Abstract
Neuroendocrine tumors (NETs) of the gastrointestinal tract have a propensity for hepatic metastases. Surgical resection for hepatic metastases remains the gold standard for long-term survival, but many patients present with multifocal tumors, precluding surgery with increasing use of chemoembolization. However, there are few studies examining long-term survival factors. We reviewed our 15-year experience with chemoembolization in 123 patients with unresectable NET liver metastases, whose prognosis was evaluated upon baseline clinical factors. There were 64 males (53%) and 59 females (47%). Average age at presentation was 56 years (range: 14.3–85.5 years). Abdominal pain (44%) was the most common presenting symptom, followed by diarrhea (30%) and weight lost (22%). Patients underwent an average 7.3 cycles of chemoembolization (range 1–32 cycles). Responses: 62% of patients had PR; 24% had stable disease and 14% had tumor progression. Overall 3-, 5- and 10-year survivals were 59, 36 and 20% of patients with a mean follow-up of 3.2 years (range 2 weeks–18.3 years) and mean survival of 3.3 years. Univariate analysis showed that age greater than 60 years had worse outcome (P < 0.01), as did baseline serum albumin of ≤3.5 g/dL and prothrombin time >13 s. Location of the primary tumor (P = 0.68), gender (P = 0.4) and serum NET peptide levels did not influence survival. However, multivariate analysis showed that a low baseline serum albumin level was an independent factor for prognosis (P = 0.003). Chemoembolization for unresectable NETs metastatic to liver is useful for tumor size reduction, symptom palliation and can be associated with prolonged survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neuroendocrine tumors (NETs), commonly referred to as carcinoids, consist of a heterogeneous group of malignancies derived from enterochromaffin cells, which are part of the amine precursor uptake and decarboxylation (APUD) system. NETs can thus arise from a broad spectrum of anatomic sites [1, 2]. These tumors share similar histological characteristics and biochemical profiles, including the ability to produce a number of amines, polypeptides and prostaglandins [3, 4]. The incidence has been estimated to be as high as 8.4 cases per 100,000 people, although the actual incidence may be significantly higher if one includes subclinical cases [3, 5, 6]. A significant proportion of patients have systemic metastases at the time of presentation [6]. Overall, hepatic metastases with NETs are the commonest site of spread, ranging from 10 to 65% of cases [6, 7] Published reports of NETs frequently focused on the prognosis of patients who are found to have primary neuroendocrine tumors [8, 9]. A group of NET patients with unresectable liver metastases outlive patients with comparable tumor burdens from most other cancers.
Since the rate of progression differs widely between patients, the management modalities for metastatic NETs are numerous [10–13] Surgical management of resectable metastatic disease remains the best option for cure, with 5-year survivals estimated between 50 and 79% [8, 14–17] However, a number of modalities including systemic chemotherapy, somatostatin analogues, cryotherapy, radiofrequency ablation, percutaneous alcohol injection, hepatic transplantation and chemoembolization have been used to treat patients with unresectable metastatic disease [15, 18]. Chemoembolization has been reported to be an effective strategy for the treatment of metastatic NETs isolated to the liver [18–20] and has been accepted as either a bridge to surgical therapy or a primary treatment for unresectable liver tumors [1, 18, 20]. Comparison of the various modalities showed that chemoembolization treatments seem to offer both symptomatic improvements and survival advantage compared to medical therapy alone [8, 16]. We review here our experience with chemoembolization therapy for the treatment of metastatic NETs to the liver at a large tertiary referral center over a 15-year period, with a focus on prognostic factors.
Materials and methods
Patients
We retrospectively analyzed the data of 123 patients with unresectable metastatic NETs to the liver treated with chemoembolization at the University of Pittsburgh Medical Center between 1990 and 2005. All patient data were gathered from electronic chart review and a previously computerized database. This database comprised a list of patients seen at a large tertiary center concentrating on the management of patients with unresectable liver tumors using multiple chemoembolization treatments by one of us (BC). In all patients, diagnosis was confirmed by biopsy, using routine histology and immunochemical examination. Baseline and monthly follow-up laboratory blood tests included routine complete blood count, prothrombin time, creatinine, routine liver function test. A NET peptide panel was performed at baseline, and the peptides that were elevated in an individual patient were repeated in follow-up. Octreotide scans were used to assess disease spread at baseline only, or if progression was suspected. Follow-up records for survival were obtained from clinic notes or phone calls.
