 years (range 2–72 years) with a male to female ratio of 1.5:1. Most common histologic subtypes were synovial sarcoma (36 %) and leiomyosarcoma (16 %). Median tumor size was 12 cm (range 1.6–30 cm). Twenty-four (20 %) patients were treated with multimodality therapy and 80 % patients received systemic chemotherapy alone. At a median follow-up of 10 months (range 1–66 months), the 2-year EFS and OS were 10 and 19 %, respectively, with a median EFS and OS of 6 and 10 months, respectively. Univariate analysis identified albumin ≤4 g/dl (p = 0.001), histologic subtypes other than synovial sarcoma (p = 0.02), non-extremity tumors (p = 0.03) and single modality treatment (p = 0.03) as factors predicting poor EFS; however, for OS, hemoglobin ≤10 g/dl (p = 0.02), tumor size >10 cm (p = 0.01) and single modality treatment (p = 0.04) were identified as poor prognostic factors. Multivariate analysis identified only serum albumin ≤4 g/dl (p = 0.002, HR 0.47, 95 % CI 0.29–0.75) associated with poor EFS; however, for OS, hemoglobin ≤10 g/dl (p = 0.009, HR 0.49, 95 % CI 0.29–0.83), tumor size >10 cm (p = 0.003, HR 2.11, 95 % CI 1.28–3.47) and single modality treatment (p = 0.01, HR 0.47, 95 % CI 0.25–0.86) emerged as poor prognostic factors.
Conclusions
Serum albumin, tumor size, hemoglobin and treatment modality affect survival in metastatic STS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
About 10 % patients with soft tissue sarcoma (STS) present with metastatic disease at baseline and 40–60 % patients with high-grade STS develop metastases despite curative local treatment. The median survival after diagnosis of distant metastases is 12 months [1–3]. The development of metastases poses a major clinical problem, as metastatic disease is seldom amenable to a curative treatment. Once metastases are detected, the treatment is based mainly on palliative chemotherapy which produces an overall response rate of about 25 % in the first-line setting [4, 5]. Surgical resection is well accepted as a standard approach for the treatment of metastases from STS isolated to the lungs, and many studies investigating this technique have reported an overall 5-year survival ranging from 30 to 40 % [6, 7].
Due to paucity of the literature specifically addressing the prognostic factors in metastatic STS, we aimed to study the outcome of patients with metastatic STS treated at our institute and identify prognostic factors affecting survival.
Materials and methods
This was a retrospective analytical study. Medical records of all patients with histologically proven STS treated between June 2003 and December 2012 at our cancer center were reviewed. Those with metastatic disease at presentation to our institute were identified and analyzed. Patients were included whether denovo with metastases or if they had developed metastases after previous treatment for their local site before presenting to our institute. Exclusion criteria were patients who had received chemotherapy before presenting to our institute, those who did not take treatment at our center, those who received ≤2 cycles of chemotherapy at our center and were lost to follow-up without progression or event and patients with rhabdomyosarcoma. The histologic diagnosis of all tumors diagnosed outside our centre was confirmed by members of the pathology department of our institute. The tumor characteristics included tumor size, primary tumor site, histology, grade and sites of metastatic disease.
Evaluation of primary lesion was done by magnetic resonance imaging (MRI) or contrast-enhanced computed tomography (CECT) depending on the site of lesion. Metastatic workup included CECT scan of thorax, abdomen and pelvis and bone scan wherever required.
Treatment and response evaluation
Treatment consisted of systemic chemotherapy, surgery and radiotherapy. Systemic chemotherapy was the primary therapy given to all patients and different regimens were used depending on the tumor histology. Patients who had complete response (CR) or partial response (PR) at primary site and/or metastatic sites after chemotherapy were taken up for local treatment either in the form of surgery or radiotherapy or both, and surgery of the metastatic sites wherever feasible. The choice of local therapy was made on an individual basis depending on primary tumor site and resectability of tumor after chemotherapy with care to avoid long-term morbidity and disfigurement.
