Abstract
Circulating tumor cells (CTC) and cancer stem cells (CSC) have been proposed as tools for detection and characterization of disease and individualization of therapy in patients with many solid tumors. Several automated and semi-automated techniques for identification and isolation of these cells from blood have been proposed and reviewed mostly focusing on their feasibility. In this mini review we summarize the recent relevant literature on this topic and discuss the clinical usability of measuring CTC and CSC in peripheral blood in patients with hepatocellular carcinoma (HCC). Besides literature, the basis for this evaluation was the authors’ experience with treating HCC and research experience on CSC and CTC. Few original reports and reviews have been published focusing on CTC and CSC in HCC. Though HCC is one of the five most common malignancies worldwide only recently these cells have come into focus for detection and characterization of this disease that is characterized by high plasticity and malignancy. A focused and prospective validation of the clinical usability of detecting these cells in HCC is still needed, but results seem promising that they may add great benefit for early detection and individualization of therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
With over 600,000 newly diagnosed cases per year, hepatocellular carcinoma (HCC) is one of the most common tumors worldwide with a significant rise in incidence in Western nations over the last years [1]. In Germany, for example, liver cancer ranges at position 6 for the cancer-related causes of death among men. HCV infection, alcohol abuse and, more recently, non-alcoholic fatty liver disease have been identified as the main risk factors for developing HCC in this country [2]. If patients are being diagnosed when presenting with symptoms, the 5-year survival rate ranges between 10 and 20 %, while early diagnosis escalates this rate to more than 50 % [3]. In patients with cirrhosis regular screening for HCC is suggested, but currently there are no cost-efficient and commonly available screening tools for other high-risk groups. Curative therapeutic options are limited to early stages and include mostly resection or orthotopic liver transplantation. High recurrence rates after resection and liver transplantation, most likely due to minimal residual disease, and the fact that the majority of patients are diagnosed in an advanced stage make palliative, often localized approaches including selective internal radiation therapy (SIRT) and transarterial chemoembolization (TACE) necessary. Up to now, there are no reliable early markers of relapse or response to surgical or interventional therapy. Serum-based markers such as alpha-fetoprotein (AFP), des-gamma-carboxyprothrombin (DCP) or the lectin 3-fraction of AFP (AFP-L3) are incapable of predicting the clinical outcome with sufficient accuracy and reproducibility in most cases. Tissue-derived molecular markers lack the possibility of monitoring the patient during or after treatment, since this would require repeated biopsies and hence increased risks for the patient. Therefore, the development of minimally invasive diagnostic methods is necessary.
Circulating tumor cells
Circulating tumor cells (CTCs) detected in the peripheral blood of HCC patients may represent a possible solution for this diagnostic dilemma. Though these cells have been frequently described in breast and lung cancer, only few studies have analyzed CTCs in patients with HCC using indirect methods such as quantitative real-time reverse-transcriptase polymerase chain reaction (qRT-PCR), an approach often used in gastrointestinal malignancies [4], or direct visualization of circulating epithelial cells. The main obstacle to the broad clinical application of available automated CTC detection methods is the high plasticity and variability of these cells particularly due to the epithelial-to-mesenchymal-transition (EMT). EMT inevitably leads to decreased detection of CTCs with techniques based mostly on assumed epithelial characteristics of these cells. Remarkably, changes from epithelial to mesenchymal cell characteristics are significantly correlated to treatment response [5].
CTCs are defined as tumor cells that either originate from the primary tumor or from metastases of this tumor. However, it is still not yet fully understood whether these identifiable circulating cells may represent the primary tumor or its metastases or even an entity of its own kind. Whether CTCs might be a surrogate of the tumor tissue is an important question regarding their utilization as a so-called “liquid bisopy.” Bearing in mind the heterogeneity of solid malignancies, one could argue that methods focusing on certain subtypes of CTCs, e.g., with epithelial characteristics, may only detect a small subpopulation of the tumor. Thus, relying on these cells for treatment decisions or to monitor therapeutic success may be an inadequate approach. This is an essential predicament already discussed for more than 3 decades [6] as tumors are very heterogeneous, and it is not clear whether single cell subpopulations that circulate in the bloodstream really imply altered prognosis or response to therapy. Still, CTC analysis including all subpopulations would certainly add to an improved monitoring of therapeutic response. The current standard procedure of targeted therapy includes obtaining biomarkers from the primary therapy-naïve tumor not considering the high plasticity of tumors when confronted with cytotoxic or biological agents. The alterations in CTC quantity or quality during therapy could therefore be used to react to changes in responsiveness to treatment in a timelier manner than available imaging techniques without the requirement of highly invasive procedures.