All patients reviewed underwent chemoembolization after surgical resection was deemed impossible. Informed consent was obtained from all patients prior to their treatments. Patients with portal vein occlusion, encephalopathy, massive ascites, uncontrolled coagulopathy or cardiac failure were excluded from treatments. Patients were generally admitted the night before their treatment and hydrated, prior to undergoing therapy. Post-procedural routine consisted of intravenous hydration and anti-emetics, followed by resumption of oral intake, assessment of liver functions and coagulation profiles, followed by discharge the following day. Patients generally underwent pre-procedural CT or MRI scan of the abdomen to identify the largest lesions suitable for embolization. Following cannulation of the feeding hepatic vessels, the dominant hepatic masses were embolized using a combination of embolizing agent plus chemotherapy, which was delivered sub-selectively to the feeding artery for single lesions, or to a lobar artery for multiple lesions. Even bilobar tumors were treated to a single lobe at any one therapy session to limit hepatotoxicity. Choice of embolic agents was chosen at the discretion of the radiologist (Gelfoam® or Biospheres®). Choice of chemotherapy agents varied depending on previous treatments, with most patients receiving doxorubicin at 40 mg/m2 as the starting dose. Many patients received streptozotocin at 2 gm/m2 if they were intolerant of doxorubicin or had previous doxorubicin exposure. All patients received intravenous hydration, anti-emetics and analgesics before and after chemoembolization. Patients with carcinoid symptoms were also given concomitant systemic Sandostatin. No carcinoid crises were experienced. Additional systemic chemotherapy was not given due to tolerance considerations. LAR treatments were repeated every 2 months till radiological stabilization, then every 3–4 months. Radiographic follow-up of hepatic tumor burden was performed using CT or MRI scans prior to the next treatment. Responses were assessed using RECIST criteria.
Clinical variables reviewed included gender, age, location of primary, presenting symptoms, number of chemoembolization treatments, size of metastases and serum hormones. Age at diagnosis represents the time liver metastases were discovered. Location of primary was defined as foregut (respiratory tract, stomach, duodenum, biliary tract), midgut (small bowel, appendix, right colon), hindgut (left colon, rectum) or unknown/liver (primary liver neuroendocrine tumors and metastatic disease with unknown primaries) [21]. Follow-up and survival were calculated from the time liver metastases were first diagnosed.
Statistical analysis
Data were reported as mean ± standard error. Comparison for survival between different variables, plotted according to the method of Kaplan–Meier, was determined using the log-rank test. The radiological response based on scan measurements of tumor sizes following treatment was examined using the paired t-test. Correlation between the number of chemoembolization cycles and survival was also determined using Spearman’s method with significance set at P = 0.01.
Results
Patient baseline characteristics are shown in Table 1. Out of 123 patients, 66 were male (53%) and 59 were female (43%). The average age of patients at the time of diagnosis for liver metastases was 56 years (range: 14–85 years). Patients were followed for a mean of 3 years (range: 0.03–18 years). Forty-two percent (n = 52) of the patients were older than 60 at the time of diagnosis for liver metastases. A small subset of individuals was under 40 years of age (n = 17). The most common site of the primary tumor was foregut (n = 61). Fifteen of the 61 foregut primary tumors were in the pancreas at the time of presentation, and these were resected prior to treatment of the liver metastases. A substantial number of patients had unknown primaries (n = 38), precluding any intervention for the primary site.
Most patients presented with symptoms with only 9 patients being discovered incidentally. The most common clinical presentation was abdominal pain (40%), followed by diarrhea (25%), weight loss (20%), nausea and vomiting (11%), flushing (11%) and fatigue (9%). Thirty-five patients (32%) had symptoms of carcinoid syndrome, consisting of diarrhea, flushing or palpitations. A significant proportion of the tumors elaborated hormones without associated symptoms, with the majority producing pancreatic polypeptide (n = 54), followed by serotonin (n = 36), gastrin (n = 25), glucagon (n = 6), insulin (n = 4), VIP (n = 3), somatostatin (n = 1) and ACTH (n = 1).
An average, 7 cycles of chemoembolization were administered to this cohort of 123 patients. There were no technical delivery failures and no > grade 2 treatment toxicities, despite several patients having >70% of the liver involved by tumor mass. Responses (Table 2) by CT/MRI were the following: Partial responses (PR) were found in 76 patients (62%), and an additional 30 patients (24%) had minor responses or tumor stabilization, for a total disease control (PR + CR + stable) of 106 patients (86%). Seventeen patients (14%) showed progressive disease while undergoing chemoembolization. Responses were seen across all the NET subtypes, including insulinomas and glucagonomas.
