Abstract
Background
Patients developing stress-induced cardiomyopathy (SIC) after subarachnoid hemorrhage (SAH) have increased risk of vasospasm, delayed cerebral ischemia and death. We evaluated whether high-sensitive troponin T (hsTnT) and N-terminal pro B-type natriuretic peptide (NTproBNP) are useful biomarkers for early detection of SIC after SAH.
Methods
Medical records of all patients admitted to our NICU with suspected or verified SAH from January 2010 to August 2014 were reviewed. Patients in whom echocardiography was performed and blood samples for measurements of hsTnT and/or NTproBNP were obtained, within 72 and 48 h, respectively, after onset of symptoms, were included. SIC was defined as reversible left ventricular segmental hypokinesia diagnosed by echocardiography.
Results
A total of 502 SAH patients were admitted during the study period, 112 patients fulfilled inclusion criteria and 25 patients fulfilled SIC criteria. Peak levels of hsTnT and NTproBNP were higher in patients with SIC (p < 0.001). hsTnT had its peak on admission, while NTproBNP peaked at days 2–4 after onset of symptoms. A hsTnT > 89 ng/l or a NTproBNP > 2,615 ng/l obtained within 48 h after onset of symptoms had a sensitivity of 100 % and a specificity of 79 % in detecting SIC.
Conclusions
The cardiac biomarkers, hsTnT and NTproBNP, are increased early after SAH and levels are considerably higher in patients with SIC. These biomarkers are useful for screening of SIC, which could make earlier diagnosis and treatment of SIC in SAH patients possible.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Acute cardiac events are common in patients with subarachnoid hemorrhage (SAH). These cardiac events vary in clinical importance from subclinical electrocardiographic changes [1] to troponin leakage [2] or severe heart failure [3–6]. Several mechanisms causing the cardiac dysfunction are proposed [7, 8]; however, over-simulation of the myocardial beta-receptors is most likely involved in the pathogenesis [9–14]. The deteriorated cardiac function usually presents as regional hypokinesia of the left ventricle (LV), not following coronary artery distribution [3–6]. Because of the association with enhanced sympathetic activity, this condition is often denoted as stress-induced cardiomyopathy (SIC), but is also referred to as Tako-tsubo cardiomyopathy [15]. Although SIC generally is reversible [6, 11], patients with SIC after SAH have an increased risk of cerebral vasospasm, delayed cerebral ischemia and death [16–18]. Early detection and treatment of SIC in SAH patients could be crucial to ensure optimal cerebral blood flow and to potentially improve long-term outcome.
Patients with Tako-tsubo cardiomyopathy usually present with a modest increase in troponins and a major increase in B-type natriuretic peptide (BNP) or N-terminal pro B-type natriuretic peptide (NTproBNP) [19]. The specificity of troponins and NTproBNP for detection of myocardial injury/heart failure in general ICU patients is low, as troponins and NTproBNP are increased also in other conditions such as sepsis, ARDS and renal failure [20–24]. However, these conditions are not often seen in SAH patients on admission, why NTproBNP and troponins possibly could be used as specific cardiac biomarkers for detection of SIC in SAH patients. Previous studies have shown that BNP is increased in patients with myocardial dysfunction after SAH [25], but has not been used for detection of SIC in SAH patients. Studies on troponin I for detection of SIC in SAH patients have shown inconsistent results [17, 26–28]. A new method of measuring troponin was introduced recently, high-sensitive troponin T (hsTnT), which has a higher sensitivity and specificity for detection of myocardial injury than previous methods of measuring troponin [29]. The primary aim of this study was therefore to evaluate whether hsTnT or NTproBNP could be useful as biomarkers for early detection of SIC in SAH patients.
Materials and Methods
Patient Inclusion
The study was approved by the Regional Ethics Committee. In this retrospective observational study, we reviewed medical records of all patients admitted to our neuro-intensive care unit (NICU), from January 2010 to August 2014, with suspected or verified spontaneous, non-traumatic SAH. Inclusion criteria were performed echocardiography within 72 h after onset of symptoms and measurements of hsTnT and/or NTproBNP within 48 h after onset of symptoms. Since 2010, hsTnT was introduced as routine in our hospital laboratory, and since 2012, we introduced routine serial measurements of hsTnT and NTproBNP in all patients with subarachnoid hemorrhage on admission and the following 3 days. NTproBNP is the natriuretic peptide used routinely at our hospital for detection of LV failure. Patients were treated according to the recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference [30].
