Abstract
Glioblastoma is the worst and most common primary brain tumor. Here, we demonstrated the role of CD73, an enzyme responsible for adenosine (ADO) production, in glioblastoma progression. ADO increased glioma cell viability via A1 receptor sensitization. CD73 downregulation decreased glioma cell migration and invasion by reducing metalloproteinase-2 and vimentin expression and reduced cell proliferation by 40%, which was related to necrosis and sub-G1 phase blockage of cell cycle. Those effects also involved the stimulation of Akt/NF-kB pathways. Additionally, CD73 knockdown or enzyme inhibition potentiated temozolomide cytotoxic effect on glioma cells by decreasing the IC50 value and sensitizing cells to a non-cytotoxic drug concentration. CD73 inhibition also decreased in vivo rat glioblastoma progression. Delivery of siRNA-CD73 or APCP reduced tumor size by 45 and 40%, respectively, when compared with control. This effect was followed by a parallel 95% reduction of ADO levels in cerebrospinal fluid, indicating the role of extracellular ADO in in vivo glioma growth. Treatment did not induce systemic damage or mortality. Altogether, we conclude that CD73 is an interesting target for glioblastoma treatment and its inhibition may provide new opportunities to improve the treatment of brain tumors.

ᅟ
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nucleotides are released by a variety of cell types in response to stress signals, such as injury, hypoxia, and inflammatory condition in the tumor microenvironment (TME) [1]. These molecules are metabolized by ectonucleotidases, including NTPDases, which hydrolyze ATP and ADP to AMP; ecto-5′-nucleotidase (CD73) and prostatic acid phosphatase (PAP), which convert AMP to adenosine (ADO); and adenosine deaminase (ADA), which deaminates ADO to inosine [2,3,4]. Therefore, these ectozymes play a crucial role in tumor progression by controlling extracellular nucleotide/nucleoside levels, such as ATP and ADO, and the purinoceptor-mediated signaling in cells that constitute TME [5, 6]. Although PAP was recently described as an ADO source in glioblastoma stem cells (GSC) [5], studies from literature and from our group point to the importance of the CD39-CD73 axis in ADO production in TME [7,8,9,10,11,12,13,14,15,16,17,18]. In line with this, the present study focused on investigating the participation of CD73 in glioblastoma growth.
CD73 is a protein bound to the outer surface of the plasma membrane by a glycosylphosphatidylinositol (GPI) anchor and localized within lipid rafts [7]. CD73 overexpression has been reported in a variety of cancer cells and tumor patient biopsies, including gliomas and breast cancer, being associated with worse disease-free survival in glioblastoma patients [8,9,10]. CD73 has enzymatic and non-enzymatic functions in cancer progression [10,11,12,13]. Notably, extracellular ADO production through CD73 activity plays important roles in cancer immunosuppression, angiogenesis, cell proliferation, and chemoresistance via P1 purinoceptor sensitization [14,15,16,17,18,19,20]. In addition to its enzymatic function, CD73 participates in tumor cell-extracellular matrix interactions, which may favor tumor spread and metastasis [11, 17, 21]. Therefore, CD73 is a promising candidate for the development of new therapeutic strategies in cancer treatment.
Glioblastoma is the worst and most common primary brain tumor [22]. Standard treatment strategies include complete surgical resection followed by radio- and systemic chemotherapy with temozolomide (TMZ) [23]. However, patients with glioblastoma exhibit a median survival of only 12 months after diagnosis [24, 25]. Given that glioblastomas are highly resistant to most cytotoxic drugs and radiotherapy, the characterization of new biological targets is extremely necessary in order to offer therapeutic options for patients. In this regard, CD73 has emerged as an important component of cancer malignancy; however, its roles in in vitro and in vivo glioma progression have not been fully elucidated. Therefore, the aim of this investigation was to evaluate the potential of CD73 as a target for glioblastoma therapy.
Materials and Methods
Characterization of CD73 Role in In Vitro Glioma Progression
Cell Lines
C6 and U87MG cell lines were purchased from American Type Culture Collection (ATCC; Rockville, Maryland, USA). Cells were maintained in low-glucose Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (FBS), penicillin/streptomycin (100 U/L), and fungizone (0.1%) (Gibco, Carlsbad, California, USA) in a humidified incubator with 5% CO2.
Cell Treatment and CD73 Knockdown
Rat C6 or human U87MG glioma cell lines were seeded at 5 × 103 cells/well in DMEM/10% FBS in 96-well plates. Cultures were exposed for 24, 48, or 72 h to ADO (product of CD73 enzyme activity; 0.1, 1, 10, 25, 50, 100, and 250 μM), AMP (CD73 substrate; 1, 10, 50,100, 250, 500, and 1000 μM), APCP (CD73 pharmacological inhibitor; 1, 10, 25, 50,100, 250, and 500 μM) [17], DPCPX (selective A1R antagonist; 1 μM) [26], Zm241385 (selective A2aR antagonist; 1 μM) [27], IB-MECA (selective A3R agonist; 20 μM) [28], caffeine (non-selective P1R antagonist; 10 μM) [29], and dipyridamole (nucleoside uptake inhibitor; 100 μM) [16]. Appropriate controls containing 0.01% DMSO (vehicle) were included (Table S1).
For CD73 knockdown, two siRNA sequences against rat CD73 (NCBI Nt5e, NM_021576.2, rCD73-siRNA-961, and rCD73-siRNA-980) and one siRNA sequence against human CD73 (NCBI NG_028214.1, hCD73-siRNA-441) were generated. Control cells were transfected with a scramble green fluorescent protein (GFP)-siRNA sequence (Table 1). Glioma cells were transfected using Lipofectamine RNAiMax reagent (Invitrogen, Carlsbad, California, USA) by reverse transfection (10 nM siRNA) according to manufacturer’s instructions. CD73 knockdown was confirmed after 72 h of transfection by flow cytometry (FACS), immunofluorescence staining, and AMPase activity determination as described below.
To assess glioma sensitivity to TMZ, C6 or U87MG cells were either treated with APCP (100 μM) for 72 h, or CD73 knockdown was performed. Afterwards, cells were exposed to a concentration curve of TMZ (12.5, 25, 50, 100, 250, 500, and 1000 μM) for additional 72 h. Control cells were treated with vehicle (0.01% DMSO). Cell viability was determined using MTT method as described below. Cells treated with vehicle DMSO, APCP, or siRNA-CD73 alone were considered controls. IC50 value was defined as the drug concentration that inhibited 50% of cell viability when compared with the untreated control.
MTT Assay
For cell viability assay, 5 × 103 cells/well were seeded in 96-well plates. After 24 h, culture medium was replaced with fresh medium, and cells were exposed to treatments as described above. Following 24, 48, or 72 h of treatment, cell viability was assessed by 3(4,5-dimethyl)-2,5diphenyltetrazolium bromide (MTT) assay (Sigma, St. Louis, Missouri, USA). This method is based on the ability of viable cells to reduce MTT and form a blue formazan product. MTT solution was added to incubation medium in the wells at a final concentration of 0.5 mg/mL. After 90 min, medium was removed, DMSO was added to the wells, and plates were shaken. Cell viability was measured according to the optical density (OD) at 492 nm. This value was calculated using Prism 5.0 software (Prism GraphPad Software, San Diego, USA) according to the following formula: cell viability rate (%) = (OD492 of treated cells/OD492 of control) × 100%.
Flow Cytometry
CD73 expression in glioma cells was evaluated by flow cytometry using rabbit anti-rat or anti-human CD73 primary antibodies (Ab) (1:100; http://ectonucleotidases-ab.com). Briefly, cells were incubated for 60 min with primary Ab prepared in PFA buffer (PBS, 1% FBS, 0.1% sodium azide) followed by 30 min incubation with FITC-conjugated secondary anti-rabbit Ab (1:100; Kirkegaard Perry Laboratories, USA) with a minimum of two washes with PFA after each incubation. Cell surface fluorescence was measured in a FACSCalibur Flow Cytometer (BD Biosciences, San Jose, California, USA). Data were expressed as CD73 expression rate (%) = (mean fluorescence intensity (MFI) of treated cells/MFI of control) × 100%.