The epithelial cell adhesion molecule (EpCAM) is exclusive to epithelial and tumorous cells and is often used to test for CTCs in the bloodstream of patients with solid malignancies. Identification of CTCs based on EpCAM positivity allows the differentiation from mesenchymal and hematopoietic cells. Furthermore, EpCAM is a promising candidate protein for cancer targeting agents. Nonetheless, recent studies were able to show that this molecule may only be part of the equation as the heterogeneity and plasticity of CTCs make a holistic methodology based on certain characteristics, only present in some of the cells, impossible. One reason for this problem is that during metastasis the EMT leads to loss of epithelial surface (EpCAM) and intracellular (cytokeratin, CK) characteristics and to acquisition of mesenchymal features associated with expression of vimentin and N-cadherin [7]. This results in the loss of cell-cell adhesion and gain of mobility, invasiveness and apoptotic resistance. More importantly, these characteristics may well serve these cells and therefore the malignant process in defying cytotoxic therapy. Gradilone and his collaboration group identified circulating mesenchymal tumor cells in patients with breast cancer in addition to epithelial ones and were able to correlate these findings to a poor prognosis [8]. Interestingly, cellular signaling pathways that play a role in triggering EMT such as the Wnt-, Notch- and Hedgehog-pathways are also involved in the development of CSCs [8]. Mani and colleagues described a link between EMT and stemness states in breast cancer models [9]. Their results illustrated a direct connection of less differentiated stem cells with the mesenchymal-appearing cells generated by EMT [10].
Independently of the heterogeneity and plasticity of CTCs as well as their missing specific cell surface markers, an average of one CTCs in 106–107 leucocytes challenges isolation and detection [11]. Basically, the established methods consist of two steps—enrichment and identification. A first density gradient centrifugation step allows a preselection of peripheral blood mononuclear cells (PBMNC). From this cell suspension, further positive or negative selection of specific CTC subtypes can be made, e.g., by employing immunomagnetic beads either directed against tumor-associated surface proteins such as EpCAM (epithelial) or OB-cadherin (mesenchymal) or against hematopoietic markers such as the common leucocyte antigen CD45. Available enrichment techniques based on various systems including the CellSearch® platform (Veridex, Raritan, NJ) using antibodies against EpCAM, keratin and CD45 to enumerate CTCs in breast, colon and prostate cancer as well as the filtration by size ISET (Isolation by Size of Epithelial/Throphoblastic Tumor cells) platform are described elsewhere [12–17]. However, especially the automated approaches are challenged by a lack of flexibility, needed to address the great differences in subpopulations and size that CTCs inherit before or during treatment [10, 18, 19]. Microscopy-based approaches allow additional morphological examinations of CTCs as well as downstream applications such as fluorescence in situ hybridization (FISH) analysis for instance employing the so-called FICTION protocol (fluorescence immunophenotyping, and interphase cytogenetics as a tool for the investigation of neoplasms) [20] or dual-colorimetric RNA-ISH assay [5] for individualized treatment decisions. An interesting non-enrichment fluid-phase biopsy approach using a high-definition (HD) CTC technology based on morphological characterization and high-throughput counting was reported by Marrinucci et al. [21]. The results displayed the ability of an enrichment-free assay to identify significant CTC numbers in the majority of patients with prostate, breast and pancreatic cancers.
