Abstract
Objectives
To evaluate the initial response and outcomes (quality of life and presence of side effects) in patients with advanced neuroendocrine tumours (NET) after treatment with radiolabelled somatostatin analogues: 90Y-DOTAT-yr3-octreotide (90Y-DOTATOC) and 177Lu-DOTA-Tyr3- octreotate (177Lu-DOTATATE).
Material and methods
The study included 5 patients with advanced NET referred to European centres for treatment with 90Y-DOTATOC and 177Lu-DOTATATE after lack of response to conventional treatment. The mean age was 45.6 years (29-68 years). Response to therapy was assessed according to: (1) RECIST criteria, as complete response, partial response, stable disease or disease progression, (2) post-treatment survival time and (3) quality of life, using the Karnofsky performance index.
Results
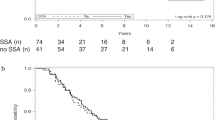
All patients survived for >20 months after treatment; mean survival time was 28 months. At the time of writing, three of the patients are alive after 20, 26 and 37 months. Partial response was observed in one patient, stable disease in three and disease progression in the fifth patient. A good-to-excellent post-treatment quality of life was observed in all patients.
Conclusion
Therapy with radiolabelled somatostatin analogues showed promising results in patients with advanced NET, with a partial response or disease stabilisation in four of the five patients, who have enjoyed an extended survival period and an improved quality of life.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Clark O, Ajani JA, Kulke M et al (2005) Neuroendocrine tumors. In: National Comprehensive Cancer Network. Clinical Practice Guidelines in Oncology
Barakat MT, Meeran K, Bloom SR (2004) Neuroendocrine tumours. Endocr Relat Cancer 11:1–18
Valdes Olmos RA, Hoefnagel CA, Bais E et al (2001) Avances terapeúticos de medicina nuclear en Oncología. Rev Esp Med Nucl 20:547–557
Cremonesi M, Ferrari M, Zoboli S et al (1999) Biokinetics and dosimetry in patients administered with (111)In-DOTA-Tyr(3)-octreotide: implications for internal radiotherapy with (90)YDOTATOC. Eur J Nucl Med 26:877–886
Waldherr C, Pless M, Maecke HR et al (2001) The clinical value of [90YDOTA]-D-Phe1-Tyr3-octreotide (90Y-DOTATOC) in the treatment of neuroendocrine tumours: a clinical phase II study. Ann Oncol 12:941–945
Waldherr C, Pless, M, Maecke HR et al (2002) Tumor response and clinical benefit in neuroendocrine tumors after 7.4 GBq 90YDOTATOC. J Nucl Med 43:610–616
Kwekkeboom DJ, Teunissen JJ, Bakker WH et al (2005) Radiolabeled somatostatin analogue [177Lu-DOTA0,Tyr3]octreotate in patients with endocrine gastroenteropancreatic tumors. J Clin Oncol 23:2754–2762
Paganelli G, Zoboli S, Cremonesi M et al (2001) Receptor-mediated radiotherapy with 90Y-DOTADPhe1- Tyr3-octreotide. Eur J Nucl Med 28:426–434
Bodei L, Cremonesi M, Grana C et al (2004) Receptor radionuclide therapy with 90Y-[DOTA]0-Tyr3-octreotide (90Y-DOTATOC) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging 31:1038–1046
Valkema R, De Jong M, Bakker WH et al (2002) Phase I study of peptide receptor radionuclide therapy with [In-DTPA] octreotide: The Rotterdam experience. Semin Nucl Med 32:110–122
Bodei L, Cremonesi M, Zoboli S et al (2003) Receptor-mediated radionuclide therapy with 90YDOTATOC in association with amino acid infusion: a phase I study. Eur J Nucl Med Mol Imaging 30:207–216
Esser JP, Krenning EP, Teunissen JJ et al (2006) Comparison of [(177)Lu-DOTA (0),Tyr (3)]octreotate and [(177)Lu-DOTA (0),Tyr (3)]octreotide: which peptide is preferable for PRRT? Eur J Nucl Med Molec Imaging 33:1346–1351
Reubi JC, Macke HR, Krenning EP (2005) Candidates for peptide receptor radiotherapy today and in the future. J Nucl Med 46[Suppl 1]:67–75
Rolleman EJ, de Jong M, Valkema R et al (2006) Inhibition of kidney uptake of radiolabeled somatostatin analogues: amino acids or gelofusine? J Nucl Med 47:1730–1731
Vegt E, Wetzels JF, Russel FG et al (2006) Renal uptake of radiolabeled octreotide in human subjects is efficiently inhibited by succinylated gelatin. J Nucl Med 47:432–436
Behr TM, Behe M, Kluge G et al (2002) Nephrotoxicity versus anti-tumourefficacy in radiopeptide therapy: facts and myths about the Scylla and Charybdis. Eur J Nucl Med Mol Imaging 29:277–279
Kwekkeboom DJ, Bakker WH, Kooij PP et al (2001) [177Lu-DOTA0Tyr3]octreotate: comparison with [111In-DTPA0]octreotide in patients. Eur J Nucl Med 28:1319–1325
Kwekkeboom DJ, Bakker WH, Kam BL et al (2003) Treatment of patients with gastro-enteropancreatic (GEP) tumours with the novel radiolabelled somatostatin analogue [177Lu-DOTA0, Tyr3]octreotate [abstract]. Eur J Nucl Med Mol Imaging 30:417–422
Kwekkeboom DJ, Bakker WH, Teunissen JJ et al (2003) Treatment with Lu-177-DOTA-Tyr3-octreotate in patients with neuroendocrine tumors: interim results [abstract]. Eur J Nucl Med Mol Imaging 30[Suppl 2]:231
DeJong M, Valkema R, Jamar F et al (2002) Somatostatin receptor-targeted radionuclide therapy of tumors: preclinical and clinical findings. Semin Nucl Med 32:133–140
Kwekkeboom DJ, Mueller-Brand J, Paganelli G et al (2005) Overview of results of peptide receptor radionuclide therapy with 3 radiolabeled somatostatin analogs. J Nucl Med 46:62–66
Kwekkeboom DJ, de Herder WW, Kam BL et al (2008) Treatment with the radiolabeled somatostatin analog [177 Lu-DOTA 0,Tyr3]octreotate: toxicity, efficacy, and survival. J Clin Oncol 26: 2124–2130
Breeman WA, de Jong M, Kwekkeboom DJ et al (2001) Somatostatin receptor-mediated imaging and therapy: basic science, current knowledge, limitations and future perspectives. Eur J Nucl Med 28:1421–1429
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muros, M.A., Varsavsky, M., Iglesias Rozas, P. et al. Outcome of treating advanced neuroendocrine tumours with radiolabelled somatostatin analogues. Clin Transl Oncol 11, 48–53 (2009). https://doi.org/10.1007/s12094-009-0310-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-009-0310-5