Abstract
MicroRNA-210 (miR-210) plays important roles in the regulation of cell growth, angiogenesis, and apoptosis in different cancer type. Previous study of miRNA expression profiling found that miR-210 was significantly elevated in osteosarcoma samples. However, its roles in this disease have not been fully elucidated. Thus, the aim of this study was to investigate the association of miR-210 expression with clinicopathologic features and prognosis in patients with osteosarcomas. Quantitative real-time reverse transcriptase-polymerase chain reaction analysis was performed to detect the expression level of miR-210 in cancerous and noncancerous bone tissues from 92 children treated for primary osteosarcomas. MiR-210 expression was significantly increased in osteosarcoma tissues compared with that in corresponding noncancerous bone tissues (P < 0.001). In addition, miR-210 upregulation more frequently occurred in osteosarcoma tissues with large tumor size (P = 0.02), poor response to preoperative chemotherapy (P = 0.008), and positive metastasis (P = 0.01). Moreover, miR-210 upregulation was associated with significantly decreased overall survival (P = 0.007) and progression-free survival (P = 0.01). In the Cox proportional hazard model, it was confirmed that its expression in the biopsy samples was an independent prognostic factor of unfavorable survival in osteosarcoma (for overall survival: P = 0.01; for progression-free survival: P = 0.02). These findings suggested that miR-210 upregulation showed a strong correlation with tumor aggressive progression of pediatric osteosarcoma and could help prognostic screening of patients with this malignancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteosarcoma is the most common malignant tumor of bone arising from primitive bone-forming mesenchymal cells and a leading cause of cancer-related death among children and adolescents [1]. It accounts for 60 % of pediatric bone tumors and has an annual incidence rate of 5.6 per million [2]. Despite recent advances in these multimodality treatments, the 5-year overall survival rate of osteosarcoma patients is only 28 % because of high frequency of pulmonary metastasis [3]. There are several factors, including the axial localization of the primary tumor, the tumor diameter of more than 8 cm, and the unfavorable histological response to preoperative chemotherapy, that influence the prognosis [4]. However, they have not been well established for the prediction of overall survival. Therefore, it is necessary to investigate the fundamental molecular mechanisms underlying the histological heterogeneity, drug resistance, and development of metastasis in order to identify novel markers for the diagnosis, the prognosis, and the treatment for patients with this disease.
MicroRNAs (miRNAs), the small (usually 18–25 nucleotides in length) and noncoding RNAs of endogenous origin, mainly function as negative regulators of gene expression [5]. The mature miRNA either destabilizes the target mRNA transcript or blocks its translation or both. In this way, miRNAs have the potential to regulate various critical biological processes, such as cell cycle, apoptosis, and differentiation [6]. Recently, expressions of miRNAs have been found to be deregulated in many sarcomas and are implicated in sarcoma pathobiology. Gene regulatory networks mediated by miRNAs in each sarcoma type could be potentially investigated in the prevention, diagnosis, prognosis, and as multimodal treatment options in these cancers [7]. Lulla et al. [8] identified 22 differentially expressed miRNAs in osteosarcomas. Interestingly, they found that miR-210, a key player in the hypoxic response, was significantly elevated in osteosarcoma tissues compared with the corresponding controls. According to our literature retrieval, miR-210 has been demonstrated to be upregulated in all cell types in hypoxic conditions [9]. It was also found to be a positive regulator of osteoblastic differentiation through the inhibition of activin A receptor type 1B [10]. Given the importance of this miRNA, further investigation on the role of miR-210 in osteosarcoma is required to be performed.
The aim of the present study was to investigate whether the assessment of miR-210 expression in biopsy samples in large series of clinically homogeneous pediatric osteosarcoma patients with at least 5-year follow-up is useful for predicting survival.
Materials and methods
Patients and tissue samples
This study was approved by the Research Ethics Committee of Shanghai Children’s Medical Center Affiliated to Shanghai Jiaotong University School of Medicine, Zhujiang Hospital, Xuhui central hospital, and Shanghai Sixth People’s Hospital, China. Written informed consent was obtained from all of the patients. All specimens were handled and made anonymous according to the ethical and legal standards.