In HCC, the lack of specific cell surface antigens challenges CTC detection. To date, only a few studies have been performed to identify or quantify these cells [18, 22–30]. Table 1 briefly summarizes the development of CTC research in the field of HCC during the last decade. The reports are very heterogenous regarding the study material. Some used patient blood samples; others used cell lines or mouse models. Also the applied CTC enrichment and detection methods varied, probably depending on the state of the art at the time, the availability of detection assays and the individual background of the studies, respectively. Waguri et al. [22] postulated that hTERT mRNA expression after immunomagnetic separation using EpCAM and anti-CD45 antibodies might be a specific tool for CTC detection in HCC. They reported a CTC detection rate of 53 % (29 out of 55 HCC cases). Vona et al. [23] used the ISET machine in advanced HCC and found that 52 % of a total of 44 patients had detectable CTCs. Moreover, they were able to correlate the appearance of CTCs with multifocal tumors, portal vein thrombosis and the Child status. The EpCAM-based enrichment method after density gradient centrifugation was comprehensively assessed by Guo et al. [24] who spiked healthy blood with a serial dilution of HepG2 cells and reported that as few as 10 cells in 5 ml blood could be detected using RT-PCR with primers specific for AFP. In clinical samples, the positive detection rate ranged from 53 to 93 % depending on the Child-Pugh class with a total positive detection rate of 73 %. Yang et al. [25] used flow cytometry to assess the distribution of CD45− CD90+ cells in 34 blood samples from liver cancer patients resulting in a detection rate of 90 %, but none in normal subjects or patients with cirrhosis. Xu et al. [26] reported the development and validation of an EpCAM-independent magnetic cell separation system mediated by the interaction of ASGPR1 being exclusively expressed on hepatocytes with its ligand. CTCs were then identified by Hep-Par-1 staining. An average of 24 ± 19 CTCs per 5 ml blood was detected in 81 % of HCC patients, suggesting that the variation ranged from 1 to 9 tumor cells per ml in the examined patients. Fan et al. [27] combined an in vivo flow cytometry technique with a GFP-transfected HCC mouse model to monitor CTC dynamics continuously and noninvasively using the ear artery. Based on multiple calculations, they reported a detection rate of 7.81 CTCs per ml blood and thus a 1.8-fold higher sensitivity than whole blood analysis using conventional flow cytometry. Liu et al. [29] investigated the expression of intercellular adhesion molecule 1 (ICAM-1) in HCC blood samples using flow cytometry. They found that 0.3 % of the cells in an HCC blood sample were CD45− ICAM-1+ cells and that those cells displayed greater sphere-forming and tumorigenic capacities and increased expression of stemness-related genes compared to CD45-ICAM- cells. The first report to identify the stem cell-like characteristics of EpCAM-positive CTCs and their prognostic significance using the standardized CellSearch System in HCC patients was published by Sun et al. [28]. Preoperatively, they detected EpCAM-positive cells in 66.7 % of the cases (82 out of 123) ranging from 1–34 cells per 7.5 ml blood and hypothesized that a preoperative count of >2 might be an independent prognostic indicator for recurrence. Also Schulze and colleagues used the CellSearch System to test for EpCAM-positive tumor cells in the peripheral blood of 59 HCC patients. Their study resulted in a detection rate of 30.5 % and a significant association of quantifiable epithelial CTCs with overall survival, BCLC staging, vascular invasion and the serum-based tumor marker AFP [30, 31]. Nel et al. [18] applied a negative CTC selection technique by depleting hematopoietic cells using immunomagnetic beads directed against CD45 and subsequent multi-immunofluorescence staining to detect various CTC subpopulations in patients with HCC. The described method is based on individual CTC profiles and subsequent cell type ratios rather than absolute cell numbers. They detected cells with mesenchymal features such as N-cadherin+/CD45− and vimentin+/CD45− as well as cells showing both epithelial and mesenchymal characteristics such as N-cadherin+/CK+/CD45− and vimentin+/CK+/CD45− cells. The results in this study group indicated that an increase in epithelial cells was associated with worse treatment outcome in patients with HCC. Recently, in a systemic review Gao et al. [32] analyzed the value of CTC detection in HCC and gastric cancer and pointed out limitations due to small cohorts and potential bias in the studies reported so far. The review indicated that large multicenter trials are required to confirm the prognostic relevance of CTCs in HCC patients.