Response to chemotherapy was assessed radiologically after 2 cycles and at completion of therapy. Complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) were defined according to Revised Response Evaluation Criteria in Solid Tumors version 1.1 [8].
Survival analysis and statistics
Patients, tumor and treatment variables were analyzed for their relation to outcome. Any event and death from any cause were used as the end points in this study. Event-free survival (EFS) was calculated from the date of diagnosis to the date of disease progression or death from any cause. Overall survival (OS) was calculated from the date of diagnosis to the date of death from any cause. All times were calculated from the first registration date to our institute to the last day of follow-up or death from any cause. Data were censored on 31 December 2014. Actuarial survival was determined by Kaplan–Meier analysis. The relations of patient, tumor, and treatment characteristics to outcome were tested by univariate analysis using log-rank. P value of ≤0.05 was considered statistically significant. Multivariate analysis was performed using Cox proportional hazards model, and only variables that were statistically significant in univariate analysis were included in the final Cox model. Patients who were lost to follow-up (LFU) or had treatment abandonment after 2 cycles of chemotherapy were also included for EFS and OS analysis and outcome in these patients was confirmed by telephonic contact. Treatment abandonment was included for survival analysis in the present study as it has been proposed that patients who were non-compliant and had treatment abandonment should be included in survival analysis for studies from developing nations to provide a true picture of outcome from these countries [9]. STATA/SE 9.0 (StataCorp LP, Texas, USA) was used for statistical analysis.
Results
Clinicopathologic profile
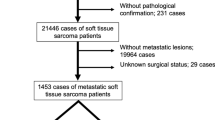
Of 950 patients of STS, 215 (23 %) patients presented with metastatic disease. Ninety-six patients were excluded (40 patients who did not take treatment, 45 patients with rhabdomyosarcoma and 11 patients who took ≤2 cycles of chemotherapy). So, here we have analyzed 119 patients and their clinicopathologic characteristics are summarized in Table 1. Median age was 37 years (range 2–72 years) with a male:female ratio of 1.5:1. Median duration of symptoms was 7 months (range 1–120 months). The primary tumor sites were extremity in 54 %, trunk in 18 %, retroperitoneum in 15 %, visceral in 10 % and head–neck in 3 % patients. Most common histologic subtypes were synovial sarcoma in 36 % and leiomyosarcoma in 16 % patients. Other histologic subtypes constituted 48 % of all patients which are shown in Supplementary Table 1. Median tumor size was 12 cm (range 1.6–30 cm) and 109 (92 %) patients had tumor size >5 cm.
Metastatic sites
One hundred and two (86 %) patients had metastases in lungs. Seventy-six (64 %) patients had metastases in lungs alone and 26 (22 %) patients had lung metastases in combination with other sites. Sixteen (13 %) patients had lymph node metastases. Out of 16 (13 %) patients with liver metastases, liver-only metastases were found in 4 patients and combination with other sites was found in 12 patients. Bone metastases were present in 11 (9 %) patients, 2 had bone metastases alone and 9 had in combination with other sites. Other rare sites of metastases were adrenals in 2 patients, brain in 1 patient and skin in 1 patient.
Treatment
Twenty-four (20 %) patients were treated with multimodality therapy and 95 (80 %) patients received single modality, i.e. systemic chemotherapy alone.
Chemotherapy
Systemic chemotherapy was given to all patients with palliative intent and patients having CR/PR at primary or metastatic sites were taken for surgery. The chemotherapeutic regimens used are shown in Supplementary Table 2. Most common regimen consisted of a combination of ifosfamide and doxorubicin in 55 % patients followed by single agent doxorubicin in 31 % patients.
Surgery
Surgery was done in 24 (20 %) patients, out of which 10 patients underwent wide local excisions and amputation was performed on 14 patients. Metastasectomy for lung metastases was performed on 6 patients.
Radiotherapy
Eighteen (15 %) patients received external beam radiotherapy. Definitive radiotherapy in the dose of 45–60 Gy was received by 4 patients and palliative radiotherapy in the dose of 8–36 Gy was received by 14 patients.