For quantitative real-time reverse transcriptase-polymerase chain reaction (qRT-PCR) analysis, 92 pediatric patients with osteosarcomas (age range: 4–20 years, median 13 years) and corresponding noncancerous bone tissue samples from the same specimens were collected from Shanghai Children’s Medical Center Affiliated to Shanghai Jiaotong University School of Medicine, Zhujiang Hospital, Xuhui central hospital, and Shanghai Sixth People’s Hospital, China, from 1999 to 2008. After establishing the diagnosis, all patients were treated with a preoperative chemotherapy lasting 4 months, using either the combination of an anthracycline (doxorubicin) and high-dose methotrexate or the combination of etoposide, ifosfamide, and high-dose methotrexate. The postoperative treatment was determined by the histologic system established by Huvos et al. [11]. The following clinical parameters were analyzed: age, gender, site of tumor, tumor size, presence of pathological fracture and distant metastasis, histological subtype of osteosarcoma, type of surgery, and histological response to preoperative chemotherapy graded on the basis of the amount of remaining viable tumor cells in the resected specimen (a 10 % cutoff was used to discriminate between good and poor response as proposed in Salzer-Kuntschik Classification [12]). The clinicopathological information of the patients is shown in Table 1. Tumor biopsies were collected before neoadjuvant therapy and were fresh-frozen, stored at −80 °C, and histologically characterized by the pathologist.
All 92 pediatric patients with osteosarcoma received follow-up. The median follow-up was 82 months (range: 10–133 months). During the follow-up period, 36 patients (36/92, 39.1 %) died of disease. Metastases developed in 31 patients at a mean of 16.2 months (range 5–49 months) after the original diagnosis. The median overall survival and progression-free survival (PFS) of patients were 30 months (95 % confidence interval [CI], 26.4–42.7 months) and 22 months (95 % CI, 18.3–32.9 months), respectively.
miRNA qRT-PCR assay
The expression levels of miR-210 in osteosarcoma and corresponding noncancerous tissues were detected by qRT-PCR assay. Briefly, total RNA from tissue samples was extracted with TRizol reagent (Invitrogen, Breda, the Netherlands) according to the manufacturer’s instructions. cDNA was reverse transcribed from total RNA samples using specific miRNA primers from the TaqMan MicroRNA Assays and reagents from the TaqMan MicroRNA Reverse Transcription kit (Applied Biosystems, Foster City, CA, USA) according to manufacturer’s instructions. Products were amplified by PCR using TaqMan Universal PCR Master Mix kit (Applied Biosystems). Primers were purchased from Applied Biosystems: hsa-miR-210 (CUGUGCGUG UGACAGCGGCUGA) and RNU6B (CGCAAGGAUGACACGCAAAUUCGUGAAGCGUUCCAUAUUUUU). Values for miR-210 were normalized to the expression levels of RNU6B. Real-time PCR was performed in ABI 7500 (Applied Biosystems).
Statistical analysis
The software of SPSS version 13.0 for Windows (SPSS Inc, IL, USA) and SAS 9.1 (SAS Institute, Cary, NC) was used for statistical analysis. Continuous variables were expressed as \( \overline{X} \pm s \). The paired t test was used to evaluate the differences in miR-210 expression levels in osteosarcoma and corresponding noncancerous bone tissues. The chi-square test was used to show differences in categorical variables. Patient survival and their differences were determined by Kaplan–Meier method and log-rank test. Cox regression (Proportional hazard model) was adopted for multivariate analysis of prognostic factors. Differences were considered statistically significant when P was less than 0.05.
Results
Elevated expression of miR-210 in pediatric osteosarcoma tissues
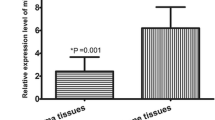
The expression levels of miR-210 in osteosarcoma and corresponding noncancerous bone biopsy samples were detected by qRT-PCR and normalized to RNU6B. Compared to noncancerous bone tissues, the expression levels of miR-210 in osteosarcoma tissues were distinctly elevated (Fig. 1). As the statistic results, the relative level of miR-210 expression normalized to RNU6B in osteosarcoma tissues (mean ± SD: 3.3 ± 0.8) was significantly higher than that in corresponding noncancerous bone tissues (mean ± SD: 2.0 ± 0.7; P < 0.001). The median of miR-210 expression levels in all 92 patients with osteosarcoma was 3.2. We divided the patients into two groups according to their expression levels of miR-210 using its median as a cutoff: high miR-210 expression group (n = 48, mean ± SD: 3.8 ± 0.5) and low miR-210 expression group (n = 44, mean ± SD: 2.8 ± 0.4).