Immunofluorescence Microscopy
C6 and U87MG glioma cells were seeded at 2 × 104 cells/well in DMEM/10% FBS onto a coverslip. Following 72 h, cells were fixed in 10% phosphate-buffered formalin/95% acetone, and sections were incubated for 90 min at room temperature (RT) with primary Ab (rabbit anti-rat or rabbit anti-human CD73 (1:1000; http://ectonucleotidases-ab.com), followed by incubation with FITC-conjugated secondary anti-rabbit Ab (1:1000; Kirkegaard Perry Laboratories, USA) for 60 min at RT. Sections were counterstained with DAPI blue (1:10,000; Sigma, St. Louis, MO, USA) for 5 min. Images were captured using a digital camera connected to a microscope (Olympus BX-50, Tokyo, Japan).
Ectonucleotidase Activity
C6 and U87MG glioma cells were seeded in 24-well plates (2 × 104 cells/well) and AMPase activity was determined after 72 h of CD73 knockdown or APCP treatment. Enzyme reaction was started by the addition of incubation medium containing 2 mM MgCl2, 120 mM NaCl, 5 mM KCl, 10 mM glucose, 20 mM HEPES (pH 7.4), and 2 mM AMP as substrate. Incubation proceeded for 10 min at 37 °C. The reaction was stopped with 10% trichloroacetic acid (TCA). The released inorganic phosphate (Pi) was assayed by malachite green method using KH2PO4 as standard [30]. Protein was measured by Coomassie blue method [31] using serum albumin as standard. Activity was reported as nanomoles Pi per minute per milligram of protein.
Scratch-Wound Assay
C6 and U87MG glioma cell lines were seeded in 12-well plates (5 × 103 cells/well). Cells were exposed to CD73-siRNA complexes or treated with APCP (1, 10, and 100 μM) or ADO (1 μM) in DMEM/10% FBS. After 72 h, a cell monolayer wound was performed with a P200 pipette tip in SFB reduced conditions (DMEM/0.5% FBS). Next, DMEM/0.5% SFB medium was replaced with fresh medium in the presence of treatments (APCP or ADO). Control cells were exposed to DMEM/0.5% FBS or to GFP-siRNA complexes. Following 0, 6, 12, and 24 h of scratch, images were captured using an inverted microscope (× 4 magnification) connected to a digital camera (Olympus BX-50, Tokyo, Japan). Cell migration was determined by measuring the width of the wound divided by two and subtracting this value from the initial half-width distance of the wound. Data were expressed as the percentage of migration compared with DMEM/0.5% FBS or GFP-siRNA-treated cells.
Migration and Invasion Transwell Assays
Cell migration and invasion were evaluated using a 24-well transwell chamber (Greiner, Frickenhausen, Germany) according to Iser et al. [32]. To study cell invasion, 50 μL of 1 mg/mL Matrigel was used to coat the upper chamber of the transwell insert. For this, 2.5 × 104 C6 glioma cells treated as described above (2.1.7) were seeded in the upper chamber of an 8-μm pore size insert in DMEM only and allowed to migrate toward a DMEM/10% FBS placed at the lower chamber. After 24 h, non-migrating or non-invading cells in the upper chamber were removed with a cotton swab and the remaining cells were fixed in methanol for 15 min. Cells that migrated to or invaded the lower surface of the membrane were stained with 0.5% crystal violet diluted in 20% methanol. Images of stained cells were captured using a digital camera connected to a microscope (Olympus BX-51, Tokyo, Japan). Four visual fields were randomly chosen for each assay. Cell quantitation was performed by processing all obtained images using ImageJ software (http://imagej.nih.gov/ij/). The average number of migrating or invading cells in these four fields was considered as the cell migration or invasion potential of the group.
RNA Isolation and Quantitative PCR
Total RNA from C6 glioma cell line was isolated using Trizol reagent (Life Technologies, CA, USA) in accordance with manufacturer’s instructions. RNA concentration and purity were assessed by Nanodrop ND1000 spectrophotometer (Nanodrop Technologies, Rockland, USA). The cDNA was synthesized with M-MLV Reverse Transcriptase (Sigma, St. Louis, Missouri, USA) from 1 μg total RNA. Real-time PCR samples were prepared in 12.5 μL (final volume) which was composed of 6.25 μL FastSYBRGreen Master Mix (Applied Biosystems, CA, USA), 0.4 μL primer pair solution (0.2 μM final concentration of each primer), 5.05 μL of water, and 1 μL of diluted cDNA. Primer sequences and the expected PCR fragment sizes are listed in Table 2. Real-time PCRs were carried out in the Applied Biosystems StepOnePlus Real-Time PCR cycler and performed in duplicates. All results were analyzed by the 2−ΔΔCt method [33]. GAPDH expression was used as the internal control gene for all relative expression calculations.
Cell Counting Assay
C6 or U87MG glioma cells were seeded at 5 × 104 cells/well in DMEM/10% FBS in 12-well plates. Following treatment and CD73 knockdown procedures, medium was removed; cells were detached with 0.25% trypsin solution (Gibco BRL, Carlsbad, USA) and counted with trypan blue exclusion. Data were expressed as the percentage of proliferation inhibition rate compared with DMEM/10% FBS or GFP-siRNA treated cells (control set at 0%).
Clonogenic Assay
For the clonogenic assay, cells were exposed to APCP (1, 10, and 100 μM) or CD73-siRNA. After 72 h, the treatment was removed; cells were washed twice with PBS, harvested and seeded at a density of 100 cells/well in a 6-well plate. After 14 days, colonies were fixed with 100% methanol, followed by staining with 0.1% crystal violet. The number of colonies was counted and single colonies were photographed for analysis. Data were expressed as the percentage of colony number compared with DMEM/10% FBS or GFP-siRNA treated cells (control set at 100%).
Propidium Iodide Assay
Cell damage was assessed by fluorescent image analysis of propidium iodide (PI) uptake. At the end of treatment, C6 glioma cells were incubated with PI (7.5 μM) for 1 h. PI fluorescence was excited at 515–560 nm using an inverted microscope (Olympus BX-50, Tokyo, Japan) fitted with a standard rhodamine filter. Images were captured using a digital camera connected to the microscope.
Cell Cycle Assay
After treatments, cells were trypsinized, centrifuged, and resuspended in 1.5 mL 70% ethanol followed by incubation with staining buffer (10 mM PBS, 1% v/v Triton x-100, 20 mg/mL RNAse, and 2 mg/mL PI, pH 7.4) for 30 min in the dark. DNA content was analyzed in a FACSCalibur Flow Cytometer (BD Biosciences, San Jose, USA).
Akt and NF-kB Expression Assay
Human U87MG glioma cells were seeded in 6-well plates at density of 1 × 105 cells/well and treated with APCP (100 μM) or exposed to CD73-siRNA complexes. Following 72 h of treatment, ADO (1 μM) was added and the treatment remained for further 48 h. Control cells were exposed to DMEM/10% FBS or GFP-siRNA scramble. Cells were washed with PBS and fixed with Fosflow BD Fix buffer for 10 min at 37 °C. Cells were then permeabilized with BD Phosflow Perm buffer for 30 min at 4 °C and incubated with mouse anti-human-pAkt-PE antibody (pT308, Cat. No. 558275; BD Biosciences) and Alexa Fluor®488 mouse anti-human-NF-kB-p65 (pS529, Cat. No. 558423; BD Biosciences) for 30 min at RT in the dark. Cell fluorescence was measured with a FACSCalibur Flow Cytometer according to the manufacturer’s instructions (BD Biosciences, San Jose, USA).
Characterization of CD73 Role in In Vivo Glioma Progression
Glioma Implantation
C6 glioma cells were cultured to approximately 90% confluence and a total of 1 × 106 cells in 3 μL of DMEM/10% FBS was injected in the right striatum at a depth of 6.0 mm (coordinates relative to bregma, 3.0 mm lateral and 0.5 posterior) of male Wistar rats (250–300 g, 8 weeks old) anesthetized by intraperitoneal (i.p.) administration of ketamine and xylazine. Animals were euthanized following 23 days of glioma implantation [34]. All procedures used in the present study followed the Guide for the Care and Use of Laboratory Animals from NIH and were approved by the Ethical Committee of Universidade Federal de Ciências da Saúde de Porto Alegre (protocol number 293/14).
Treatment of Animals
Seven days after glioma implantation, animals were randomly divided into four groups as follows: (1) control (5 μL of PBS); (2) APCP (2 μg prepared in 5 μL of PBS); (3) scramble-GFP-siRNA (9 pg of sequence complexed with 5 μL of Lipofectamine RNAiMax); and (4) CD73–980-siRNA (9 pg of sequence complexed with 5 μL of Lipofectamine RNAiMax). Treatments were administered to ketamine/xylazine anesthetized rats via intracerebroventricular (icv) injection into the cisterna magna once a week for 3 weeks (days 7, 14, and 21) using an infusion pump at rate of 0.5 μL/min. After 23 days of glioma implantation, rats were decapitated and brains were removed, sectioned, and frozen. Blood samples were collected from all animals for posterior enzyme assays (alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, and urea), and serum or cerebrospinal fluid (CSF) were collected to determine levels of purine compounds. For body weight analysis, the experimental groups were compared with control group.