Response and toxicity
Overall, 9 (8 %) patients achieved CR, 18 (15 %) patients achieved PR and 38 (32 %) patients had SD. The overall response rate with chemotherapy was 23 % (including CR and PR). Fifty-four (45 %) patients developed progression on treatment. Twenty-one (18 %) patients developed Grade I and II and 3 (2.5 %) patients developed Grade III and IV toxicities. The most common toxicity was myelosuppression in 18 patients and 1 patient died due to febrile neutropenia.
Survival
At the end of last follow-up, 107 patients were dead (98.5 % deaths due to disease progression), 6 patients were alive (1 in CR and 5 with disease) and follow-up information for 6 patients was not known.
Median follow-up was 10 months (range 1–66 months). The 2-year EFS and OS were 10 and 19 %, respectively, with a median EFS of 6 months and a median OS of 10 months (Supplementary Figs. 1, 2).
Prognostic factors
Univariate analysis
For EFS, albumin ≤4 g/dl (p = 0.001), histologic subtypes other than synovial sarcoma (p = 0.02), non-extremity tumors (p = 0.03) and single modality treatment (p = 0.03) emerged as poor prognostic factors. However, for OS, univariate analysis identified hemoglobin ≤10 g/dl (p = 0.02), tumor size >10 cm (p = 0.01) and single modality treatment (p = 0.04) as poor prognostic factors (Table 2).
Multivariate analysis
Multivariate analysis for EFS identified only serum albumin ≤4 g/dl (p = 0.002, HR 0.47, 95 % CI 0.29–0.75) associated with poor EFS (Fig. 1), whereas for OS, tumor size >10 cm (p = 0.003), hemoglobin ≤10 g/dl (p = 0.009) and single modality treatment (p = 0.01) emerged as poor prognostic factors (Table 3) (Fig. 2a, b, c).
Discussion
The median survival of our cohort was 10 months with 2-year OS of 19 % which is comparable to the published literature [1, 10, 11]. Ifosfamide and doxorubicin combination was most commonly used in our study and an overall response rate of 23 % was found which is consistent with the literature [12–17]. Due to varied histologic subtypes in our cohort with small number of patients in each group, it is difficult to make statistical comparisons between different chemotherapeutic regimens. Most studies on STS are severely hampered by the admixture of a variety of histologic subtypes, making it difficult to assess the clinical activity of any given treatment.
Surgical resection for the treatment of pulmonary metastases has shown improvement in 5-year survival ranging from 30 to 40 % [6, 7, 11, 18]. Since the number of patients undergoing pulmonary metastasectomy in our study was very less, it is difficult to draw any conclusion on its benefit.
Little is known about the prognostic factors in metastatic STS. There is only one previous study by Van Glabbeke et al. [1] specifically addressing this cohort of patients which recognized factors such as extrapulmonary metastases and histologies like leiomyosarcoma and malignant fibrous histiocytoma to be associated with poor prognosis whereas good performance status, absence of liver metastases, low histopathologic grade, long time since diagnosis of the primary tumor, few metastases, and young age had favorable outcome [1]. We found low serum albumin, tumor size >5 cm, low hemoglobin and single modality treatment as adverse prognostic factors in this group.
Serum albumin has been described as an independent prognosticator of survival in various cancers [19–23]. A study by Barreto-Andrade et al. [24] analyzing a group of 61 patients with STS found serum albumin as independent prognostic factor of survival in these patients. Another study by Biswas et al. [25] done in metastatic Ewing’s sarcoma found low serum albumin as a poor prognosticator of disease outcome. Our study confirms the association of low serum albumin level with poor survival in metastatic STS.
Tumor size is well recognized as an independent prognostic indicator in localized STS and is a determinant of both local recurrence as well as metastatic disease [3, 24, 26, 27]. Bauer et al. [3] found larger primary tumors in patients with metastatic STS than patients with localized disease: median 11 cm versus 7 cm, respectively; however, the prognostic impact of tumor size in metastatic STS was not found. Our study is the first of its kind which establishes tumor size as an independent prognostic factor for metastatic disease. The relatively poorer survival in our cohort might be explained by large tumor size; notably 98 % of this cohort had tumor size >5 cm and the median tumor diameter was 12 cm.
