Abstract
Background
The impact of ventriculostomy-associated infections (VAI) on intracerebral hemorrhage (ICH) outcomes has not been clearly established, although prior studies have attempted to address the incidence and predictors of VAI. We aimed to explore VAI characteristics and its effect on ICH outcomes at a population level.
Methods
ICH patients requiring ventriculostomy with and without VAI were identified from 2002 to 2011 Nationwide Inpatient Sample using ICD-9 codes. A retrospective cohort study was performed. Demographics, comorbidities, hospital characteristics, inpatient outcomes, and resource utilization measures were compared between the two groups. Pearson’s Chi-square and Wilcoxon–Mann–Whitney tests were used for categorical and continuous variables, respectively. Logistic regression was used to analyze the predictors of VAI.
Results
We included 34,238 patients in the analysis, of whom 1934 (5.6 %) had VAI. The rate of ventriculostomy utilization in ICH increased from 5.7 % in 2002–2003 to 7.0 % in 2010–2011 (trend p < 0.001) and the rate of VAI also showed a gradual upward trend from 6.1 to 7.0 % across the same interval (trend p < 0.001). The VAI group had significantly higher inpatient mortality (41.2 vs. 36.5 %, p < 0.001) and it remained higher after controlling for baseline demographics, hospital characteristics, comorbidity, and systemic infections (adjusted OR 1.38, 95 % CI 1.22–1.46, p < 0.001). The VAI group had longer length of hospital stay and higher inflation adjusted cost of care. Predictors of VAI included higher age, males, higher Charlson’s comorbidity scores, longer length of stay, and presence of systemic infections mainly pneumonia and sepsis.
Conclusion
VAI resulted in higher inpatient mortality, more unfavorable discharge disposition, and higher resource utilization measures in ICH patients. Steps to mitigate VAI may help improve ICH outcomes and decrease hospital costs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
External ventriculostomy drains (EVD) offer an alternate conduit for cerebrospinal fluid drainage, and serve as a life saving approach in intracerebral hemorrhage (ICH) patients with hydrocephalus or intraventricular hemorrhage (IVH) or both [1]. Additionally, they have also been used to administer recombinant tissue-type plasminogen activator (rt-PA) for clot lysis in patients with IVH [2]. EVDs, however, have significant complications, the most notable being hemorrhage [3] and infection (meningitis or ventriculitis) [4]. The incidence of ventriculostomy-associated infections (VAI) has been reported to vary from 1 to 28.2 %, with a mean reported rate of 7.9 % [5, 6]. Factors predisposing to VAI include craniotomy, systemic infection, depressed cranial fracture, and IVH to name a few [5]. Greater emphasis on ventriculostomy insertion techniques and use of impregnated catheters may decrease VAI, although trends over time are not known. Whether the duration of ventriculostomy correlates with VAI remains an area of debate.
Limitations of prior studies describing VAI incidence and predictors include retrospective design, small to modest sample size and single center data. A recent sub study of the Clot Lysis: Evaluating Accelerated Resolution of Intraventricular Hemorrhage Phase III (CLEAR-III) addressed these limitations and showed a VAI rate of 4.4 % [6]. With therapies such as intraventricular fibrinolysis on the rise over the last decade [7], it is important to delineate VAI characteristics at a population level, particularly in the absence of such data. We hence aimed to explore the incidence, trends over time, predictors, and outcomes of VAI using a large national health database.
Methods
Data Source
Discharge data were obtained from the Nationwide Inpatient Sample (NIS) of the Healthcare Cost and Utilization Project (HCUP) from 2002 to 2011 [8]. NIS represents a 20 % stratified random sample of all inpatient admissions to non-federated US hospitals. The database provides information on patient demographics, hospital characteristics, primary and secondary diagnoses, inpatient procedures, comorbidities, and case-severity measures. All diagnoses and procedures are recorded using International Classification of Diseases version 9 Clinical Modification (ICD-9-CM) codes.