Pathological Analysis and Tumor Quantification
At least three hematoxylin and eosin (HE) sections (4 μm frozen tissue) from each animal were analyzed by a pathologist blinded to the experiment. For tumor size quantification, images were captured using a digital camera connected to a microscope (× 2 magnification; Nikon Eclipse TE300) and the tumor area (mm2) was determined using ImageJ software. The total volume (mm3) of the tumor was computed by the multiplication of the slice sections and by summing the segmented areas [34].
Analysis of Purine Compound Levels in Serum or Cerebrospinal Fluid
Cerebrospinal fluid (CSF) was drawn (80–100 μL/rat) by direct puncture of cisterna magna using an insulin syringe (27 gauge × 1/2 in length) in Wistar rats anesthetized with ketamine/xylazine on the 23rd day after glioma implantation. Samples were stored at − 80 °C for further analysis. Serum proteins were denatured with 1 M HClO4. All samples were then centrifuged (13,000×g for 1 min at 4 °C), supernatants were neutralized with 4 N KOH and clarified by a second centrifugation (13,000×g for 1 min at 4 °C). Samples were collected and stored at − 80 °C for further analysis.
ATP, ADP, AMP, adenosine, inosine, and hypoxanthine were analyzed by HPLC according to Bertoni et al. [11]. Briefly, 50 μL of samples (CSF or serum) were applied to a reversed-phase HPLC Shimadzu Prominence (Shimadzu, Kyoto, Japan) using a Shimadzu column Shim-pack CLC (M) C18 (150 × 4.6 mm × 5 μm) attached to a guard column Shimadzu Shim-pack GVP-ODS (4.6 × 10 mm). The elution was carried out by applying a linear gradient from 100% solvent A (60 mM KH2PO4 and 5 mM of tetrabutylammonium chloride, pH 5.0) to 100% of solvent B (solvent A plus 30% methanol) over a 42-min period (flow rate at 1.0 mL/min). Mobile phases were filtered through a 0.45 μm Millipore filter prior to analysis, and all the applied reagents were HPLC grade. The amount of purine compounds and metabolic residues were measured by absorption at 254 nm. The retention time of standards was used as parameter for identification and quantification by comparison of the peak area. Purine levels were expressed as micromolars.
Statistical Analysis
Statistics were performed using GraphPad Prism 5 software (San Diego, USA); data were expressed as mean ± SD and subjected to one-way analysis of variance (ANOVA) followed by Tukey-Kramer post hoc test (for multiple comparisons) or Student’s t test as appropriate. Differences were considered significant for a p value of *p < 0.05, **p < 0.01, and ***p < 0.001, respectively.
Results
CD73 Exerts a Proliferative Stimulus in Glioblastoma Cell Lines Via ADO Production and A1 Receptor Sensitization
First, to determine CD73 role in in vitro glioma progression, rat C6 and human U87MG glioma cell lines were treated with increasing concentrations of either the enzyme product (ADO), the substrate (AMP), or the pharmacological inhibitor (APCP), and after 24, 48, or 72 h of treatment, cell viability was evaluated (Fig. 1). Alternatively, knockdown of the CD73 gene was performed using siRNA technology. Following 72 h of silencing protocol, CD73 expression and activity was decreased, respectively, by 60/70% in C6 and 70/70% in U87MG cells (Fig. S1e, f, and j). Similarly, APCP treatment decreased AMPase activity in a concentration-dependent manner in all evaluated cell lines (Fig. S1g, h).
CD73 expression and enzyme activity modulates glioma cell viability. Glioma cells were exposed to increasing concentrations of ADO (a, b), AMP (c, d), and APCP (e, f) as indicated, or CD73 was knockdown by CD73-siRNA (g, h). Cell viability was assessed in C6 (left) and U87MG (right) glioma cells by MTT following 24, 48, and 72 h of treatment or transfection with siRNA. Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. Values represent mean ± SD of at least three independent experiments carried out in triplicate. Data were analyzed by ANOVA followed by post hoc comparisons (Tukey-Kramer test). *p < 0.05, **p < 0.01, and ***p < 0.001, significantly different from control cells. ADO, adenosine; APCP, adenosine 5′-(α,β-methylene)diphosphate: CD73 enzyme inhibitor
Adenosine treatment exhibited differential effects on C6 and U87MG glioma cell lines and induced a biphasic effect on cell viability, which was time and concentration dependent. When C6 cells were exposed to ADO, a significant increase in cell viability was observed after 24 h of treatment at lowest concentrations, reaching up to 50% following 1 μM ADO exposure. However, further increases in ADO concentration (up to 250 μM) did not affect cell viability, neither did longer exposure times (48 and 72 h) (Fig. 1a). Interestingly, ADO had no effect on U87MG cell viability after 24 h of treatment. However, after 48 h, low (1 and 10 μM) and high (250 μM) ADO concentrations increased U87MG cell viability by 30, 40, and 50%, respectively. In contrast with C6 cells, higher ADO concentrations and longer exposure times (72 h) reduced U87MG viability by up to 50% when cells were treated with 250 μM ADO (Fig. 1b).
The substrate of CD73 (AMP) had a similar effect in the two cell lines, leading to a decrease in cell viability at higher concentrations and longer times of exposure. In this sense, 1 mM AMP reduced ~ 50% glioma cell viability following 72 h of treatment (Fig. 1c, d). Finally, APCP treatment reduced C6 cell viability by up to 30%, reaching a plateau at the 50 μM concentration, while in U87MG, this effect was observed at higher APCP concentrations (250 and 500 μM) (Fig. 1e, f). Corroborating the influence of CD73 on glioma viability, CD73 knockdown decreased glioma viability by ~ 20% in both cell lines (Fig. 2g, h). Therefore, low ADO concentrations promoted glioma cell viability, while AMP and strategies to inhibit endogenous extracellular ADO formation, such as CD73 pharmacological inhibition or knockdown, had the opposite effect.
Adenosine increases glioma cell viability via A1 receptor sensitization. Analysis of caffeine, dipyridamole, DPCPX, Zm241385, and IB-MECA treatments in C6 and U87 glioma cell viability. Glioma cells were exposed to caffeine (10 μM), dipyridamole (100 μM), DPCPX (1 μM), Zm241385 (1 μM), and IB-MECA (20 μM) 30 min prior ADO (1 μM). Cell viability was assessed by MTT after 24 h (C6; a–c) or 48 h (U87MG; b–d) of treatment. Control cell were exposed to DMSO (0.01%). Values represent the mean ± SD of at least three independent experiments carried out in triplicate. Data were analyzed by ANOVA followed by post hoc comparisons (Tukey-Kramer test). *p < 0.05, **p < 0.01, and ***p < 0.001, significantly different from control cells. Caffeine, non-selective P1R antagonist; dipyridamole, uptake nucleoside inhibitor; DPCPX, selective A1 antagonist; Zm241385, selective A2A antagonist; IB-MECA, selective A3 agonist
To better understand how ADO increases glioma viability, C6 and U87MG cells were treated with the non-selective P1 receptor (P1R) antagonist caffeine (10 μM) and the nucleoside uptake inhibitor dipyridamole (100 μM) for 30 min prior to 1 μM ADO exposition, as described in “Materials and Methods.” In accordance with P1R participation, caffeine prevented the proliferative effect induced by ADO in both cell lines, resulting in levels comparable with the control (Fig. 2a, b). The inhibition of nucleoside uptake by dipyridamole prevented the increase of cell viability promoted by ADO (Fig. 2a, b). Interestingly, dipyridamole alone also decreased U87MG cell viability by 50% when compared with the control (Fig. 2b).