Low hemoglobin level before surgery was found to have negative prognostic impact on clinical outcome in patients with localized STS in a study by Szkandera J et al. [28]. Our study confirms the poor impact of low baseline hemoglobin on survival in metastatic disease as well.
The benefit of surgery for primary site in metastatic STS is not clear. However, clear benefit of treatment for primary tumor site was found in our study as patients who underwent multimodality treatment had better survival rates than patients who received systemic chemotherapy alone (28 vs 17 %, p value = 0.04). This study emphasizes upon addressing the primary tumor site in metastatic disease as well either by surgery or radiotherapy. However, more studies are required to draw any meaningful conclusion.
In conclusion, this is a large single-institutional study on metastatic STS which identifies serum albumin, tumor size, hemoglobin and treatment modality as factors predictive of outcome in metastatic disease.
References
Van Glabbeke M, van Oosteroom A, Oosterhuis J, Mouridsen H, Crowther D, Somers R, et al. Prognostic factors for the outcome of chemotherapy in advanced soft tissue sarcoma; an analysis of 2,185 patients treated with anthracycline-containing first-line regimens ñ a European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol. 1999;17:150–7.
Coindre JM, Terrier P, Guillou L, Le Doussal V, Collin F, Ranchere D, et al. Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas. Cancer. 2001;91:1914–26.
Bauer HC, Trovik CS, Alvegard TA, Berlin O, Erlanson M, Gustafson M, et al. Monitoring referral and treatment in soft tissue sarcoma: study based on 1851 patients from the Scandinavian Sarcoma Group Register. Acta Orthop Scand. 2001;72:150–9.
Karavasilis V, Seddon BM, Ashley S, Al-Muderis O, Fisher C, Judson I. Significant clinical benefit of first-line palliative chemotherapy in advanced soft-tissue sarcoma: retrospective analysis and identification of prognostic factors in 488 patients. Cancer. 2008;112:1585–91.
Santoro A, Tursz T, Mouridsen H, Verweij J, Steward W, Somers R, et al. Doxorubicin versus CYVADIC versus doxorubicin plus ifosfamide in first-line treatment of advanced soft tissue sarcomas: a randomized study of the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. J Clin Oncol. 1995;13:1537–45.
Billingsley KG, Burt ME, Jara E. Pulmonary metastases from soft tissue sarcoma: analysis of patterns of diseases and postmetastasis survival. Ann Surg. 1999;229:602–10.
Pastorino U, Buyse M, Frielder G. Long- term results of lung metastasectomy: prognostic analyses based on 5206 cases. J Thorac Cardiovasc Surg. 1997;113:31–49.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Mostert S, Arora RS, Arreola M, Bagai P, Friedrich P, Gupta S, et al. Abandonment of treatment for childhood cancer: position statement of a SIOP PODC Working Group. Lancet Oncol. 2011;12(8):719–20. doi:10.1016/S1470-2045(11)70128-0.
Italiano A, Mathoulin-Pelissier S, Cesne AL, Terrier P, Bonvalot S, Collin F, et al. Trends in survival for patients with metastatic soft-tissue sarcoma. Cancer. 2011;117:1049–54.
Bedi M, King DM, Charlson J, Whitfield R, Hackbarth DA, Zambrano EV, et al. Multimodality management of metastatic patients with soft tissue sarcomas may prolong survival. Am J Clin Oncol. 2014;37:272–7.
Sleijfer S, Seynaeve C, Verweij J. Using single-agent therapy in adult patients with advanced soft tissue sarcoma can still be considered standard care. Oncologist. 2005;10:833–41.
Penel N, Bui BN, Bay JO, Cupissol D, Ray-Coquard I, Piperno-Neumann S, et al. Phase II trial of weekly paclitaxel for unresectable angiosarcoma: the ANGIOTAX study. J Clin Oncol. 2008;26:5269–74.