Data Collection
The following data were retrieved from medical records: findings on echocardiography (see below), levels of hsTnT and NTproBNP, neurological status on admission according to the World Federation of Neurological Surgeons (WFNS) grade for SAH [31] and aneurysm position. Furthermore, data on arterial blood pressure, heart rate, dose of vasopressors (norepinephrine) and use of inotropes on admission to NICU and at the time of echocardiography were obtained. Findings on electrocardiogram on admission and findings on chest X-ray at the time of echocardiography were also obtained. We identified patients fulfilling criteria for severe SIRS/sepsis, ARDS/neurogenic pulmonary edema (NPE) and acute kidney injury (AKI) or anemia during first three days after admission. Fluid intake during this period was also recorded. Clinical variables evaluated for their ability to detect SIC were as follows: hsTnT and NTproBNP on days 1, 2 and 3 after onset of symptoms, WFNS grade, ECG changes, systolic blood pressure, heart rate on admission and pulmonary edema/signs of heart failure on chest X-ray at time of echocardiography. WFNS grade 1–3 was defined as low-grade SAH, and WFNS grade 4–5 was defined as high-grade SAH. Severe SIRS/sepsis, ARDS/NPE and AKI were defined according to standard definitions [32–34]; anemia was defined as hemoglobin levels <10.0 g/dl. SIC was defined as: (1) transient hypokinesia, akinesia or dyskinesia of the LV segments (2) the absence of other credible pathological conditions (e.g., ischemia, myocarditis) explaining the LV regional dysfunction (3) modest elevation of troponin, i.e., disparity between the troponin levels and the amount of ventricular dysfunction, according to the Gothenburg criteria of SIC [35, 36].
Biomarker Analysis and Echocardiographic Evaluation
hsTnT was analyzed with the Roche high-sensitive troponin T assay with a coefficient of variation of 3.4 % [37]. NTproBNP was analyzed with the Elecsys® assay (Roche) on Cobas platform with a coefficient of variation of 3.8 %. Left ventricular ejection fraction (LVEF) was calculated with Simpson’s biplane method. Sixteen LV segments were evaluated individually and scored on the basis of its motion and thickening as followed: score 1 (normokinesia), score 2 (hypokinesia), score 3 (akinesia) and score 4 (dyskinesia). LV wall motion score index (WMSI) was derived as the sum of all scores divided by the number of segments [38]. The echocardiographist was blinded to biomarker levels of the individual patient.
Statistics
All continuous variables were checked for normal distribution with inspection of histogram distribution and Shapiro–Wilks test. Normally distributed continuous variables are presented as mean ± standard deviation, and non-normally distributed continuous variables are presented as median with interquartile range. T test was used to compare means between two groups in continuous normally distributed variables. Mann–Whitney U test was used to compare medians between two groups in continuous non-normally distributed variables. Fisher’s exact test was used for comparing incidences between two groups with binary variables. ROC curves were used to determine sensitivity, specificity and cutoff levels for variables to detect SIC. To compare levels of hsTnT and NTproBNP over time in patients with and without SIC, a linear mixed model was used. hsTnT and NTproBNP were logarithmized and set as dependent variables, and an autoregressive covariation matrix was used. For determining variables independently associated with levels of hsTnT and NTproBNP, a linear regression model was used. Peak levels of hsTnT and NTproBNP were logarithmized and set as dependent variables. In a univariate regression, variables with a p value < 0.10 were included in a multivariate analysis and variables <0.05 in the multivariable analysis were considered significant. All models were verified with scatterplot of residuals/predicted values and normally distribution of residuals. The results presented from the models are anti-logged. p values < 0.05 were considered significant. Software used was IBM SPSS Statistics version 22.0.
Day 1 was defined as the first 24 h after onset of symptoms.
Results
Patient Inclusion
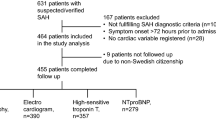
A total of 502 patients with suspected or verified SAH were admitted during the study period; 356 of these had a verified SAH. A total of 178 patients were examined with echocardiography, and 135 of these were examined within 72 h after onset of symptoms. Biomarkers were obtained within 48 h after onset of symptoms in 112 of these patients (Fig. 1). The most common indication for echocardiography (more than one possible) was high dose of vasopressor/inotropes (n = 71) followed by arrhythmias (n = 22), electrocardiographic changes indicating ischemia (n = 22), pulmonary edema (n = 20), substantial increase in cardiac enzymes (n = 17), difficult weaning (n = 14) and increased lactate with low central venous saturation (n = 5). Other indications for echocardiography were evaluation of heart valves because of the presence of cardiac murmurs (n = 12) and assessment of cardiac function in patients with known heart disease (n = 6).