Those effects are probably related to an increased ADO availability in the extracellular environment, which in turn activates P1 receptors in a differential and selective fashion (A1 > A2A > A2B > A3) [35]. Therefore, cells were further exposed to selective receptor antagonists to investigate which of them were involved in the biphasic ADO effects in glioma cell viability. Interestingly, the selective A1 receptor antagonist DCPCX (1 μM) prevented ADO effect in C6 and U87MG cells, with viability comparable with the control, while the A2A receptor antagonist Zm241385 (1 μM) did not induce any changes. Finally, the selective A3R agonist IB-MECA (10 μM) induced a decrease of ~ 20% in glioma cell viability, which was not different from the control. Taken together, this data points to a key role of A1R in proliferative actions mediated by ADO, which could be sensitized by low ADO concentrations, a condition that is possibly present at the TME as a result of CD73 activity [8, 9]. Indeed, CD73 has been characterized as the main extracellular ADO source [36]. Its increased expression reported in glioblastoma may be important for both ADO production and AMP scavenger activity, since AMP is a potential cytotoxic nucleotide. Therefore, a strategy aiming to downregulate/inhibit CD73 expression may be promising.
Downregulation of CD73 Negatively Regulates In Vitro Glioma Growth Via Akt/NF-kB Pathways and Potentiates TMZ Cytotoxicity
To further evaluate the impact of CD73 knockdown and pharmacological inhibition on in vitro glioma cell proliferation, a set of experiments was performed aiming to identify potentially involved cellular pathways. CD73 silencing resulted in ~ 30 and ~ 40% inhibition of C6 and U87MG cell proliferation, respectively, as assessed by cell counting, and ADO treatment restored the proliferation in those cells (Fig. 3a, b). ADO effect was stronger in U87MG when compared with C6 cell line. Additionally, CD73 knockdown in C6 cells decreased colony formation by 35% (Fig. 3c, d); promoted positive PI staining, suggestive of necrosis (Fig. 3e); and induced a discrete 4% cell population at Sub-G1 cell cycle phase, indicating apoptosis (Fig. 5f). APCP treatment reduced cell proliferation by 20% in C6 cells at 10 and 100 μM, while in U87MG, the 25% proliferation reduction was observed only at a higher concentration (100 μM). In addition, all APCP tested concentrations decreased C6 colony formation index in 33% and caused a 2% increase in the sub-G1-phase cell population (Fig. 3a–f).
CD73 downregulation reduces glioma cell proliferation and malignancy parameters. Glioma cells were exposed to increasing concentrations of APCP as indicated, CD73 was knockdown by CD73-siRNA, and cells were exposed or not to ADO (1 μM). Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. Cell proliferation was assessed by cell counting in C6 (a) and U87MG (b); colony formation was analyzed by clonogenic assay in C6 cells (c, d); necrosis was evaluated by PI incorporation in C6 cells (e). Fluorescence (right) and phase contrast (left) (× 4 magnification); FACS was used for determination of cell cycle distribution (sub-G1, G1/G0, S, and G2/M) in C6 cells (f). Results were analyzed by one-way ANOVA, followed by Tukey’s multiple comparisons test. All experiments were repeated at least three times. **p < 0.01 and ***p < 0.001, significantly different from control groups; ###p < 0.001, significantly different from scramble GFP-siRNA-transfected cells; &p < 0.05 and &&&p < 0.001, significantly different from CD73-siRNA transfected cells treated with ADO. ADO, adenosine; APCP, adenosine 5′-(α,β-methylene)diphosphate: CD73 enzyme inhibitor
Cell signaling via p-Akt and NF-kB-p65 pathways are well-known modulators of glioblastoma proliferation [37,38,39,40]. To investigate the mechanisms involved in the antiproliferative effects induced by CD73 dowregulation or enzyme inhibition, p-Akt and NF-kB-p65 levels were investigated using flow cytometry in U87MG cells. Interestingly, CD73 knockdown upregulated p-Akt and NF-kB-p65 by 200 and 80%, respectively, when compared with control cells, and ADO supplementation did not reverse this effect. Moreover, ADO or APCP treatments did not cause changes in these pathways. These results suggest that blockade of CD73 expression promotes p-Akt/NF-kB-p65 signaling in glioma cells and that these effects are independent of ADO signaling (Figs. 4 and 5).
CD73 knockdown positively modulates Akt signaling pathway in glioma cells. U87MG glioma cells were exposed to APCP (100 μM), CD73 was knockdown by CD73-siRNA, and cells were exposed or not to ADO (1 μM) for 48 h. Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. p-Akt binding was measured by FACS. a–f Histograms represent unstained cells (gray) and treated cells (white). g Quantitative analysis of p-Akt immunocontent. Values represent means ± SD and were analyzed by one-way ANOVA followed by post hoc comparisons (Tukey). Asterisk, significantly different from the control DMEM/10% FBS group, number sign, significantly different from scramble GFP-siRNA group; and ampersand, significantly different from adenosine (ADO)-treated group
CD73 knockdown positively modulates NF-kB signaling pathway in glioma cells. U87MG glioma cells were exposed to APCP (100 μM), CD73 was knockdown by CD73-siRNA, and cells were exposed or not to ADO (1 μM) for 48 h. Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. NF-kB-p65 binding was measured by FACS. a–f Histograms represent unstained cells (gray) and treated cells (white). g Quantitative analysis of NF-kB-p65 immunocontent. Values represent means ± SD and were analyzed by one-way ANOVA followed by post hoc comparisons (Tukey). Asterisk, significantly different from the control DMEM/10% FBS group; number sign, significantly different from scramble GFP-siRNA group; and ampersand, significantly different from adenosine (ADO)-treated group
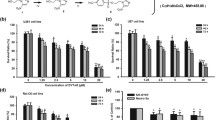
CD73 participation in chemoresistance was also investigated (Fig. 6). Glioma cells were treated with 100 μM TMZ alone, which did not induce a cytotoxic effect (Fig. 6), or in combination with APCP, the CD73 enzyme inhibitor. TMZ treatment reduced cell viability by 20% on average in glioma cells which were previously silenced for CD73 or treated with APCP (Fig. 6a, b). In addition, IC50 value for TMZ was decreased by ~ 100 μM when combined therapies were applied (Fig. 6c). Taken together, this data indicate that CD73 inhibition potentiates TMZ cytotoxicity in glioblastoma cells.
CD73 downregulation potentiates TMZ-induced glioma cytotoxicity. C6 and U87MG glioma cells were exposed to APCP (100 μM) or CD73 was knockdown by CD73-siRNA prior 72 h of TMZ (100 μM). Cell viability was determined by MTT assay following 72 h of treatment. Values represent means ± SD and were analyzed by one-way ANOVA followed by post hoc comparisons (Tukey). Asterisk, significantly different from the control DMEM/10% FBS group; number sign, significantly different from APCP or CD73-siRNA alone. APCP, adenosine 5′-(α,β-methylene)diphosphate: CD73 enzyme inhibitor; TMZ, temozolomide
CD73 Regulates In Vitro Glioma Cell Migration and Invasion Via ADO Production, Metalloproteinase-2, and Vimentin Expression
In addition to generating extracellular ADO via AMPase activity, CD73 has been reported as an important cell-cell and cell-extracellular matrix adhesion protein. Both CD73 roles are related to cell migration and adhesion processes in glioma cells [11, 17]; however, the mechanisms involved remain unclear. Experiments were performed in CD73-silenced cells treated either with ADO (1 μM) or in the presence of CD73 enzyme inhibitor APCP. Cell migration was analyzed by scratch-wound and transwell assays, whereas cell invasion was performed in Matrigel-coated transwell inserts. Further, analysis of metalloproteinase-2 (MMP-2) and vimentin mRNA expression was performed. The scratch-wound assay indicates that ADO induced a sustained C6 and U87MG glioma cell migration through 24 h of analysis. CD73 knockdown (Fig. 7a, b) or inhibition by APCP (Fig. 7c, d) impaired glioma cell migration, while supplementing ADO to silenced cells recovered cell migration potential (Fig.7a, b; Fig. 2S). Similar results were obtained analyzing C6 glioma cell migration and invasion using transwell apparatus. ADO increased cell migration and invasion by ~ 18 and ~ 40%, respectively (Fig. 7e–i), which was followed by a parallel increase of 150% in MMP-2 and 360% in vimentin expression (Fig. 7g, h). Conversely, CD73 knockdown decreased by cell migration and invasion potential by ~ 60 and ~ 80%, respectively (Fig. 7e–i). At this condition, MMP-2 expression was abolished, and vimentin was decreased by 80% (Fig. 7g, h). ADO treatment in CD73-silenced cells restored migration and invasive potential of glioma cells without significantly increasing MMP-2 and vimentin expression. Consistent with a positive stimulus of ADO for cell migration, APCP reduced migratory and invasive potentials by 40 and 75%, respectively (Fig. 7e–i). APCP also induced an unexpected 210% increase in vimentin expression (Fig. 7h). These data suggest that CD73 and ADO play a role in glioma cell migration and invasion, and that these effects are mediated at least in part by MMP-2 and vimentin expression modulation.