Case Selection
Cases with a primary diagnosis of non-traumatic ICH were identified using the ICD-9-CM code 431 [9]. Subsequently, only ICH patients requiring ventriculostomy were selected, using procedure code 02.2 (prior to October, 1, 2011) and 02.21 (from October, 1, 2011) [10]. Cases with age <18 years, traumatic brain injury, brain malignancy, and cerebral vascular malformations were excluded to restrict our population to those with primary ICH. Additionally, we also excluded patients with early death (<48 h) and those with initiation of palliative care (code V66.7). As the unit of the NIS database is discharge after hospitalization, rather than an individual patient, cases transferred out to another hospital were excluded to prevent double counting of the same patient. VAI was defined as any patient with ventriculostomy who developed meningitis (ICD-9-CM 320.x, 321.x, 322.x). Figure 1 shows the case selection strategy used in the study.
Comorbidity and Severity Adjustment
A cumulative score was calculated for each case using the Charlson comorbidity index [11, 12]. The index is a weighted score of 17 different comorbidities using ICD-9-CM codes, and has been validated for outcome adjustment for administrative datasets on ischemic stroke and ICH [13, 14]. Case severity was determined using the all patient refined diagnosis-related groups (APR-DRGs) to assess risk of mortality using an algorithm developed by 3M Health Information Systems. This 4 point ordinal scale (minor, moderate, major, and extreme risk of mortality) is derived from age, primary and secondary diagnoses, and procedures [15]. The APR-DRG methodology has been validated to predict mortality in administrative datasets and has been used as a severity indicator in prior studies, including those relating to ICH [7, 16, 17].
Outcomes Measures
The primary outcomes of interest were inpatient mortality. Resource utilization measures used in the study were length of hospital stay and overall cost of care. Cost of care was obtained using HCUP cost-to-charge ratios and total hospital charges, and was adjusted for inflation to obtain US 2013 dollar values using yearly inflation rates published by US Department of Labor-Bureau of Labor statistics [18].
Statistical Analysis
Comparisons were made using χ 2 and Wilcoxon rank sum tests for categorical and continuous variables, respectively. Multivariate logistic regression was used to adjust for known potential confounding variables in assessing the effect of VAI on outcomes. The following covariates were included in all regression models: age, gender, race/ethnicity, hospital characteristics (location, teaching status, geographic region, bed size, and ICH case volume quartile), modified Charlson comorbidity index, 3 M APR-DRG risk of mortality subclass, coronary artery disease, diabetes mellitus, hypertension, atrial fibrillation, dyslipidemia, anemia, valvular disease, anticoagulation-associated hemorrhage, thrombocytopenia, blood components transfusion, performance of cerebral angiography, craniectomy and craniotomy, prolonged mechanical ventilation, and systemic infections. We studied 10-year temporal trends of utilization of ventriculostomy in ICH and among the cases included in the analysis. Significance of trend was tested by χ 2 test for linear association. As recommended by HCUP, population estimates were obtained by complex sample analyses that consider weights, clustering, and stratification used for NIS sampling [19]. All analyses were performed using IBM SPSS version 20 (IBM Corporation, NY, USA) with statistical significance set at p < 0.05. No adjustment was made for multiple comparisons due to the exploratory nature of the analysis.
Results
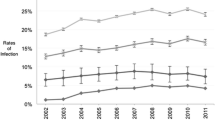
A total of 619,166 ICH admissions were recorded from 2002 to 2011 of whom 43,569 (7.1 %) had EVDs placed (Fig. 1). After applying the eligibility criteria, 34,238 patients were included for analysis. The overall incidence of VAI was 5.6 % (1934 patients). The rate of ventriculostomy utilization in ICH increased from 5.7 % in 2002–2003 to 7.0 % in 2010–2011 (trend p < 0.001) and the rate of VAI also showed an gradual upward trend from 6.1 to 7.0 % across the same interval (trend p < 0.001) as shown in Fig. 2.
Patients with VAI were younger (69.2 vs. 60.7 %, p < 0.001), and had a higher proportion of males (63.4 vs. 55.5 %, p < 0.001). The incidence of VAI was higher in urban hospitals, teaching hospitals, and hospitals with high ICH case volumes. Majority of VAI patients had private insurance (35.5 vs. 29.7 %, p < 0.001). The VAI cohort had lower modified Charlson’s comorbidity scores (p = 0.005), but the overall case severity as assessed by 3M APR-DRGs risk of mortality was higher in the VAI group (extreme likelihood of dying: 56.6 vs. 41.5 %, p < 0.001). Among inpatient procedures, craniectomy was more common in patients with VAI (2.4 vs. 1.7 %, p = 0.038), as were tracheostomy (p = 0.025) and gastrostomy (p < 0.001). Rates of VAI did not differ with intraventricular rt-PA infusion (Tables 1, 2, and 3).