Mean survival for the entire cohort was 5.47 years. Overall 3-, 5- and 10-year survivals were 59, 36, and 20%. Univariate analysis (Table 3) revealed that age >60 years was a negative predictor of survival (P = 0.03). Gender, site of primary tumor and hormone production did not predict survival. Patients with unknown primaries, who thus must have had their primary tumors left untreated, had comparable survival to patients who underwent surgical management of their primaries.
Pre-treatment laboratory evaluation revealed that albumin <3.5 g/dL and PT >13 s were statistically significant in predicting unfavorable outcome. Hormone production including pancreatic polypeptide, serotonin, chromogranin and gastrin did not affect survival. Multivariate analysis showed that albumin <3.5 g/dL was an independent unfavorable prognostic factor for survival, with an Exp(B) of 2.708 (P = 0.003, 95% CI: 1.412, 5.192).
Discussion
Many patients remain asymptomatic until their tumors are metastatic. About 10% of patients already have hepatic metastases at presentation although some report that to be as high as 50–65% of patients [6, 7]. Unlike patients with isolated primary NETs, a large proportion of patients (92%) seen in this series were symptomatic from their tumor burdens. Carcinoid syndrome, manifested by either diarrhea (25%) or by flushing (11%), was not prevalent even when patients had developed large tumor burdens. The majority of patients presented with abdominal pain and discomfort (40%) due to their liver tumor size. Several patients had symptoms related to liver dysfunction. Serum levels of hormone production also did not correlate with symptoms, as a large number of patients produced non-functional hormones with pancreatic polypeptide being the commonest, followed by serotonin.
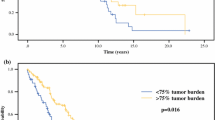
The patients with NET hepatic metastases have several treatment options. On the one hand, the tumors are clearly malignant culminating in symptoms both from tumor bulk and from hormone production, which is terminal in the majority of cancers. On the other hand, the indolent growth rate of many of these tumors offers a unique opportunity to intervene [7]. When tumor bulk precludes surgical extirpation, various modalities are utilized for palliation. Since the hepatic artery alone supplies these hepatic tumors, this represents an opportunity for selective drug delivery [7, 19]. Overall patient survival in this cohort was comparable to previous observations [16, 22]. Long-term survival was 35.5% at 5 years and 19.5% at 10 years in our series. The 3.3-year median survival is a reminder that even with chemotherapy responses; this can be a lethal disease. Chamberlain et al. showed that patients with over 75% liver involvement by tumor had only 33% 5-year survival [16].
Analysis of multiple clinical variables showed that age, albumin level and prothrombin time influenced survival. Location of the primary did not influence overall survival in this series of patients. A recent small study, using combination chemotherapy, might yield longer survival [23]. Unlike pancreatic adenocarcinomas, location was not a significant predictor in this series of patients. Neither gender nor hormone affected survival (Table 3). Laboratory findings showed that albumin and PT are significant predictors of outcome. Similar to the results of chemoembolization for inoperable hepatocellular carcinoma [24], markers of synthetic function of the liver are significant predictors of outcome (Table 3).
Because of the limited number of patients available, prospective randomized clinical trials comparing chemoembolization therapy versus other modalities are difficult in single-institution studies of NET. However, the use of chemoembolization for symptomatic control is increasingly accepted [11, 18–20, 22]. We found that baseline low serum albumin levels, high prothrombin time and old age are variables identifying patients at risk of poorer survival.
Abbreviations
- NET:
-
Neuroendocrine tumor
- CT:
-
Computerized tomography
- PR:
-
Partial response
References
O’Toole D, Maire F, Ruszniewski P. Ablative therapies for liver metastases of digestive endocrine tumours. Endocr Relat Cancer. 2003;10:463–8.
Creutzfeldt W. Carcinoid tumors: development of our knowledge. World J Surg. 1996;20:126–31.
Lips CJ, Lentjes EGWM, Hoppener JWM. The spectrum of carcinoid tumours and carcinoid syndromes. Ann Clin Biochem. 2003;40:612–27.
Moertel CG. Karnofsky memorial lecture: an odyssey in the land of small tumors. J Clin Oncol. 1987;5(10):1503–22.
Berge T, Linell F. Carcinoid tumors, frequency in a defined population during a 12-year period. Acta Pathol Microbiol Scand A. 1976;84:322–30.