CD73 downregulation reduces glioma cell migration and invasion by modulating MMP-2 and vimentin expression. C6 and U87MG glioma cells were exposed to increasing concentrations of APCP as indicated, CD73 was knockdown by CD73-siRNA, and cells were exposed or not to ADO (1 μM). Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. The average of initial wound width was measured and defined as 0% of cell migration. Images were collected at increasing times following scratch-wound, as indicated, using an inverted microscope. a–d Lines represent the mean percentage of migrating cells relative to negative control (DMEM/0.5% FBS). e, f Migration and invasion assays, respectively. C6 glioma cells were seeded in DMEM only in the upper chamber of uncoated or Matrigel-coated chambers of transwells. DMEM/10% FBS was added to the lower chamber, and cell migration and invasion was determined following 24 h of incubation. Columns represent the mean percentage of cells migrating to or invading the lower chamber. g, h Relative MMP-2 and vimentin mRNA expression in C6 cells was determined by qPCR using 2−ΔΔCt method. mRNA expression was normalized against GAPDH levels and expressed relatively to control (cells maintained in DMEM/0.5% FBS). i Representative light microscopy image of crystal violet-stained cells.*p < 0.05,**p < 0.01, and ***p < 0.001, significantly different from control cells; #p < 0.05, ##p < 0.01, ###p < 0.001, significantly different from adenosine (ADO)-treated group, as determined by ANOVA followed by Tukey’s test. All experiments were repeated at least three times
CD73 Knockdown or Enzyme Inhibition Suppress In Vivo Glioblastoma Growth
Considering the promising effect of CD73 silencing or inhibition in impairing in vitro glioma proliferation, we evaluated the potential of CD73 as a target for in vivo antiglioma therapy. In line with this, C6 glioma cells were implanted by intracranial injection into adult Wistar rats, and the animals were treated with either the selective CD73 enzyme inhibitor APCP or CD73siRNA sequences complexed with lipofectamine, as described in “Materials and Methods.” Control animals were treated with either PBS or GFP-siRNA-scramble sequence. Twenty-three days after glioma implantation, rat brains were frozen and the tissue blocks were processed for HE staining. Corroborating the previous findings, which indicate an in vitro proliferative role for CD73 in glioblastoma, APCP treatment reduced tumor volume by 40% (108.3 ± 36.3 mm3), while CD73 silencing reduced tumor volume by 45% (97.2 ± 52.9 mm3) when compared with control group PBS (173.2 ± 24.2 mm3) or GFP-siRNA-scramble (137.6 ± 33.1 mm3) (Fig. 8a–e). In addition, in one animal from each group (APCP and CD73-siRNA), a complete tumor remission was observed, remaining at the end of treatment only regions of necrosis and edema at glioma implantation site. Interestingly, histopathological analysis revealed increased necrosis and lymphocytic infiltration in CD73-siRNA-treated animals compared with the control group (Table 3; Fig. 9). These data indicate that CD73 downregulation may be an interesting strategy to control in vivo glioma growth.
CD73 inhibition decreases in vivo glioma growth. C6 glioma cells were implanted in rat brain, and the treatment was performed via icv as indicated. Tumor size was measured 23 days after glioma implantation by analyzing three HE sections of each tumor. HE images were captured using a digital camera connected to a microscope, and total volume (mm3) was determined using ImageJ Software. Values represent the mean ± SD of at least five animals per group. Data were analyzed by ANOVA followed by post hoc comparisons (Tukey’s test). *p < 0.05, significantly different from control groups (saline)
Representative HE sections of implanted tumors denote histological characteristics that define glioblastoma multiforme. Necrosis (N), peritumoral edema (PE), lymphocytic infiltration (LI), and vascular proliferation (V). Note that CD73-siRNA treatment increased necrosis and lymphocytic infiltration in implanted gliomas. The complete histopathological analysis is shown in Table 3; × 20 magnification (× 40 magnification insert)
To evaluate the impact of CD73 knockdown or inhibition by APCP on nucleotide/nucleoside levels, HPLC analysis was performed in CSF and serum. Data from literature show that levels of ATP, ADP, AMP, ADO, inosine, and hypoxanthine in CSF from healthy rats are very low, at the picomoles per microliter range [41]. However, our data show that rats submitted to glioma implantation model exhibited on average a 1000-fold increase of these levels, emphasizing the importance of purinergic signaling in glioma progression (Table 4).
In CSF, APCP-treated group achieved a 95% reduction of ADO levels, confirming CD73 inhibition (0.217 ± 0.08 μM), and a 550% increase in hypoxanthine levels (2.993 ± 0.95 μM) when compared with the control (PBS group) (ADO 6.828 ± 3.39 μM and hypoxanthine 0.458 ± 0.33 μM). In addition, in the CD73-silenced group, ADP and ADO levels were reduced by 80 and 97%, respectively (0.264 ± 0.05 and 0.220 ± 0.04 μM), and the hypoxanthine level increased (440%—2.489 ± 0.29 μM) when compared with the GFP-siRNA control group (7.420 ± 2.61 and 2.024 ± 0.15 μM) (Table 4).
In serum, APCP treatment promoted a decrease in ATP (80%—2.673 ± 0.78 μM), ADP (95%—0.106 ± 0.07 μM), ADO (30%—5.556 ± 0.48 μM), and inosine levels (60%—1.060 ± 0.16 μM) when compared with the control group ATP (15.407 ± 1.34 μM), ADP (2.392 ± 0.92 μM), ADO (7.593 ± 0.84 μM), and inosine (2.349 ± 0.43 μM). Moreover, in the group treated with CD73-siRNA complexes, a decrease in ATP (50%—7.753 ± 0.97 μM), ADP (99%—0.018 ± 0.03 μM), and inosine (50%—1.183 ± 0.09 μM) levels was observed (Table 4). Notably, although serum purine levels were altered in animals, no significant damage was detected (Table 4).
Finally, it is important to note that animals treated with CD73-siRNA or APCP did not exhibit mortality, behavioral changes, or weight loss (data not shown). Neither changes in renal and hepatic damage markers (ALT, AST, creatinine, and urea) (Table 5) nor histopathological changes in heart, liver, kidney, lung, and spleen were observed (Fig. S3), indicating the safety of these treatments.
Discussion
The present study demonstrates that CD73 knockdown or inhibition decreases in vitro and in vivo glioblastoma growth, suggesting CD73 as an interesting target for brain tumor therapy. First, the role of CD73 in in vitro glioma growth was investigated. We found that CD73 favors glioblastoma cell growth via ADO generation, and A1R sensitization exerts a proliferative stimulus to glioma cells. In line with this, CD73 downregulation decreased cell colony number, cell proliferation and potentiated TMZ cytotoxicity via Akt and NF-kB signaling pathways. Cell migration and invasion were also affected when targeting CD73, which was associated, at least in part, by decreasing expression of MMP-2 and vimentin. Finally, CD73 downregulation also decreased in vivo glioma growth, which was followed by decreased ADO levels in CSF, indicating the participation of this nucleoside in glioma progression.
Our research group has previously shown that alterations on purinergic signaling are involved in glioma progression [11, 17, 42,43,44,45,46,47]. Glioma cells exhibit low ATPase/ADPase activities and high AMPase activity when compared with normal astrocytes [42]. In this regard, higher CD73 expression in gliomas contributes to cell proliferation [17] and modulates cell adhesion and tumor cell-extracellular matrix interactions [11]. However, the mechanisms involved in these effects in gliomas were not determined. Additionally, data from the literature show that extracellular ADO plays an important role in brain physiology, has essential neuromodulatory, anticonvulsive, and neuroprotective roles [48, 49]. In the context of cancer, ADO accumulation favors tumor growth, metastasis, angiogenesis, chemoresistance, and immunosuppression [8, 9]. Other ectonucleotidases, such as PAP, are also expressed by cancer cells and may be important for ADO generation from AMP hydrolysis in acid TME [4,5,6,7, 36]. PAP selective expression in GSC under hypoxia conditions has recently been described and deserves further analysis. Here, we hypothesized that strategies to blockade CD73 would decrease ADO availability in TME, which would in turn decrease tumor progression. Corroborating this idea, the results presented here show the positive role of CD73 in the control of migration, invasion, malignancy, chemoresistance to TMZ and in vivo glioma progression. Conversely, CD73 inhibition by APCP or siRNA technology prevented the positive actions of CD73 on tumor growth. These data reinforce its potential as a therapeutic target for glioblastoma.