Circulating cancer stem cells
The intratumoral heterogeneity is believed to be caused by subpopulations of cells that are genetically identical but display distinct phenotypic states such as CSCs and non-CSCs. The abilities of self-renewal and tumor initiation define CSCs and are relevant to metastasis. Chaffer and Weinberg [33] hypothesized the presence of intrinsic and induced CSC subtypes within a tumor, which may explain the pathological heterogeneity. Intrinsic CSCs are believed to exist in the primary tumor from the beginning of tumorigenesis. Induced CSCs can be formed when cells such as myofibroblasts, macrophages or mesenchymal stem cells undergo the process of EMT and enter a CSC-like stage. Therefore, CSCs are described as non-equivalent to CTCs. Only CTCs that have the ability to form ectopic metastasis have CSC characteristics and are known as circulating CSCs [12]. As with CTCs, circulating CSCs may be recognized by certain characteristics not common in other cells identifiable in the bloodstream, i.e., hematopoietic cells, by using multimarker approaches including staining for CD45 and stem cell-specific markers. They have been successfully isolated from peripheral blood in patients with liver cancer using anti-CD90-coated microbeads and subsequent counterstaining against CD45 and CD44 [34]. Yet, there is no definitive marker for liver-derived CSCs in the bloodstream as none of the defined CSC markers are exclusive to liver cancer stem cells [35]. Yamashita et al. [36] postulated that EpCAM is one of the promising markers to define aggressive subpopulations of CSCs. They are believed to have colony-forming potential and can produce EpCAM+ as well as EpCAM− cells. Additional CD133 staining revealed an EpCAM+ CD133+ subpopulation with tumorigenic capacity indicating that EpCAM as well as CD133 may be hepatic CSC markers; EpCAM is suggested to be the better marker with regards to the enrichment technique. As mentioned above, EpCAM-positive cells can be isolated from blood by various approaches including immunomagnetic bead selection. Furthermore, the additional staining of other cell surface molecules such as CD44 may increase the specificity in identifying circulating CSCs [37]. In the treatment of solid malignancies, tumor recurrence and resistance to therapy during systemic or local-ablative approaches are the main obstacles. While tumors with high turnover and rapid proliferation respond well to available cytotoxic agents, HCC shows a different behavior. Up to now, systemic treatment in HCC has failed to reach good response rates and significant survival benefits [38]. CSCs may escape therapeutic approaches by being composed of quiescent slow-cycling subpopulations that will not respond to antiproliferative agents [39]. Although EpCAM+/CK19+ cells seem to be the mainstay of CSCs in liver cancer, these cells are not the only ones with proven stemness and therefore tumor-forming characteristics [40]. Viatour and colleagues [40] used a mouse model to show that deleting three members of the retinoblastoma 1 (Rb1) gene family, namely Rb, p107, and p130, therefore inactivating a pathway generally silenced in human HCC by a diversity of host mechanisms, results in the development of liver tumors with histopathological features and gene expression profiles similar to human HCC. Therefore, they proved that in their mouse model cancer initiation was associated with a specific expansion of CSC populations. Measuring liver cancer stem cells in the bloodstream of patients before local ablative or systemic treatment and after the initial cycle may contribute valuable information to the clinician for identifying those patients who will respond to treatment and those with a prolonged time to progression. CSCs themselves may also be an important target for new treatment strategies as the classical approach targeting rapid proliferative cells also influences healthy cell populations leading to the typical side effects of systemic treatment such as neutropenia, significant gastrointestinal mucositis and diarrhea (Sorafenib) or dramatic changes in the skin constitution leading to hand-foot syndrome and therefore limiting cancer patients activities of daily life (ADL). Moreover, targeting the active and proliferative cell population of the tumor may only result in a short gain—bulk reduction—but a long-term loss since these cells are not the tumor-forming population [41]. An innovative methodology in CSC-targeted therapy is the siRNA-mediated downregulation of signaling pathways involved in carcinogenesis. Yamashita et al. [42] reported that silencing of EpCAM using RNA interference in Hep3B cells resulted in growth inhibition. Another approach to target CSCs is the use of monoclonal antibodies directed against CSC surface markers and conjugated to an antitumor or cytotoxic drug [43]. Smith et al. [44] reported a decreased proliferation rate of Hep3B cells and delayed tumor growth in a SCID mouse model after targeting CD133+ cells with anti-CD133 antibody conjugated to monomethyl auristatin. Yang et al. [34] showed that treatment of CD90+ CD44+ cells with an antibody against CD44 prevented tumor metastasis in a nude mouse model. A brief summary of current CSC markers for liver cancer and relevant targeted therapeutic approaches were reported by Lee et al. [45]. It has been shown that human mesenchymal stem cells (MSC) have the ability to migrate to and proliferate within tumor sites [46]. Therefore, MSCs might be utilized as cellular vehicles to deliver therapeutic genes introduced by viral vectors. First studies targeting stem cells using transduced MSC resulted in tumor growth suppression in mouse models [47]. But before viral-based treatments are ready for prime time, serious issues such as the immunogenicity of viral vectors have to be resolved. Furthermore, the lack of specific CSC markers will result in inhibition of non-CSCs. Presently, targeting of CSCs is not yet fully understood. Development of localized treatment combinations targeting CSCs directly in the liver may be promising as it will not only result in bulk reduction but probably also reduce the small tumor-forming cell subpopulation. Although CSC analysis may not be usable for treatment decisions at the moment, identifying and characterizing the individual tumor cell composition and the constitution of circulating non-hematopoietic stem cells in the blood of patients before therapy may add important information to the process of personalizing therapy. Patients with increased numbers of CSCs may not benefit from systemic or even local ablative approaches as they do not respond to treatments that rely on antiproliferative effects. Identification of these cells is already possible and should be evaluated during prospective treatment of patients with liver cancer.