Bax N, Woods H, Batchelor A, Jennings M. Clinical manifestations of carcinoid disease. World J Surg. 1996;20:142–6.
Proye C. Natural history of liver metastasis of gastroenteropancreatic neuroendocrine tumors: place for chemoembolization. World J Surg. 2001;25:685–8.
Shebani KO, Souba WW, Finkelstein DM, Stark PC, Elgadi KM, Tanabe KK, et al. Prognosis and survival in patients with gastrointestinal tract carcinoid tumors. Ann Surg. 1999;229(6):815–23.
Pape U-F, Bohmig M, Berndt U, Tiling N, Wiedenmann B, Plockinger U. Survival and clinical outcome of patients with neuroendocrine tumors of the gastroenteropancreatic tract in a German referral center. Ann N Y Acad Sci. 2004;1014:222–33.
Woodside KJ, Townsend CM, Evers BM. Current management of gastrointestinal carcinoid tumors. J Gastrointest Surg. 2004;8(6):742–56.
Yao KA, Talamonti MS, Nemcek A, Angelos P, Chrisman H, Skarda J, et al. Indications and results of liver resection and hepatic chemoembolization for metastatic gastrointestinal neuroendocrine tumors. Surgery. 2001;130(4):677–85.
Siperstein AE, Berber E. Cryoablation, percutaneous alcohol injection, and radiofrequency ablation for treatment of neuroendocrine liver metastases. World J Surg. 2001;25(6):693–6.
Florman S, Toure B, Kim L, Gondolesi G, Roayaie S, Krieger N, et al. Liver transplantation for neuroendocrine tumors. J Gastrointest Surg. 2004;8(2):208–12.
Azimuddin K, Chamberlain RS. The surgical management of pancreatic neuroendocrine tumors. Surg Clin North Am. 2001;81(3):511–25.
Sutcliffe R, Maguire D, Ramage J, Rela M, Heaton N. Management of neuroendocrine liver metastases. Am J Surg. 2004;187:39–46.
Chamberlain R, Canes D, Brown K, Saltz L, Jarnagin W, Fong Y, et al. Hepatic neuroendocrine metastases: does intervention alter outcomes? J Am Coll Surg. 2000;190(4):432–45.
Nave H, Mossinger E, Feist H, Lang H, Raab H-R. Surgery as primary treatment in patients with liver metastases from carcinoid tumors: a retrospective, unicentric study over 13 years. Surgery. 2001;129:170–5.
Roche A, Girish BV, de Baere T, Baudin E, Boige V, Elias D, et al. Trans-catheter arterial chemoembolization as first-line treatment for hepatic metastases from endocrine tumors. Eur Radiol. 2003;13:136–40.
Allison D, Modlin I, Jenkins W. Treatment of carcinoid liver metastases by hepatic-artery embolisation. Lancet. 1977;2(8052–8053):1323–5.
Drougas JG, Anthony LB, Blair TK, Lopez RR, Wright JK, Chapman WC, et al. Hepatic artery chemoembolization for management of patients with advanced metastatic carcinoid tumors. Am J Surg. 1998;175:408–12.
Carr BI, Kondragunta V, Buch S, Branch R. Therapeutic equivalence in survival for hepatic arterial chemoembolization and 90Yttrium microspheres (Y90) treatments in unresectable HCC. Cancer. 2010;116:1305–14.
Gupta S, Yao JC, Ahrar K, Wallace MJ, Morello FA, Madoff DC, et al. Hepatic artery embolization and chemoembolization for treatment of patients with metastatic carcinoid tumors: the M.D. Anderson experience. Cancer J. 2003;9(4):261–7.
Vogl TJ, Gruber T, Naguib NNN, Hammersting R, Nour-Eldin NA. Liver metastases of neuroendocrine tumors: treatment with hepatic transarterial chemotherapy using two therapeutic protocols. Am J Roentgen. 2009;193:941–7.
Carr BI, Buch SC, Kondragunta V, Pancoska P, Branch RA. Tumor and liver determinants of prognosis in unresectable HCC: a case cohort study. J Gastroenterol Hepatol. 2008;23:1259–66.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dong, X.D., Carr, B.I. Hepatic artery chemoembolization for the treatment of liver metastases from neuroendocrine tumors: a long-term follow-up in 123 patients. Med Oncol 28 (Suppl 1), 286–290 (2011). https://doi.org/10.1007/s12032-010-9750-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-010-9750-6