Interestingly, in this context, our data showed that CD73 knockdown promoted activation of p-Akt/NF-kB-p65 signaling. Although these pathways have been classically related to cell proliferation and survival [37,38,39,40], a disturbance in Akt signaling has also been associated with the antiglioma activity of natural compounds [37,38,39,40, 50]. Accordingly, our data suggest that Akt/NF-kB-p65 activation in CD73 knockdown cells may be related to decreased glioma progression. Nonetheless, the overstimulation of these proteins as a compensatory survival signaling in injured glioma cells, as observed in previous studies [51, 52], could not be excluded. Therefore, whether the increase of Akt/NF-kB-p65 activity promoted by CD73 knockdown is associated to cell death or to a resistance process remains to be evaluated in further studies. Indeed, Akt/NF-kB-p65 biological effects are dependent on the stimulation degree, which may result in opposite outcomes [50, 53]. To the best of our knowledge, this is the first study demonstrating Akt/NF-kB-p65 modulation by CD73 in an ADO-independent way.Our data show that CD73 plays a role in the control of glioma cell migration and invasion through an adenosinergic pathway. In this context, CD73 downregulation decreased cell migration and invasion, while ADO supplementation reversed these effects. Others have previously shown that RNAi-mediated knockdown of CD73 in breast cancer cells inhibited their invasion potential [54]. However, previous studies did not address the mechanism of action by which CD73 may modulate tumor-cell migration and invasion. Interestingly, our data suggest that it may be through modulation of the expression of MMP-2 and vimentin. The migration and invasion potentials of malignant tumors require the proteolytic degradation of ECM components. Previous data has established that MMPs are associated with pathological hallmarks of glioblastoma aggressiveness and its inhibition might be useful for treating brain tumor invasiveness [55, 56]. In addition, vimentin, a protein of intermediate filament family, participates in several cell signal pathways to modulate the motility and invasion of cancer cells; in patients with glioblastoma, low vimentin expression is associated with longer survival and a better response to temozolomide therapy [57, 58]. Therefore, we suggest a novel mechanism by which CD73 controls migration, invasion and malignancy of the glioma.
Glioblastoma cells are characterized by high chemoresistance to antitumor drugs [22, 23]. Therefore, the available chemotherapy for treatment of brain tumors has, for decades, made only a very limited contribution. In the present study, we reveal CD73 as a possible target to potentiate TMZ cytotoxicity against glioblastoma cells. Our data showed that CD73 blockade increased sensitivity to TMZ by lowering its IC50 and by inducing a decrease of cell viability of two highly chemoresistant glioma cell lines following a non-cytotoxic drug concentration treatment. Currently, studies have shown that CD73 suppresses chemotherapy efficacy, conferring glioma chemoresistance to vincristine in a P1 receptor-dependent effect [18]. Indeed, CD73 knockdown or activity inhibition, as well as A3 antagonism, decreased multiple drug resistance protein-1 (MRP1) expression and glioblastoma stem-like cell proliferation following chemotherapeutic treatment [18,19,20]. In addition, CD73 mediates multiresistance in leukemia by interacting with TRAIL receptor [59], and breast cancer doxorubicin resistance by suppressing adaptive antitumor immune response via ADO receptor activation [60]. In this context, modulation of glioma chemoresistant phenotype by CD73 inhibition may play a role in TMZ therapy.
In addition, we demonstrated that the TME of glioblastoma in vivo presents high concentrations of ADO in the CSF, in the micromolar range. This data is consistent with studies showing high ADO levels in the TME, while levels in healthy tissue are around the nanomolar range [41]. On the other hand, siRNA-CD73 or APCP downregulated the activity of the CD73. Consistently, both APCP treatment and CD73 knockdown reduced ADO levels in CSF when compared with untreated animals, restoring the concentrations of this nucleoside to the nanomolar range, which is found in healthy tissues. This treatment also reduced tumor size and increased necrosis and lymphocyte infiltration in the siRNA-CD73 group. In view of the data presented, we suggest that the reduction of the tumor growth after CD73 downregulation can be attributed to decreased ADO. In addition, we observed a decrease in ADP levels in the siRNA-CD73 group in CSF, which can justify the 5% higher decrease in tumor volume when compared with the APCP group (which did not cause change in ADP levels), since our research group has already demonstrated that the accumulation of ADP results in bigger and more malignant gliomas [46]. The present analysis reinforces the safety of CD73 inhibition, since no alterations were observed in behavior, hepatic or renal function, nor in histopathological characteristics of kidney, liver, lung, spleen, and heart tissues.
In conclusion, we found that CD73 silencing or enzyme inhibition decreased in vitro and in vivo glioblastoma growth. In addition, CD73 blockage potentiates TMZ induced glioma cytotoxicity. However, additional studies are needed to develop a formulation that increases the bioavailability of drugs to central nervous system and allows for a less invasive route of administration in the future.
Abbreviations
- TMZ:
-
Temozolomide
- ADO:
-
Adenosine
- CD73:
-
ecto-5′-nucleotidase
- APCP:
-
Adenosine 5′-(α,β-methylene)diphosphate
- TME:
-
Tumor microenvironment
- MMP-2:
-
Metalloproteinase-2
- icv:
-
Intracerebroventricular
- CSF:
-
Cerebrospinal fluid
- GPI:
-
Glycosylphosphatidylinositol
- DPCPX:
-
8-Cyclopentyl-1,3-dipropylxanthine
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- HPLC:
-
High-performance liquid chromatography
- MDR:
-
Multiple drug resistance
- Zm241385:
-
4-(2-[7-Amino-2-(2-furyl)[1,2,4]triazolo[2,3-a][1,3,5]triazin-5-ylamino]ethyl)phenol
- IB-MECA:
-
1-Deoxy-1-[6-[[(3-iodophenyl)methyl]amino]-9H-purin-9-yl]-N-methyl-β-d-ribofuranuronamide
References
Idzko M, Ferrari D, Eltzsching HK (2014) Nucleotide signalling during inflammation. Nature 15:310–317. https://doi.org/10.1038/nature13085
Di Virgilio F, Adinolfi E (2017) Extracellular purines, purinergic receptors and tumor growth. Oncogene 36:293–303. https://doi.org/10.1038/onc.2016.206
Burnstock G (2017) Purinergic signalling: therapeutic developments. Front Pharmacol 8:661. https://doi.org/10.3389/fphar.2017.00661
Zimmermann H (2009) Prostatic acid phosphatase, a neglected ectonucleotidase. Purinergic Signal 5(3):273–275. https://doi.org/10.1007/s11302-009-9157-z
Whiteside TL (2017) Targeting adenosine in cancer immunotherapy: a review of recent progress. Expert Rev Anticancer Ther 17:527–535. https://doi.org/10.1080/14737140.2017.1316197
Burnstock G, Knight GE (2017) The potential of P2X7 receptors as a therapeutic target, including inflammation and tumour progression. Purinergic Signal 14:1–18. https://doi.org/10.1007/s11302-017-9593-0
Zimmermann H, Zebisch M, Sträter N (2012) Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal 8(1):105–106. https://doi.org/10.1007/s11302-011-9256-5.
Antonioli L, Novitskiy SV, Sachsenmeier KF, Fornai M, Blandizzi C, Haskó G (2017) Switching off CD73: a way to boost the activity of conventional and targeted antineoplastic therapies. Drug Discov Today 22:1686–1696. https://doi.org/10.1016/j.drudis.2017.06.005
Kazemi MH, Raoofi Mohseni S, Hojjat-Farsangi M, Anvari E, Ghalamfarsa G, Mohammadi H, Jadidi-Niaragh F (2018) Adenosine and adenosine receptors in the immunopathogenesis and treatment of cancer. J Cell Physiol 233:2032–2057. https://doi.org/10.1002/jcp.25873
Ludwig HC, Rausch S, Schallock K, Markakis E (1999) Expression of CD 73 (ecto-5′-nucleotidase) in 165 glioblastomas by immunohistochemistry and electronmicroscopic histochemistry. Anticancer Res 19:1747–1752
Cappellari AR, Vasques GL, Bavaresco L, Braganhol E, Battastini AM (2012) Involvement of ecto-5′-nucleotidase/CD73 in U138MG glioma cell adhesion. Mol Cell Biochem 359:315–322. https://doi.org/10.1007/s11010-011-1025-9
Bertoni APS, de Campos RP, Tsao M, Braganhol E, Furlanetto TW, Wink MR (2018) Extracellular ATP is differentially metabolized on papillary thyroid carcinoma cells surface in comparison to normal cells. Cancer Microenviron 11:61–70. https://doi.org/10.1007/s12307-018-0206-4
Allard B, Beavis PA, Darcy PK, Stagg J (2016) Immunosuppressive activities of adenosine in cancer. Curr Opin Pharmacol 9:7–16. https://doi.org/10.1016/j.coph.2016.04.001.