In the multivariable logistic regression model, VAI was associated with higher in-hospital mortality (OR 1.38, 95 % CI 1.22–1.46, p < 0.001). Rates of permanent CSF shunting were higher with VAI (OR 1.78, 95 % CI 0.49–2.11, p < 0.001). In terms of resource utilization measures, VAI patients had longer length of stay (31 vs. 16 days with non-VAI, p < 0.001) and significantly higher total cost of care ($91170.4 vs. $47927.8 with non-VAI, p < 0.001). In the multivariable logistic regression model assessing predictors of VAI, higher age, males, higher Charlson’s comorbidity scores, longer length of stay and presence of systemic infections mainly pneumonia and sepsis. Procedures performed during hospitalization such as craniotomy or craniectomy were not associated with VAI (Table 4). We also performed linear regression analyses in different subgroups, which included high volume ICH centers only, patients on mechanical ventilation ≥96 h, patients transferred in from another hospital, and patients with the highest tertile of Charlson’s comorbidity scores (Supplemental Table 1). The rationale behind choosing these subgroups was to increase the probability of including patients with similar risk factors and mortality risk, in the absence of availability of ICH severity factors such as hematoma volume, location and presence of intraventricular hemorrhage. Both cost of care and hospital length of stay were significantly higher in the VAI group across all subgroups.
Discussion
Our analysis, the largest to date performed at a national level, showed that VAI was an independent predictor of in-hospital mortality, composite unfavorable discharge outcomes, and higher resource utilization measures. Interestingly, the rates of VAI have only gone up marginally over the past decade.
Mortality has not been the focus of prior VAI studies. We observed a mortality rate of 41.2 % with VAIs (vs. 36.5 % in the no VAI group), and nearly 50 % of VAI patients ended up with poor discharge outcomes compared to 35 % without VAI. Possible explanations for these findings include: worse neurologic outcome due to VAI itself, larger IVH requiring longer EVD duration, and unmeasured complications such as increased intracranial pressure, ICH expansion and ventriculostomy-associated hemorrhage. IVH is a significant risk factor for VAI and is also an independent predictor of 30-day ICH mortality and 90-day functional outcomes [1, 20]. Another plausible reason could be higher rates of new or expanded ICH/IVH following ventriculostomy. For instance, overall hemorrhage rates associated with EVDs are said to vary from 1 to 48 %, with a symptomatic ICH rate of 0 to 14.6 % [12]. The CLEAR-III trial reported a total EVD-related hemorrhage rate of 17.2 % with 2.4 % symptomatic hemorrhages [12]. Lack of specific ICD-9 codes for IVH, and absence of data on EVD duration, ICH and IVH volume, and neurologic assessment scales precluded further exploration of these variables in the analysis. The higher utilization of tracheostomy and gastrostomy tubes in patients with VAI compared to those without, suggests these patients had worse neurologic function. Systemic infections and sepsis were also more common in VAI patients, which may be attributable to post-stroke (or post-ICH) immunodepression marked by lymphopenia and upregulation of anti-inflammatory cytokines [21]. The consequence of this cascade is a higher rate of systemic infections that subsequently worsen clinical outcomes [22]. Conversely, systemic infections may act as potential sources for hematogenic spread to the central nervous system resulting in meningitis, which may have been diagnosed as VAI. Lack of availability of timeline of different infections prevented us from further studying the number of VAIs preceded by systemic infections.