Patel SR, Gandhi V, Jenkins J, Papadopolous N, Burgess MA, Plager C, et al. Phase II clinical investigation of gemcitabine in advanced soft tissue sarcomas and window evaluation of dose rate on gemcitabine triphosphate accumulation. J Clin Oncol. 2001;19:3483–9.
Grosso F, Jones RL, Demetri GD, Judson IR, Blay JY, Le Cesne A, et al. Efficacy of trabectedin (ecteinascidin-743) in advanced pretreated myxoid liposarcomas: a retrospective study. Lancet Oncol. 2007;8:595–602.
Santoro A. Advanced soft tissue sarcoma: how many more trials with anthracyclines and ifosfamide? Ann Oncol. 1999;10:151–4.
Bramwell VH, Anderson D, Charette ML, Sarcoma Disease Site Group. Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev. 2003;(3):CD003293.
Donaldson SS, Meza J, Breneman JC, Crist WM, Laurie F, Qualman SJ, et al. Results from the IRS-IV randomized trial of hyperfractionated radiotherapy in children with rhabdomyosarcoma–a report from the IRSG. Int J Radiat Oncol Biol Phys. 2001;51:718–28.
Ballmer PE, Ochsenbein AF, Schutz-Hofmann S. Transcapillary escape rate of albumin positively correlates with plasma albumin concentration in acute but not in chronic inflammatory disease. Metabolism. 1994;43:697–705.
Maltoni M, Amadori D. Prognosis in advanced cancer. Hematol Oncol Clin North Am. 2002;16:715–29.
Onate-Ocana LF, Aiello-Crocifoglio V, Gallardo-Rincon D, Herrera-Goepfert R, Brom-Valladares R, Carrillo JF, et al. Serum albumin as a significant prognostic factor for patients with gastric carcinoma. Ann Surg Oncol. 2007;14:381–9.
Heys SD, Walker LG, Deehan DJ, Eremin OE. Serum albumin: a prognostic indicator in patients with colorectal cancer. J R Coll Surg Edinb. 1998;43:163–8.
Seve P, Ray-Coquard I, Trillet-Lenoir V, Sawyer M, Hanson J, Broussolle C, et al. Low serum albumin levels and liver metastasis are powerful prognostic markers for survival in patients with carcinomas of unknown primary site. Cancer. 2006;107:2698–705.
Barreto-Andrade JC, Medina-Franco H. Serum albumin is an independent prognostic factor for survival in soft tissue sarcomas. Rev Invest Clin. 2009;61:198–204.
Biswas B, Agarwala S, Rastogi S, Khan SA, Mohanti BK, Sharma DN, et al. High burden of metastases and poor outcome in pelvic PNET. Pediatr Blood Cancer. 2013;60(9):E97–9. doi:10.1002/pbc.24552.
Coindre JM, Terrier P, Bui NB, Bonichon F, Collin F, Le Doussal V, et al. Prognostic factors in adult patients with locally controlled soft tissue sarcoma. A study of 546 patients from the French Federation of Cancer Centers Sarcoma Group. J Clin Oncol. 1996;14:869–77.
Pisters PW, Leung DH, Woodruff J, Shi W, Brennan MF. Analysis of prognostic factors in 1,041 patients with localized soft tissue sarcomas of the extremities. J Clin Oncol. 1996;14:1679–89.
Szkandera J, Gerger A, Liegl-Atzwanger B, Stotz M, Samonigg H, Ploner F, et al. Pre-treatment anemia is a poor prognostic factor in soft tissue sarcoma patients. PLoS One. 2014;9(9):e107297. doi:10.1371/journal.pone.0107297.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Iqbal, N., Shukla, N.K., Deo, S.V.S. et al. Prognostic factors affecting survival in metastatic soft tissue sarcoma: an analysis of 110 patients. Clin Transl Oncol 18, 310–316 (2016). https://doi.org/10.1007/s12094-015-1369-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1369-9