A total of 91 patients (81 %) were admitted on day 1 after onset of symptoms, and the remaining 21 patients (19 %) were admitted on day 2 after onset of symptoms. Sixty-six patients (58 %), 24 patients (22 %) and 22 patients (20 %) had echocardiography performed on days 1, 2 and 3 after onset of symptoms, respectively. A reduction in the degree of hypokinesia in patients with SIC was verified between 2 and 21 days after first echocardiography. Serum hsTnT was obtained in all 112 patients, and serial measurements (≥2 samples during first 3 days after onset of symptoms) were performed in 108 patients. Serum NTproBNP was obtained in 93 patients, and serial measurements were performed in 81 patients.
Patient Characteristics
Study cohort characteristics of the 112 included patients are presented in Table 1. Twenty-five patients fulfilled echocardiographic criteria for SIC. The incidence of SIC was significantly higher in women (88 % of SIC patients) and in patients with high-grade SAH (72 % of SIC patients). The incidence of SIC was significantly lower in patients with a history of hypertension (12 % of SIC patients). There were no differences between the patients with and without SIC with respect to age, the need for ventricular drainage, aneurysm position and treatment. Coronary angiography was performed in one patient with SIC and was without coronary stenosis.
Hemodynamic and Pulmonary Variables and Other Complications in non-SIC and SIC Patients
Data on hemodynamic and pulmonary variables are presented in Table 2. On admission, systolic blood pressure (SBP) and diastolic blood pressure were lower, heart rate tended to be higher (p = 0.076), and the incidences of ST elevations and/or negative T-waves were higher in the SIC patients. At the time of echocardiography, SBP was lower, while the doses of norepinephrine and number of patients treated with inotropes were higher in SIC patients. The incidence of pulmonary edema tended (p = 0.084) to be higher in the SIC patients. LVEF was significantly lower in patients with SIC. Median number of hypokinetic segments was significantly higher in patients with SIC. Three non-SIC patients with prior myocardial infarction had irreversible hypokinetic areas in the LV, which was attributed to prior myocardial infarction. ARDS/NPE was the only complication more frequent in patients with SIC (Table 2).
hsTnT and NTproBNP in Non-SIC and SIC Patients
Peak levels of hsTnT and NTproBNP were significantly higher in patients with SIC (Fig. 2). Both hsTnT and NTproBNP were significantly higher in patients with SIC at all days (Fig. 3). hsTnT had its peak level on day 1 after onset of symptoms followed by a daily decline, both in patients with and without SIC (Fig. 3). Levels of NTproBNP were significantly higher on days 2, 3 and 4 after onset of symptoms, both in patients with and without SIC (Fig. 3).
Levels of hsTnT and NTproBNP in patients with and without SIC. Levels of hsTnT and NTproBNP were higher in SIC patients at all days. hsTnT had its peak on day 1 after onset of symptoms with a daily decline in both groups. NTproBNP had peak levels at day 2 after onset of symptoms in both groups, the difference between days 2, 3 and 4 were not significant in any of the groups. SIC stress-induced cardiomyopathy. *p < 0.05
hsTnT, NTproBNP and Clinical Variables for Detection of SIC
A hsTnT > 89 ng/l on day 1 after onset of symptoms, a NTproBNP > 2,615 ng/l on day 2 after onset of symptoms and a NTproBNP > 1,830 ng/l on day 3 after onset of symptoms had best values for detection of SIC, respectively (Fig. 4a–c), with a sensitivity of 100 % and a specificity of 83–91 %. A SBP ≤ 120 mmHg on admission had the best detective value of the hemodynamic variables with a sensitivity of 84 % and a specificity of 85 % (Fig. 4d). Mean arterial pressure (AUC 0.86) and diastolic pressure (AUC 0.74) were inferior to SBP in the detection of SIC. Heart rate could not significantly detect SIC. A summary of the diagnostic value for variables to detect SIC is presented in Table 3. hsTnT and NTproBNP had best detective value of the variables tested. High-grade SAH, ECG changes and pulmonary edema on chest X-ray were all inferior to hsTnT, NTproBNP and SBP on admission in detecting SIC. If both hsTnT and NTproBNP are measured simultaneously at any time within 48 h after onset of symptoms, the above-mentioned cutoff levels had a sensitivity of 100 % and a specificity of 79 % with a negative predictive value (NPV) of 100 % and a positive predictive value (PPV) of 58 %.