Conclusions
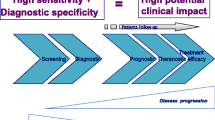
So far, scientists and clinicians have focused on identifying and quantifying certain subgroups of CSCs or CTCs. They relied on either defined surface markers or differences in the size of individual cell populations. These approaches have several limitations as they only represent small parts of the picture; therefore, changes in these cell populations during treatment may at most implicate success in bulk reduction but, as reality shows, no real survival benefits. To get a clearer picture of the individual cell composition and the constitution of the circulating non-hematopoietic cells, more holistic approaches need to be taken urgently. Therefore, all non-hematopoietic subpopulations circulating in the bloodstream need to be analyzed. The correlation of cell profiles of the primary chemo-naïve tumor and during curative or palliative treatment with clinical responses will improve our understanding of the roles that CTCs and circulating CSCs play in HCC. To this aim, large collaborative groups are needed to reach sufficient patient numbers and different clinical courses. However, the necessary techniques are already available. Clinicians should focus less on refining automated quantification of certain subgroups and more on understanding the association of circulating non-hematopoietic cells with the primary chemo-naïve tumor and the correlation of changes in the individual tumor cell composition, the CTC and CSC profiles, during treatment with the patient’s response. However, before applying CTC analysis in personalizing patient care, there has to be consensus on the gold standard, especially since the development in detection of CTCs over the last 10 years has been dramatic (Table 1).
Abbreviations
- CTCs:
-
Circulating tumor cells
- qRT-PCR:
-
Quantitative real-time reverse transcriptase polymerase chain reaction
- CK:
-
Cytokeratin
- EpCAM:
-
Epithelial cell adhesion molecule
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin 2011;61:69–90
Ertle J, Dechene A, Sowa JP, Penndorf V, Herzer K, Kaiser G, et al. Non-alcoholic fatty liver disease progresses to hepatocellular carcinoma in the absence of apparent cirrhosis. Int J Cancer 2011;128:2436–2443
Bruix J, Sherman M. American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology 2011;53:1020–1022
Hoffmann AC, Vallbohmer D, Grimminger P, Metzger R, Prenzel KL, Hoelscher AH, et al. Preoperative survivin mRNA detection in peripheral blood is an independent predictor of outcome in esophageal carcinoma. Pharmacogenomics 2010;11:341–347
Yu M, Bardia A, Wittner BS, Stott SL, Smas ME, Ting DT, et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 2013;339:580–584
Fidler IJ. Tumor heterogeneity and the biology of cancer invasion and metastasis. Cancer Res 1978;38:2651–2660
Christofori G. New signals from the invasive front. Nature 2006;441:444–450
Gradilone A, Raimondi C, Nicolazzo C, Petracca A, Gandini O, Vincenzi B, et al. Circulating tumour cells lacking cytokeratin in breast cancer: the importance of being mesenchymal. J Cell Mol Med 2011;15:1066–1070
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008;133:704–715
Nel I, Gauler T, Hoffmann AC. Circulating tumor cell composition and outcome in patients with solid tumors. Int J Clin Pharmacol Ther. 2014;52:74–75
Riethdorf S, Pantel K. Advancing personalized cancer therapy by detection and characterization of circulating carcinoma cells. Ann N Y Acad Sci 2010;1210:66–77
Bitting RL, Boominathan R, Rao C, Kemeny G, Foulk B, Garcia-Blanco MA, et al. Development of a method to isolate circulating tumor cells using mesenchymal-based capture. Methods 2013;64:129–136