This is also the first study to analyze resource utilization measures and hospital characteristics in VAI following ICH. We noted that patients with VAI had almost twice longer length of stay than non-VAI patients. This may reflect more severe ICH/IVH in the VAI cohort, or the need for longer duration of EVD increasing infection risk. Prior studies have shown that EVDs longer than 5 days have higher VAI rates compared to those with a shorter duration [23, 24]. Further, once diagnosed, completion of antibiotic therapies may have warranted continued hospitalization prolonging the length of stay. Presumably, higher rates of CSF shunt procedures and longer length of stay (particularly in the intensive care unit) drove the overall higher cost of care in the VAI cohort. As for hospital characteristics, our analysis suggests that high ICH case volume centers appear to have higher VAI rates. The observed incidence of VAI in the lowest volume centers (1st quartile) was 4.8 %, which gradually increased to 6.1 % in the highest ICH volume centers (4th quartile). We compared patient comorbidities between those admitted to high and low ICH volume centers. ICH patients admitted to high volume centers had higher Charlson comorbidity scores and higher APR-DRG risk of mortality. Higher case volume centers would have more expertise in placement and subsequent management of EVDs, not to mention standardized protocols. Hence, one may surmise that these patients had overall higher ICH severity (larger hematoma, more intraventricular hemorrhage) which may have necessitated longer duration of EVD and hence the higher VAI rate. However, data on EVD duration was not available in the NIS database.
The results of this study should be interpreted with caution due to inherent limitations of administrative databases, the retrospective and exploratory nature of the analyses, lack of well-validated ICH severity measures, and follow-up data. For severity measures, the NIS database did not have information on Glasgow coma scale or ICH/IVH volume and location. We used a previously validated DRG-based risk of mortality algorithm to partially overcome this limitation, although classification was likely imperfect. ICD-9-CM code 431 is validated to have high positive predictive value for diagnosing primary ICH from administrative datasets [9, 25], but the accuracy of the code for meningitis has not been studied. However, the code for meningitis has been used in prior studies utilizing the NIS database [26, 27]. While random ICD-9 coding errors are possible, they would bias the results toward the null, and are hence unlikely to account for the measured differences in mortality rates found in this study. The NIS database also did not have data on the duration of EVD, use of antibiotic impregnated EVDs, day of diagnosis of VAI, or use of prophylactic antibiotic therapy. Despite these limitations, large national administrative datasets such as the NIS provide a readily available opportunity to study less frequent complications like VAI at a population level.
Conclusion
VAI is associated with higher mortality, less favorable discharge outcomes, and more resource utilization measures in ICH patients. Annual incidence of VAI has, however, increased only slightly over the past decade and in parallel with apparent increased ventriculostomy utilization. Baseline patient demographics, comorbidities, hospital characteristics appear to predict VAI following ICH. Given the poor outcomes, better measures to mitigate VAIs are warranted.
References
Engelhard HH, Andrews CO, Slavin KV, Charbel FT. Current management of intraventricular hemorrhage. Surg Neurol. 2003;60:15–21 discussion 2.
Staykov D, Bardutzky J, Huttner HB, Schwab S. Intraventricular fibrinolysis for intracerebral hemorrhage with severe ventricular involvement. Neurocrit Care. 2011;15:194–209.
Gardner PA, Engh J, Atteberry D, Moossy JJ. Hemorrhage rates after external ventricular drain placement. J Neurosurg. 2009;110:1021–5.
Bota DP, Lefranc F, Vilallobos HR, Brimioulle S, Vincent JL. Ventriculostomy-related infections in critically ill patients: a 6-year experience. J Neurosurg. 2005;103:468–72.
Lozier AP, Sciacca RR, Romagnoli MF, Connolly ES Jr. Ventriculostomy-related infections: a critical review of the literature. Neurosurgery. 2002;51:170–81 discussion 81–82.
Dey M, Stadnik A, Riad F, et al. Bleeding and infection with external ventricular drainage: a systematic review in comparison with adjudicated adverse events in the ongoing Clot Lysis Evaluating Accelerated Resolution of Intraventricular Hemorrhage Phase III (CLEAR-III IHV) Trial. Neurosurgery. 2015;76:291–301.
Moradiya Y, Murthy SB, Newman-Toker DE, Hanley DF, Ziai WC. Intraventricular thrombolysis in intracerebral hemorrhage requiring ventriculostomy: a decade-long real-world experience. Stroke. 2014;45:2629–35.
Overview of the nationwide inpatient sample (nis). HCUP Databases. Healthcare Cost and Utilization Project (HCUP); 2012.