miR-210 expressions in 166 pairs of osteosarcoma and corresponding noncancerous bone tissues were, respectively, detected by real-time quantitative RT-PCR assay. The expression levels of miR-210 in osteosarcoma tissues were significantly lower than those in corresponding noncancerous bone tissues (P < 0.0001)
Elevated expression of miR-210 associates with advanced clinicopathological features of pediatric osteosarcoma
Table 1 summarized the associations of miR-210 expression with various clinicopathological parameters of osteosarcoma tissues. In patients with large tumor size (>8 cm), miR-210 upregulation occurred significantly more often than those with small tumor size (≤8 cm, P = 0.02). Taking into consideration the relationship between miR-210 expression and the response to preoperative chemotherapy, we found that patients in high miR-210 expression group had poorer response to preoperative chemotherapy than those in low miR-210 expression group (P = 0.008). The elevated expression of miR-210 was also associated with the presence of metastasis (P = 0.01). Detailed results of other statistical tests of miR-210 expression and clinicopathological parameters are presented in Table 1.
Elevated expression of miR-210 confers poor prognosis in patients with osteosarcomas
Using Kaplan–Meier method and log-rank test, the overall survival (OS, Fig. 2a, P = 0.007) and progression-free survival (PFS, Fig. 2b, P = 0.01) of pediatric osteosarcoma patients with high miR-210 expression were both significantly shorter than those with low miR-210 expression. Besides, the survival benefits were also found in those with smaller tumor size (P = 0.008 and 0.01, respectively), without metastasis (P < 0.001 and 0.001, respectively), and better response to preoperative chemotherapy (both P = 0.01) for OS and PFS.
Overall survival (a) and progression-free survival (b) curves for two groups defined by low and high expression of miR-210 in patients with pediatric osteosarcoma. The patients with high miR-210 expression had a significantly worse 5-year overall (P = 0.007) and progression-free (P = 0.01) survival rate than those with low miR-210 expression
In the Cox proportional hazard model, it was confirmed that miR-210 expression in the biopsy samples (for OS: RR 3.3, 95 % CI, 1.0–8.1, P = 0.01; for PFS: RR 2.6, 95 % CI, 0.8–7.2, P = 0.02), tumor size (for OS: RR 3.5, 95 % CI, 1.0–8.4, P = 0.01; for PFS: RR 2.9, 95 % CI, 0.8–7.6, P = 0.02), metastasis status (for OS: RR 4.2, 95 % CI, 1.6–10.3, P = 0.006; for PFS: RR 3.9, 95 % CI, 1.3–9.6, P = 0.008), and response to preoperative chemotherapy (for OS: RR 2.5, 95 % CI, 0.9–7.3, P = 0.02; for PFS: RR 2.1, 95 % CI, 0.6–6.6, P = 0.03) was independent prognostic factors of unfavorable survival in pediatric osteosarcoma (Table 2).
Discussion
The importance of miRNAs in solid tumors and their potential utility as prognostic factors have become apparent. In the current study, we firstly found that miR-210 expression was increased in pediatric osteosarcoma tissues compared with noncancerous bone tissues; Then, the upregulation of miR-210 in osteosarcoma tissues was significantly correlated with aggressive clinicopathological features; Moreover, the results of Kaplan–Meier analyses shown that osteosarcoma tissues with the increased expression of miR-210 tend to have shorter overall and progression-free survival. Finally, the multivariate analysis clearly indicated that the increased expression of miR-210 in biopsy samples may be considered as a prognostic factor in pediatric osteosarcoma for decreased survival and a greater probability of disease progression regardless of oncological treatment. To our knowledge, this is the first study to investigate the clinical significance of miR-210 in pediatric osteosarcoma patients.