Wu LJ, Pan YD, Pei XY, Chen H, Nguyen S, Kashyap A, et al. Capturing circulating tumor cells of hepatocellular carcinoma. Cancer Lett 2012;326:17–22
Chiappini F. Circulating tumor cells measurements in hepatocellular carcinoma. Int J Hepatol 2012;2012:684802
Zhang Y, Li J, Cao L, Xu W, Yin Z. Circulating tumor cells in hepatocellular carcinoma: detection techniques, clinical implications, and future perspectives. Semin Oncol 2012;39:449–460
Alix-Panabieres C, Pantel K. Technologies for detection of circulating tumor cells: facts and vision. Lab Chip 2014;14:57–62
Krebs MG, Hou JM, Sloane R, Lancashire L, Priest L, Nonaka D, et al. Analysis of circulating tumor cells in patients with non-small cell lung cancer using epithelial marker-dependent and -independent approaches. Journal of Thoracic Oncology 2012;7:306–315
Nel I, Ertlel JM, Baba HA, Weber F, Sitek B, Stephan C, et al. A novel method for the detection of different subgroups of circulating tumor cells in patients with hepatocellular carcinoma. Hepatology 2012;56:457A
Nel I, Baba HA, Ertle J, Weber F, Sitek B, Eisenacher M, et al. Individual profiling of circulating tumor cell composition and therapeutic outcome in patients with hepatocellular carcinoma. Transl Oncol 2013;6:420–428
Campos M, Luque R, Jimenez J, Martinez R, Warleta F, Sanchez-Quesada C, et al. Simultaneous phenotypic and genetic characterization of single circulating tumor cells from colon cancer patients. Histol Histopathol 2013;6(5):872–879
Marrinucci D, Bethel K, Kolatkar A, Luttgen MS, Malchiodi M, Baehring F, et al. Fluid biopsy in patients with metastatic prostate, pancreatic and breast cancers. Phys Biol 2012;9:016003
Waguri N, Suda T, Nomoto M, Kawai H, Mita Y, Kuroiwa T, et al. Sensitive and specific detection of circulating cancer cells in patients with hepatocellular carcinoma; detection of human telomerase reverse transcriptase messenger RNA after immunomagnetic separation. Clin Cancer Res 2003;9:3004–3011
Vona G, Estepa L, Beroud C, Damotte D, Capron F, Nalpas B, et al. Impact of cytomorphological detection of circulating tumor cells in patients with liver cancer. Hepatology 2004;39:792–797
Guo J, Yao F, Lou Y, Xu C, Xiao B, Zhou W, et al. Detecting carcinoma cells in peripheral blood of patients with hepatocellular carcinoma by immunomagnetic beads and rt-PCR. J Clin Gastroenterol 2007;41:783–788
Yang ZF, Ngai P, Ho DW, Yu WC, Ng MN, Lau CK, et al. Identification of local and circulating cancer stem cells in human liver cancer. Hepatology 2008;47:919–928
Xu W, Cao L, Chen L, Li J, Zhang XF, Qian HH, et al. Isolation of circulating tumor cells in patients with hepatocellular carcinoma using a novel cell separation strategy. Clin Cancer Res 2011;17:3783–3793
Fan ZC, Yan J, Liu GD, Tan XY, Weng XF, Wu WZ, et al. Real-time monitoring of rare circulating hepatocellular carcinoma cells in an orthotopic model by in vivo flow cytometry assesses resection on metastasis. Cancer Res 2012;72:2683–2691
Sun YF, Xu Y, Yang XR, Guo W, Zhang X, Qiu SJ, et al. Circulating stem cell-like epithelial cell adhesion molecule-positive tumor cells indicate poor prognosis of hepatocellular carcinoma after curative resection. Hepatology 2013;57:1458–1468
Liu S, Li N, Yu X, Xiao X, Cheng K, Hu J, et al. Expression of intercellular adhesion molecule 1 by hepatocellular carcinoma stem cells and circulating tumor cells. Gastroenterology 2013;144(1031–1041):e1010