Kokotailo RA, Hill MD. Coding of stroke and stroke risk factors using international classification of diseases, revisions 9 and 10. Stroke. 2005;36:1776–81.
Sekula RF, Cohen DB, Patek PM, Jannetta PJ, Oh MY. Epidemiology of ventriculostomy in the United States from 1997 to 2001. Br J Neurosurg. 2008;22:213–8.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45:613–9.
Bar B, Hemphill JC 3rd. Charlson comorbidity index adjustment in intracerebral hemorrhage. Stroke. 2011;42:2944–6.
Jimenez Caballero PE, Lopez Espuela F, PortillaCuenca JC, Ramirez Moreno JM, Pedrera Zamorano JD, Casado Naranjo I. Charlson comorbidity index in ischemic stroke and intracerebral hemorrhage as predictor of mortality and functional outcome after 6 months. J Stroke Cerebrovasc Dis. 2013;22:e214–8.
Edwards N, Honemann D, Burley D, Navarro M. Refinement of the medicare diagnosis-related groups to incorporate a measure of severity. Health Care Financ Rev. 1994;16:45–64.
Baram D, Daroowalla F, Garcia R, et al. Use of the All Patient Refined-Diagnosis Related Group (APR-DRG) Risk of Mortality Score as a Severity Adjustor in the Medical ICU. Clin Med Circ Respirat Pulm Med. 2008;2:19–25.
Siddiq F, Adil MM, Norby KE, Rahman HA, Qureshi AI. Rates and outcomes of neurosurgical treatment for postthrombolytic intracerebral hemorrhage in patients with acute ischemic stroke. World Neurosurg. 2014;82:678–83.
CPI inflation calculator. Databases, Tables & Calculators by Subject, 2013; 2014.
Houchens R, Elixhauser A. Final report on calculating nationwide inpatient sample (nis) variances, 2001. HCUP Method Series Report # 2003-02. 2005;2012.
Nieuwkamp DJ, de Gans K, Rinkel GJ, Algra A. Treatment and outcome of severe intraventricular extension in patients with subarachnoid or intracerebral hemorrhage: a systematic review of the literature. J Neurol. 2000;247:117–21.
Offner H, Vandenbark AA, Hurn PD. Effect of experimental stroke on peripheral immunity: CNS ischemia induces profound immunosuppression. Neuroscience. 2009;158:1098–111.
Kamel H, Iadecola C. Brain-immune interactions and ischemic stroke: clinical implications. Arch Neurol. 2012;69:576–81.
Mayhall CG, Archer NH, Lamb VA, et al. Ventriculostomy-related infections. A prospective epidemiologic study. New Engl J Med. 1984;310:553–9.
Narayan RK, Kishore PR, Becker DP, et al. Intracranial pressure: to monitor or not to monitor? A review of our experience with severe head injury. J Neurosurg. 1982;56:650–9.
Williams GR, Jiang JG, Matchar DB, Samsa GP. Incidence and occurrence of total (first-ever and recurrent) stroke. Stroke. 1999;30:2523–8.
Khetsuriani N, Quiroz ES, Holman RC, Anderson LJ. Viral meningitis-associated hospitalizations in the United States, 1988-1999. Neuroepidemiology. 2003;22:345–52.
Sonig A, Thakur JD, Chittiboina P, Khan IS, Nanda A. Is posttraumatic cerebrospinal fluid fistula a predictor of posttraumatic meningitis? A US Nationwide Inpatient Sample database study. Neurosurg Focus. 2012;32:E4.
Acknowledgments
Dr. Hanley was awarded significant research support through Grant Numbers 5U01NS062851 for Clot Lysis Evaluation of Accelerated Resolution of Intraventricular Hemorrhage III, and 1U01NS08082 for Minimally Invasive Surgery Plus r-tPA for Intracerebral Hemorrhage Evacuation (MISTIE) III.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Drs. Murthy, Moradiya and Ziai report no disclosures.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Murthy, S.B., Moradiya, Y., Shah, J. et al. Incidence, Predictors, and Outcomes of Ventriculostomy-Associated Infections in Spontaneous Intracerebral Hemorrhage. Neurocrit Care 24, 389–396 (2016). https://doi.org/10.1007/s12028-015-0199-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-015-0199-5