Allard B, Turcotte B, Stagg J (2012) CD73-generated adenosine: orchestrating the tumor-stroma interplay to promote cancer growth. J Biomed Biotechnol 2012:1–8. https://doi.org/10.1155/2012/485156
Xu S, Shao QQ, Sun JT, Yang N, Xie Q, Wang DH, Huang QB, Huang B et al (2013) Synergy between the ectoenzymes CD39 and CD73 contributes to adenosinergic immunosuppression in human malignant gliomas. Neuro-Oncology 15:1160–1172. https://doi.org/10.1093/neuonc/not067
Bahreyni A, Khazaei M, Rajabian M, Ryzhikov M, Avan A, Hassanian SM (2018) Therapeutic potency of pharmacological adenosine receptor agonist/antagonist in angiogenesis, current status and perspectives. J Pharm Pharmacol 70:191–196. https://doi.org/10.1111/jphp.12844
Bavaresco L, Bernardi A, Braganhol E, Cappellari AR, Rockenbach L, Farias PF, Wink MR, Delgado-Cañedo A et al (2008) The role of ecto-5′-nucleotidase/CD73 in glioma cell line proliferation. Mol Cell Biochem 319:61–68. https://doi.org/10.1007/s11010-008-9877-3
Quezada C, Garrido W, Oyarzún C, Fernández K, Segura R, Melo R, Casanello P, Sobrevia L et al (2013) 5′-Ectonucleotidase mediates multiple-drug resistance in glioblastoma multiforme cells. J Cell Physiol 228:602–608. https://doi.org/10.1002/jcp.24168
Uribe D, Torres Á, Rocha JD, Niechi I, Oyarzún C, Sobrevia L, San Martín R, Quezada C (2017) Multidrug resistance in glioblastoma stem-like cells: Role of the hypoxic microenvironment and adenosine signaling. Mol Asp Med 55:140–151. https://doi.org/10.1016/j.mam.2017.01.009
Torres A, Vargas Y, Uribe D, Jaramillo C, Gleisner A, Salazar-Onfray F, López MN, Melo R et al (2016) Adenosine A3 receptor elicits chemoresistance mediated by multiple resistance-associated protein-1 in human glioblastoma stem-like cells. Oncotarget 7:67373–67386. https://doi.org/10.18632/oncotarget.12033
Koszałka P, Gołuńska M, Stanisławowski M, Urban A, Stasiłojć G, Majewski M, Wierzbicki P, Składanowski AC et al (2015) CD73 on B16F10 melanoma cells in CD73-deficient mice promotes tumor growth, angiogenesis, neovascularization, macrophage infiltration and metastasis. Int J Biochem Cell Biol 69:1–10. https://doi.org/10.1016/j.biocel.2015.10.003
Di Carlo DT, Cagnazzo F, Benedetto N, Morganti R, Perrini P (2017) Multiple high-grade gliomas: epidemiology, management, and outcome. A systematic review and meta-analysis. Neurosurg Rev. https://doi.org/10.1007/s10143-017-0928-7
Arévalo ÁST, Erices JI, Uribe DA, Howden J, Niechi I, Muñoz S, Martín RS, Monrás CAQ (2017) Current therapeutic alternatives and new perspectives in glioblastoma multiforme. Curr Med Chem 24:2781–2795. https://doi.org/10.2174/0929867324666170303122241
Wijaya J, Fukuda Y, Schuetz JD (2017) Obstacles to brain tumor therapy: key ABC transporters. Int J Mol Sci 27:12–18. https://doi.org/10.3390/ijms18122544.
Stupp R, van den Bent MJ, Hegi ME (2005) Optimal role of temozolomide in the treatment of malignant gliomas. Curr Neurol Neurosci Rep 5:198–206, 2005
Bernardi A, Bavaresco L, Wink MR, Jacques-Silva MC, Delgado-Cañedo A, Lenz G, Battastini AM (2007) Indomethacin stimulates activity and expression of ecto-5′-nucleotidase/CD73 in glioma cell lines. Eur J Pharmacol 13(569):8–15
Eusemann TN, Willmroth F, Fiebich B, Biber K, van Calker D (2015) Adenosine receptors differentially regulate the expression of regulators of G-protein signalling (RGS) 2, 3 and 4 in astrocyte-like cells. PLoS One 10(8):e0134934. https://doi.org/10.1371/journal.pone.0134934
Kim TH, Kim YK, Woo JS (2012) The adenosine A3 receptor agonist Cl-IB-MECA induces cell death through Ca2+/ROS-dependent down regulation of ERK and Akt in A172 human glioma cells. Neurochem Res 37(12):2667–2677. https://doi.org/10.1007/s11064-012-0855-5
Jiang J, Lan YQ, Zhang T, Yu M, Liu XY, Li LH, Chen XP (2015) The in vitro effects of caffeine on viability, cycle cycle profiles, proliferation, and apoptosis of glioblastomas. Eur Rev Med Pharmacol Sci 19(17):3201–3207
Chan K, Delfert D, Junger KD (1986) A direct colorimetric assay for Ca-ATPase activity. Anal Biochem 157:375–380
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein—dye binding. Anal Biochem 72:248–254
Iser IC, Ceschini SM, Onzi GR, Bertoni AP, Lenz G, Wink MR (2016) Conditioned medium from adipose-derived stem cells (ADSCs) promotes epithelial-to-mesenchymal-like (EMT-like) in glioma cells in vitro. Mol Neurobiol 53(10):7184–7199. https://doi.org/10.1007/s12035-015-9585-4
Livak KJ, Schmittgen TD (2011) Analysis of relative gene expression data using real-time quantitative PCR and the 2(2delta delta C(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
da Silveira EF, Azambuja JH, de Carvalho TR, Kunzler A, da Silva DS, Teixeira FC, Rodrigues R, Beira FT et al (2017) Synthetic 2-aryl-3-((piperidin-1-yl)ethyl)thiazolidin-4-ones exhibit selective in vitro antitumoral activity and inhibit cancer cell growth in a preclinical model of glioblastoma multiforme. Chem Biol Interact 266:1–9. https://doi.org/10.1016/j.cbi.2017.02.001
Ceruti S, Abbracchio MP (2013) Adenosine signaling in glioma cells. Adv Exp Med Biol 986:13–30. https://doi.org/10.1007/978-94-007-4719-7_2
Liu TZ, Wang X, Bai YF, Liao HZ, Qiu SC, Yang YQ, Yan XH, Chen J et al (2014) The HIF-2alpha dependent induction of PAP and adenosine synthesis regulates glioblastoma stem cell function through the A2B adenosine receptor. Int J Biochem Cell Biol 49:8–16. https://doi.org/10.1016/j.biocel.2014.01.007
Majewska E, Szeliga M (2017) AKT/GSK3β signaling in glioblastoma. Neurochem Res 42(3):918–924. https://doi.org/10.1007/s11064-016-2044-4
Cahill KE, Morshed RA, Yamini B (2016) Nuclear factor-κB in glioblastoma: insights into regulators and targeted therapy. Neuro-Oncology 18(3):329–339. https://doi.org/10.1093/neuonc/nov265
Zanotto-Filho A, Gonçalves RM, Klafke K, de Souza PO, Dillenburg FC, Carro L, Gelain DP, Moreira JC (2017) Inflammatory landscape of human brain tumors reveals an NFκB dependent cytokine pathway associated with mesenchymal glioblastoma. Cancer Lett 390:176–187. https://doi.org/10.1016/j.canlet.2016.12.015
Zanotto-Filho A, Braganhol E, Klafke K, Figueiró F, Terra SR, Paludo FJ, Morrone M, Bristot IJ et al (2015) Autophagy inhibition improves the efficacy of curcumin/temozolomide combination therapy in glioblastomas. Cancer Lett 358(2):220–231. https://doi.org/10.1016/j.canlet.2014.12.044
Cruz Portela LV, Oses JP, Silveira AL, Schmidt AP, Lara DR, Oliveira Battastini AM, Ramirez G, Vinadé L et al (2002) Guanine and adenine nucleotidase activities in rat cerebrospinal fluid. Brain Res 950(1–2):74–78
Wink MR, Lenz G, Braganhol E, Tamajusuku AS, Schwartsmann G, Sarkis JJ, Battastini AM (2003) Altered extracellular ATP, ADP and AMP catabolism in glioma cell lines. Cancer Lett 198(2):211–218
Morrone FB, Oliveira DL, Gamermann P, Stella J, Wofchuk S, Wink MR, Meurer L, Edelweiss MI et al (2006) In vivo glioblastoma growth is reduced by apyrase activity in a rat glioma model. BMC Cancer 23(6):226