ROC curves for hsTnT and NTproBNP for day 1, day 2 and day 3 and for systolic blood pressure on admission. hsTnT had best detective value on day 1, while NTproBNP had best detective value on days 2 and 3. Systolic blood pressure on admission had a significant detective value with a high sensitivity and specificity. SBP systolic blood pressure, AUC area under the curve
Variables Associated with Peak Levels of hsTnT and NTproBNP (Tables 4, 5)
In a univariate analysis, SIC, age, sepsis and atrial fibrillation were significantly associated with peak levels of NTproBNP. In the multivariate analysis, SIC was the only variable significantly associated with peak levels of NTproBNP. SIC, high-grade SAH, SBP, heart rate and dose of norepinephrine on admission were significantly associated with peak levels of hsTnT in the univariate analysis, but only SIC and high-grade SAH were independently associated with peak levels of hsTnT in the multivariate analysis.
Discussion
The major findings in this retrospective study were that peak serum levels of hsTnT and NTproBNP were higher in patients developing SIC after SAH and if obtained within 48 h after onset of symptoms, had a high sensitivity and specificity of detecting SIC.
We used the latest generation of assay for troponin measurement, hsTnT, which permits detection of very low levels of troponin T and permits a faster detection of myocardial injury, than previously used assays [29]. We found that hsTnT obtained early after admission has a very high sensitivity of detecting SIC. On the other hand, we also demonstrated that NTproBNP is a better marker than hsTnT for detection of SIC at day 2, or later, after onset of symptoms. If taken simultaneously at any time within 48 h after onset of symptoms, a value below the suggested cutoff values (hsTnT > 89 ng/l, NTproBNP > 2,615 ng/l) excludes SIC, while a value above the cutoff values have a 58 % chance of detecting SIC. Although the positive predictive value was slightly lower than for each test alone (see Table 3), this approach is more practical due to less need for timing of biomarker blood sampling in relation to onset of symptoms. The excellent negative predictive value of hsTnT and NTproBNP makes these biomarkers suitable for screening of SIC in SAH patients. Even if the positive predictive value was moderate (58 %), more than half of the patients with biomarkers over our proposed cutoff levels would be diagnosed as having SIC, which should be considered acceptable for a screening test.
Previous studies have shown that plasma levels of hsTnT and NTproBNP can be increased in a variety of conditions not related to heart failure [20–24]. We used a multivariable regression model to discriminate whether the high levels of hsTnT and NTproBNP seen in SIC patients were independently associated with SIC, or to complications associated with SIC. In this model, SIC was the only independent variable associated with peak levels of NTproBNP, while both SIC and high-grade SAH were independently associated with peak levels of hsTnT. A majority of the patients developing SIC were women. A female overrepresentation of cardiac dysfunction in SAH patients has been demonstrated in several previous studies [2, 4, 5] and is well described in patients with Tako-tsubo cardiomyopathy [35]. An incidental finding was that the incidence of SIC was lower in patients with a history of hypertension. If this was not a coincidence, one could speculate that this could be due to treatment with beta-blockers and antihypertensive agents which is found to reduce incidence of SIC in previous studies [17, 39]. Unfortunately, we were not able to obtain reliable data on pre-admission medication in all patients to verify this finding in our data.
hsTnT had its peak on hospital admission followed by a daily decline, suggesting that myocardial injury occurs early in the course of the disease, in contrast with patients with myocardial ischemia, which has peak levels of hsTnT at days 2–3 after a coronary event [29, 40]. This is in line with the observation that patients with SIC had a lower systolic blood pressure upon admission, suggesting that these patients had developed cardiac injury and SIC already on arrival to the NICU.
Two small-sample studies by Parekh et al. [26] and Deibert et al. [27] successfully identified SIC in patients with SAH using troponin I, with a 100 % sensitivity and a specificity of 80–90 %. In those two studies, troponin was measured within 24 h after hemorrhage. A larger study by Kothavale et al. [28] found an association between increased levels of troponin I and SIC. However, the sensitivity was only 53 %, and the authors found troponin I to be an inadequate screening test. This could be explained by late inclusion (mean time 3.7 days). This may reflect the importance of measuring cardiac troponins early upon admission for detection of SIC and that troponin alone is probably not sufficient for early screening of SIC after SAH. Furthermore, none of the above-cited studies used ROC curves to define cutoff levels of troponin I for detection of SIC, none measured BNP or NTproBNP for detection of SIC, and in none of the studies was hsTnT measured for detection of myocardial injury.