Schulze K, Gasch C, Staufer K, Nashan B, Lohse AW, Pantel K, et al. Presence of EpCAM-positive circulating tumor cells as biomarker for systemic disease strongly correlates to survival in patients with hepatocellular carcinoma. Int J Cancer 2013;133:2165–2171
Schulze K, Beneken C, Staufer K, Nashan B, Lohse AW, Pantel K, et al. Detection of Epcam-Positive Circulating Tumor Cells in Patients with Hepatocellular Carcinoma—a Pilot Study. Hepatology 2011;54:1357a
Gao P, Jiao SC, Bai L, Wang H, Jing FF, Yang JL. Detection of circulating tumour cells in gastric and hepatocellular carcinoma: a systematic review. J Int Med Res 2013;41:923–933
Chaffer CL, Weinberg RA. A perspective on cancer cell metastasis. Science 2011;331:1559–1564
Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai P, et al. Significance of CD90 + cancer stem cells in human liver cancer. Cancer Cell 2008;13:153–166
Yoon SK. The biology of cancer stem cells and its clinical implication in hepatocellular carcinoma. Gut Liver 2012;6:29–40
Yamashita T, Ji J, Budhu A, Forgues M, Yang W, Wang HY, et al. EpCAM-positive hepatocellular carcinoma cells are tumor-initiating cells with stem/progenitor cell features. Gastroenterology 2009;136:1012–1024
Zhu Z, Hao X, Yan M, Yao M, Ge C, Gu J, et al. Cancer stem/progenitor cells are highly enriched in CD133 + CD44 + population in hepatocellular carcinoma. Int J Cancer 2010;126:2067–2078
Kim SR, Kudo M, Hino O, Han KH, Chung YH, Lee HS. Organizing Committee of Japan-Korea Liver S. Epidemiology of hepatocellular carcinoma in Japan and Korea. A review. Oncology 2008;75(Suppl 1):13–16
Libbrecht L, Roskams T. Hepatic progenitor cells in human liver diseases. Semin Cell Dev Biol 2002;13:389–396
Viatour P, Ehmer U, Saddic LA, Dorrell C, Andersen JB, Lin C, et al. Notch signaling inhibits hepatocellular carcinoma following inactivation of the RB pathway. J Exp Med 2011;208:1963–1976
Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, et al. Cancer stem cells–perspectives on current status and future directions: aACR Workshop on cancer stem cells. Cancer Res 2006;66:9339–9344
Yamashita T, Budhu A, Forgues M, Wang XW. Activation of hepatic stem cell marker EpCAM by Wnt-beta-catenin signaling in hepatocellular carcinoma. Cancer Res 2007;67:10831–10839
Chen K, Huang YH, Chen JL. Understanding and targeting cancer stem cells: therapeutic implications and challenges. Acta Pharmacol Sin 2013;34:732–740
Smith LM, Nesterova A, Ryan MC, Duniho S, Jonas M, Anderson M, et al. CD133/prominin-1 is a potential therapeutic target for antibody-drug conjugates in hepatocellular and gastric cancers. Br J Cancer 2008;99:100–109
Lee TK, Castilho A, Ma S, Ng IO. Liver cancer stem cells: implications for a new therapeutic target. Liver Int 2009;29:955–965
Klopp AH, Spaeth EL, Dembinski JL, Woodward WA, Munshi A, Meyn RE, et al. Tumor irradiation increases the recruitment of circulating mesenchymal stem cells into the tumor microenvironment. Cancer Res 2007;67:11687–11695
Uchibori R, Okada T, Ito T, Urabe M, Mizukami H, Kume A, et al. Retroviral vector-producing mesenchymal stem cells for targeted suicide cancer gene therapy. J Gene Med 2009;11:373–81
Compliance with ethical requirements and Conflict of interest
The authors declare that they have no ethical conflicts. Ivonne Nel, Paul David, Guido G. H. Gerken, Joerg F. Schlaak and Andreas-Claudius Hoffmann declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nel, I., David, P., Gerken, G.G.H. et al. Role of circulating tumor cells and cancer stem cells in hepatocellular carcinoma. Hepatol Int 8, 321–329 (2014). https://doi.org/10.1007/s12072-014-9539-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-014-9539-3