Morrone FB, Jacques-Silva MC, Horn AP, Bernardi A, Schwartsmann G, Rodnight R, Lenz G (2003) Extracellular nucleotides and nucleosides induce proliferation and increase nucleoside transport in human glioma cell lines. J Neuro-Oncol 64(3):211–218
Braganhol E, Huppes D, Bernardi A, Wink MR, Lenz G, Battastini AM (2008) A comparative study of ectonucleotidase and P2 receptor mRNA profiles in C6 cell line cultures and C6 ex vivo glioma model. Cell Tissue Res 335(2):331–340. https://doi.org/10.1007/s00441-008-0723-4
Braganhol E, Morrone FB, Bernardi A, Huppes D, Meurer L, Edelweiss MI, Lenz G, Wink MR et al (2009) Selective NTPDase2 expression modulates in vivo rat glioma growth. Cancer Sci 100(8):1434–1442. https://doi.org/10.1111/j.1349-7006.2009.01219.x
Braganhol E, Kukulski F, Lévesque AS, Fausther M, Lavoie EG, Zanotto-Filho A, Bergamin LS, Pelletier J et al (2015) Nucleotide receptors control IL-8/CXCL8 and MCP-1/CCL2 secretions as well as proliferation in human glioma cells. Biochim Biophys Acta 1852(1):120–130. https://doi.org/10.1016/j.bbadis.2014.10.014
Boison D (2013) Role of adenosine in status epilepticus: a potential new target? Epilepsia 6:20–22. https://doi.org/10.1111/epi.12268
Ballesteros-Yáñez I, Castillo CA, Merighi S, Gessi S (2017) The role of adenosine receptors in psychostimulant addiction. Front Pharmacol 8. https://doi.org/10.3389/fphar.2017.00985
Gentile MT, Ciniglia C, Reccia MG, Volpicelli F, Gatti M, Thellung S, Florio T, Melone MA et al (2015) Ruta graveolens L. induces death of glioblastoma cells and neural progenitors, but not of neurons, via ERK 1/2 and AKT activation. PLoS One 10(3):e0118864. https://doi.org/10.1371/journal.pone.0118864
Zhang T, Zheng C, Hou K, Wang J, Zhang Y, Fan Y, Zhao H, Qu X et al (2018) Suppressed expression of Cbl-b by NF-κB mediates icotinib resistance in EGFR-mutant non-small-cell lung cancer. Cell Biol Int. https://doi.org/10.1002/cbin.11026
Zhang H, Wang Q, Liu J, Cao H (2018) Inhibition of the PI3K/Akt signaling pathway reverses sorafenib-derived chemo-resistance in hepatocellular carcinoma. Oncol Lett 15(6):9377–9384. https://doi.org/10.3892/ol.2018.8536
Karin M, Lin A (2002) NF-kappaB at the crossroads of life and death. Send Nat Immunol 3(3):221–227. https://doi.org/10.1038/ni0302-221
Zhi X, Chen S, Zhou P, Shao Z, Wang L, Ou Z, Yin L (2007) RNA interference of ecto-5′-nucleotidase (CD73) inhibits human breast cancer cell growth and invasion. Clin Exp Metastasis 24:439–448
Yuan Y, Shen N, Yang SY, Zhao L, Guan YM (2015) Extracellular matrix metalloproteinase inducer and matrix metalloproteinase-2 overexpression is associated with loss of hormone receptor expression and poor prognosis in endometrial cancer. Oncol Lett 10:342–348
Musumeci G, Magro G, Cardile V, Coco M, Marzagalli R, Castrogiovanni P, Imbesi R, Graziano AC et al (2015) Characterization of matrix metalloproteinase-2 and -9, ADAM-10 and N-cadherin expression in human glioblastoma multiforme. Cell Tissue Res 362:45–60. https://doi.org/10.1007/s00441-015-2197-5
Lai SW, Huang BR, Liu YS, Lin HY, Chen CC, Tsai CF, Lu DY, Lin C (2018) Differential characterization of temozolomide-resistant human glioma cells. Int J Mol Sci 19. https://doi.org/10.3390/ijms19010127
Lin L, Wang G, Ming J, Meng X, Han B, Sun B, Cai J, Jiang C (2016) Analysis of expression and prognostic significance of vimentin and the response to temozolomide in glioma patients. Tumour Biol 37:15333–15339
Mikhailov A, Sokolovskaya A, Yegutkin GG, Amdahl H, West A, Yagita H, Lahesmaa R, Thompson LF, Jalkanen S, Blokhin D, Eriksson JE (2008) CD73 participates in cellular multiresistance program and protects against TRAIL-induced apoptosis. J Immunol 181:464–475
Loi S, Pommey S, Haibe-Kains B, Beavis PA, Darcy PK, Smyth MJ, Stagg J (2013) CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc Natl Acad Sci U S A 10:11091–11096. https://doi.org/10.1073/pnas.1222251110.
Funding
This study was supported by the Brazilian agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-Processo 422298/2016-6; 310846/2014-5), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS-Processo 16/2551-0000265-7; PRONEX-Processo 16/2551-0000473-0); J.H. Azambuja, N.E. Gelsleichter, L.R. Beckenkamp were recipients of CAPES or CNPq fellowships.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
Highlights
1) CD73/Adenosine regulates cell migration and invasion by expression of MMP-2 and vimentin;
2) Absence of CD73 induces glioma cell death via necrosis and potentiates temozolomide cytotoxicity;
3) Dowregulation of CD73 decreases glioma growth in vivo through reduction of adenosine (ADO);
4) Treatment of the animals did not induce systemic damage or mortality;
5) CD73, the enzyme responsible for the production of adenosine, is proposed to treat glioma.
Electronic Supplementary Material
Fig. S1
CD73 silencing and enzyme inhibition characterization in C6 and U87MG glioma cells. Evaluation of CD73 knockdown by siRNA-CD73 complexes. Glioma was transfected with siRNA-CD73 for 72 h, and knockdown efficiency was assessed by (a, b) FACS; AMPase activity evaluation in CD73 knockdown cells (d, e) or in APCP-treated cells (g, h) by malachite green method. j Immunofluorescence against CD73. Untreated or siRNA-GFP-transfected cells were considered controls. Values represent mean ± SD of at least three independent experiments carried out in triplicate. Data were analyzed by ANOVA followed by post hoc comparisons (Tukey-Kramer test). *p < 0.05, **p < 0.01, and ***p < 0.001, significantly different from control and #p < 0.05, ##p < 0.01, and ###p < 0.001, significantly different from siRNA-scramble (GFP) transfected cells. (PNG 260 kb)
Fig. S2
CD73 downregulation reduces glioma cell migration. C6 glioma cells were exposed to increasing concentrations of APCP as indicated, CD73 was knockdown by CD73-siRNA, and cells were exposed or not to ADO (1 μM). Control cells were exposed to DMEM or transfected with scramble GFP-siRNA. Images were collected at increasing times following scratch-wound, as indicated, using an inverted microscope. (PNG 3342 kb)
Fig. S3
Treatment of glioma-implanted rats with APCP or CD73/GFP-siRNA complexes does not promote histological changes in liver, heart, spleen, lung, and kidney tissues. To perform the histological analysis in peripheral organs, animals were killed 23 days after stereotaxic surgery and liver, kidney, heart, spleen, lung, and kidney were dissected, processed for standard hematoxylin/eosin staining, and the histological analysis was performed by a pathologist blinded to the study (× 20 magnification). The analyses were performed in five animals per group. (PNG 5420 kb)
Table S1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Azambuja, J.H., Gelsleichter, N.E., Beckenkamp, L.R. et al. CD73 Downregulation Decreases In Vitro and In Vivo Glioblastoma Growth. Mol Neurobiol 56, 3260–3279 (2019). https://doi.org/10.1007/s12035-018-1240-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1240-4