Hypoxia is a common feature of various pathological conditions including tissue ischemia, inflammation, and solid tumors [13]. miR-210 is the miRNA most frequently associated with tumor hypoxia, and recent studies have demonstrated that miR-210 is highly upregulated in hypoxic cells [14]. The stem-loop of miR-210 is located in an intron of a noncoding RNA, which is transcribed from AK123483 on chromosome 11p15.5 [15]. It plays important roles in cell survival and tumor initiation. By a microarray analysis, miR-210 has been identified as the predominant miR induced by hypoxia in pancreatic cancer cell lines as its induction was dependent on HIF-1α which regulates miR-210 by directly binding to a hypoxia responsive element on the proximal miR-210 promoter [16]. Recent studies have found that the expression of miR-210 is also upregulated in many other cancers including head and neck cancer, glioblastoma, breast cancer, non-small-cell lung cancer, malignant melanoma, prostate cancer, renal cell carcinoma, and osteosarcomas [17–20]. It functions either as a diagnostic factor or as a prognostic marker. From a diagnostic standpoint, Wang et al. [21] reported that the expression level of miR-210 was distinctly increased in the serum from patients with diffuse large B-cell lymphoma and in the plasma of pancreatic cancer patients. From a prognostic standpoint, Rothe et al. [22] found that miR-210 was significantly associated with poor clinical outcome in ER-positive, tamoxifen-treated breast cancer patients. Greither et al. [23] indicated that miR-210 might be a strong potential biomarker of prognosis in a gender-specific manner in soft-tissue sarcoma. Gee et al. [24] reported that high levels of miR-210 were associated with locoregional disease recurrence and short overall survival in patients with head and neck cancer.
According to the publication referenced above, there is no report concerning the involvement of miR-210 in pediatric osteosarcoma. The interesting aspect of our study is the association of miR-210 upregulation with the advanced tumor progression and unfavorable prognosis in pediatric osteosarcoma. However, the current study was limited by its retrospective nature which led to our results being considered exploratory. Then, the tumor sample size was relatively smaller. Further experimental validation using a large number of tumor samples should be performed.
In conclusion, these findings suggested that miR-210 upregulation showed a strong correlation with tumor aggressive progression of pediatric osteosarcoma and could help prognostic screening of patients with this malignancy. This study is hypothesis generating, and that further prospective analysis should be worth doing.
References
Broadhead ML, Clark JC, Myers DE, Dass CR, Choong PF. The molecular pathogenesis of osteosarcoma: a review. Sarcoma. 2011;2011:959248.
Ottaviani G, Jaffe N. The epidemiology of osteosarcoma. Cancer Treat Res. 2009;152:3–13.
PosthumaDeBoer J, Witlox MA, Kaspers GJ, van Royen BJ. Molecular alterations as target for therapy in metastatic osteosarcoma: a review of literature. Clin Exp Metastasis. 2011;28:493–503.
Geller DS, Gorlick R. Osteosarcoma: a review of diagnosis, management, and treatment strategies. Clin Adv Hematol Oncol. 2010;8:705–18.
Osman A. MicroRNAs in health and disease–basic science and clinical applications. Clin Lab. 2012;58:393–402.
Kong YW, Ferland-McCollough D, Jackson TJ, Bushell M. microRNAs in cancer management. Lancet Oncol. 2012;13:e249–58.
Subramanian S, Kartha RV. MicroRNA-mediated gene regulations in human sarcomas. Cell Mol Life Sci. 2012;69:3571–85.
Lulla RR, Costa FF, Bischof JM, Chou PM. de F Bonaldo M, Vanin EF, Soares MB. Identification of Differentially Expressed MicroRNAs in Osteosarcoma. Sarcoma. 2011;2011:732690.
Fasanaro P, Greco S, Lorenzi M, Pescatori M, Brioschi M, Kulshreshtha R, Banfi C, Stubbs A, Calin GA, Ivan M, Capogrossi MC, Martelli F. An integrated approach for experimental target identification of hypoxia-induced miR-210. J Biol Chem. 2009;284:35134–43.
