Abstract
Introduction
In patients, a transient decrease in peripheral blood lymphocyte counts was observed following intraperitoneal administration of the trifunctional monoclonal antibody catumaxomab (anti-human EpCAM x anti-human CD3). The aim of this study was to clarify the observed effect in a preclinical mouse model and to analyse the related mechanism of action in vitro.
Materials and methods
A related antibody, BiLu (antihuman EpCAM x anti-mouse CD3), was administered to mice and blood leukocytes were analysed. In vitro studies measured activation and cytokine secretion from human peripheral blood mononuclear cells (PBMC). For the analysis of T cell adhesion, PBMC were preincubated with catumaxomab and then co-cultured with human endothelial cells (HUVEC); T cell adhesion was assessed in the presence or absence of endothelial cell preactivation by TNFα. Adherent T cells were determined by flow cytometry.
Results
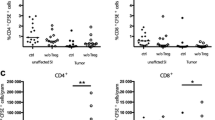
Treatment of mice with BiLu resulted in a dosedependent transient decrease in CD3+ T cells (both CD4+ and CD8+) that returned to the normal range within 48 h. Catumaxomab physiologically activated T cells in vitro (increased CD69 expression) and induced cytokine release (TNFα, IFNγ). TNFα increased expression of adhesion molecules CD54 and CD62E on endothelial cells. Furthermore, catumaxomab dose-dependently enhanced adhesion of T cells to endothelial cells. Adhesion was further increased when endothelial cells were preactivated with TNFα.
Conclusions
Catumaxomab increases adhesion of T cells to endothelial cells due to antibody-mediated activation of T cells and production of T cell cytokines that up-regulate endothelial cell adhesion molecules. These results provide a mechanistic rationale for the transient, reversible decrease in lymphocyte counts observed following catumaxomab administration in patients, which is likely to be due to redistribution of lymphocytes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Seimetz D, Lindhofer H, Bokemeyer C (2010) Development and approval of the trifunctional antibody catumaxomab (anti-EpCAM x anti-CD3) as targeted cancer immunotherapy. Cancer Treatment Rev 36:458–467
Heiss MM, Murawa P, Koralewski P et al (2010) The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: results of a prospective randomized phase II/III trial. Int J Cancer 127:2209–2221
Riesenberg R, Buchner A, Pohla H et al (2001) Lysis of prostate carcinoma cells by trifunctional bispecific antibodies (αEpCAM × αCD3). J Histochem Cytochem 49:1–7
Zeidler R, Mysliwietz J, Csanady M et al (2000) The Fc-region of a new class of intact bispecific antibody mediates activation of accessory cells and NK cells and induces direct phagocytosis of tumor cells. Br J Cancer 83:261
Zeidler R, Reisbach G, Wollenberg B et al (1999) Simultaneous activation of T-cells and accessory cells by a new class of intact bispecific antibody results in efficient tumor cell killing. J Immunol 163:1246–1252
Burges A, Wimberger P, Kümper C et al (2007) Effective relief of malignant ascites in patients with advanced ovarian cancer by a trifunctional anti-EpCAM x anti-CD3 antibody: a phase I/II study. Clin Cancer Res 13:3899–3905
Schoentag RA, Cangiarella J (1993) The nuances of lymphocytopenia. Clin Lab Med 13:923–936
Westermann J, Engelhardt B, Hoffmann JC (2001) Migration of T cells in vivo: molecular mechanisms and clinical implications. Ann Intern Med 135:279–295
van Buul JD, Hordijk PL (2004) Signaling in leukocyte transendothelial migration. Arterioscler Thromb Vasc Biol 24:824–833
Petrini B, Wasserman J, Blomgren H et al (1979) Blood lymphocyte subpopulations in breast cancer patients following post-operative adjuvant chemotherapy or radiotherapy. Clin Exp Immunol 38:361–365
Bernengo MG, Lisa F, Meregalli M, Doveil GC (1984) Changes in T and B lymphocyte subpopulations before, during and after chemotherapy for malignant melanoma. Int J Tissue React 6:505–511
Hirschhaeuser F, Leidig T, Rodday B et al (2009) Test system for trifunctional antibodies in 3D MCTS culture. J Biomol Screen 14:980–990
Schmitt M, Schmitt A, Reinhardt P et al (2004) Opsonization with a trifunctional bispecific (αCD3 × αEpCAM) antibody results in efficient lysis in vitro and in vivo of EpCAM positive tumor cells by cytotoxic T lymphocytes. Int J Oncol 25:841–848
Riechelmann H, Wiesneth M, Schauwecker P et al (2007) Adoptive therapy of head and neck squamous cell carcinoma with antibody coated immune cells: a pilot clinical trial. Cancer Immunol Immunother 56:1397–1406
Ruf P, Kluge M, Volovat C et al (2010) Pharmacokinetics, immunogenicity and bioactivity of the therapeutic antibody catumaxomab intraperitoneally administered to cancer patients. Br J Clin Pharm 69:617–625
Zeidler R, Mayer A, Gires O et al (2001) TNFα contributes to the antitumor activity of a bispecific trifunctional antibody. Anticancer Res 21:3499–3504
Amann M, D’Argouges S, Lorenczewski G et al (2009) Antitumor activity of an EpCAM/CD3-bispecific BiTE antibody during long-term treatment of mice in the absence of T-cell anergy and sustained cytokine release. J Immunother 32:452–464
Kroesen BJ, Janssen RA, Buter J et al (1995) Bispecific monoclonal antibodies for intravenous treatment of carcinoma patients: immunobiologic aspects. J Hematother 4:409–414
Janssen RA, Kroesen BJ, Buter J et al (1995) Immunomodulatory effects of intravenous BIS-1 F(ab′)2 administration in renal cell cancer patients. Br J Cancer 72:795–799
Buysmann S, Bemelman FJ, Schellekens PT et al (1996) Activation and increased expression of adhesion molecules on peripheral blood lymphocytes is a mechanism for the immediate lymphocytopenia after administration of OKT3. Blood 87:404–411
Molema G, Tervaert JW, Kroesen BJ et al (2000) CD3 directed bispecific antibodies induce increased lymphocyte-endothelial cell interactions in vitro. Br J Cancer 82:472–479
Bock HA, Gallati H, Zurcher RM et al (1995) A randomized prospective trial of prophylactic immunosuppression with ATG-Fresenius versus OKT3 after renal transplantation. Transplantation 59:830–840
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Dettmar, K., Seitz-Merwald, I., Lindemann, C. et al. Transient lymphocyte decrease due to adhesion and migration following catumaxomab (anti-EpCAM x anti-CD3) treatment in vivo . Clin Transl Oncol 14, 376–381 (2012). https://doi.org/10.1007/s12094-012-0811-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-012-0811-5