Mizuno Y, Tokuzawa Y, Ninomiya Y, Yagi K, Yatsuka-Kanesaki Y, Suda T, Fukuda T, Katagiri T, Kondoh Y, Amemiya T, Tashiro H, Okazaki Y. miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett. 2009;583:2263–8.
Huvos AG. Osteosarcoma in adolescents and young adults: new developments and controversies. Commentary on pathology. Cancer Treat Res. 1993;62:375–7.
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, Shenkier T, Cella D, Davidson NE. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med. 2007;357:2666–76.
Chi JT, Wang Z, Nuyten DS, et al. Gene expression programs in response to hypoxia: cell type specificity and prognostic significance in human cancers. PLoS Med. 2006;3:e47.
Hong L, Han Y, Zhang H, Zhao Q, Qiao Y. miR-210: a therapeutic target in cancer. Expert Opin Ther Targets. 2013;17:21–8.
He J, Wu J, Xu N, Xie W, Li M, Li J, Jiang Y, Yang BB, Zhang Y. MiR-210 disturbs mitotic progression through regulating a group of mitosis-related genes. Nucleic Acids Res. 2012 In press.
Merlo A, de Quiros SB, Secades P, Zambrano I, Balbín M, Astudillo A, Scola B, Arístegui M, Suarez C, Chiara MD. Identification of a Signaling Axis HIF-1α/MicroRNA-210/ISCU independent of SDH mutation that defines a subgroup of head and neck paragangliomas. J Clin Endocrinol Metab. 2012;97:E2194–200.
Papaconstantinou IG, Manta A, Gazouli M, Lyberopoulou A, Lykoudis PM, Polymeneas G, Voros D. Expression of MicroRNAs in patients with pancreatic cancer and its prognostic significance. Pancreas. 2012 In press.
Hong L, Yang J, Han Y, Lu Q, Cao J, Syed L. High expression of miR-210 predicts poor survival in patients with breast cancer: a meta-analysis. Gene. 2012;507:135–8.
Võsa U, Vooder T, Kolde R, Vilo J, Metspalu A, Annilo T. Meta-analysis of microrna expression in lung cancer. Int J Cancer. 2012 In press.
Wotschofsky Z, Busch J, Jung M, Kempkensteffen C, Weikert S, Schaser KD, Melcher I, Kilic E, Miller K, Kristiansen G, Erbersdobler A, Jung K. Diagnostic and prognostic potential of differentially expressed miRNAs between metastatic and non-metastatic renal cell carcinoma at the time of nephrectomy. Clin Chim Acta. 2012 In press.
Wang J, Chen J, Chang P, LeBlanc A, Li D, Abbruzzesse JL, Frazier ML, Killary AM, Sen S. MicroRNAs in plasma of pancreatic ductal adenocarcinoma patients as novel blood-based biomarkers of disease. Cancer Prev Res. 2009;2:807–13.
Rothé F, Ignatiadis M, Chaboteaux C, Haibe-Kains B, Kheddoumi N, Majjaj S, Badran B, Fayyad-Kazan H, Desmedt C, Harris AL, Piccart M, Sotiriou C. Global microRNA expression profiling identifies MiR-210 associated with tumor proliferation, invasion and poor clinical outcome in breast cancer. PLoS ONE. 2011;6:e20980.
Greither T, Würl P, Grochola L, Bond G, Bache M, Kappler M, Lautenschläger C, Holzhausen HJ, Wach S, Eckert AW, Taubert H. Expression of microRNA 210 associates with poor survival and age of tumor onset of soft-tissue sarcoma patients. Int J Cancer. 2012;130:1230–5.
Gee HE, Camps C, Buffa FM, Patiar S, Winter SC, Betts G, Homer J, Corbridge R, Cox G, West CM, Ragoussis J, Harris AL. hsa-mir-210 is a marker of tumor hypoxia and a prognostic factor in head and neck cancer. Cancer. 2010;116:2148–58.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Haiqing Cai and Lijun Lin contribute equally to this work.
Rights and permissions
About this article
Cite this article
Cai, H., Lin, L., Cai, H. et al. Prognostic evaluation of microRNA-210 expression in pediatric osteosarcoma. Med Oncol 30, 499 (2013). https://doi.org/10.1007/s12032-013-0499-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0499-6