The main limitation of this study is its retrospective design. As only patients in whom echocardiography was performed and cardiac biomarkers were measured early after hospital arrival were included, our study population was biased toward patients with a more severe heart condition. However, the major indications for echocardiography were the use of inotropes/vasopressors and the presence of arrhythmias causing hemodynamic instability and not biomarker levels. Levels of hsTnT and NTproBNP also had a wide range and were not biased toward exclusively high or low values. Obviously, we could have missed hemodynamically stable patients with SIC, and we cannot exclude that such patients would have had lower levels of hsTnT and NTproBNP than we suggest for detection of SIC. However, also hemodynamically stable patients were examined with echocardiography, and we believe that the incidence of such patients is low. Another limitation was that coronary angiography was not performed for exclusion of myocardial ischemia because of coronary artery disease. Thus, we cannot exclude the possibility that some of our patients had LV wall motion abnormalities caused by myocardial ischemia. However, previous studies have shown that coronary angiography is without coronary stenosis or vasospasm in patients with regional hypokinesia after SAH [3]. Coronary angiography is not without risks and nor recommended in the Neurocritical Care Consensus Conference [30]. All patients fulfilling the diagnosis of SIC had reversal of LV hypokinesia, which is a less likely finding in patients with LV wall motion abnormalities due coronary artery disease. The strengths of this study are that blood samples were taken close to onset of symptoms and that serial blood samples were taken in most patients. This enabled us to demonstrate the difference in time to peak hsTnT and peak NTproBNP and identify time as an important factor for detection of SIC.
In summary, we found that NTproBNP and hsTnT have an excellent negative predictive value and a moderate positive predictive value for detection of SIC, which make hsTnT and NTproBNP suitable for screening of SIC after SAH. We therefore recommend that these biomarkers should be measured early after arrival in the NICU for early detection of SIC. Early detection and treatment of SIC could potentially improve prognosis in SAH patients.
References
Burch GE, Meyers R, Abildskov JA. A new electrocardiographic pattern observed in cerebrovascular accidents. Circulation. 1954;9:719–23.
Tung P, Kopelnik A, Banki N, et al. Predictors of neurocardiogenic injury after subarachnoid hemorrhage. Stroke J Cereb Circ. 2004;35:548–51.
Kono T, Morita H, Kuroiwa T, Onaka H, Takatsuka H, Fujiwara A. Left ventricular wall motion abnormalities in patients with subarachnoid hemorrhage: neurogenic stunned myocardium. J Am Coll Cardiol. 1994;24:636–40.
Mayer SA, LiMandri G, Sherman D, et al. Electrocardiographic markers of abnormal left ventricular wall motion in acute subarachnoid hemorrhage. J Neurosurg. 1995;83:889–96.
Lee VH, Connolly HM, Fulgham JR, Manno EM, Brown JRD, Wijdicks EFM. Tako-tsubo cardiomyopathy in aneurysmal subarachnoid hemorrhage: an underappreciated ventricular dysfunction. J Neurosurg. 2006;105:264–70.
Banki N, Kopelnik A, Tung P, et al. Prospective analysis of prevalence, distribution, and rate of recovery of left ventricular systolic dysfunction in patients with subarachnoid hemorrhage. J Neurosurg. 2006;105:15–20.
Lee VH, Oh JK, Mulvagh SL, Wijdicks EFM. Mechanisms in neurogenic stress cardiomyopathy after aneurysmal subarachnoid hemorrhage. Neurocrit Care. 2006;5:243–9.
Redfors B, Shao Y, Ali A, Omerovic E. Are the different patterns of stress-induced (Takotsubo) cardiomyopathy explained by regional mechanical overload and demand: supply mismatch in selected ventricular regions? Med Hypotheses. 2013;81:954–60.
Naredi S, Lambert G, Eden E, et al. Increased sympathetic nervous activity in patients with nontraumatic subarachnoid hemorrhage. Stroke J Cereb Circ. 2000;31:901–6.
Sato K, Masuda T, Izumi T. Subarachnoid hemorrhage and myocardial damage clinical and experimental studies. Jpn Heart J. 1999;40:683–701.
Zaroff JG, Rordorf GA, Ogilvy CS, Picard MH. Regional patterns of left ventricular systolic dysfunction after subarachnoid hemorrhage: evidence for neurally mediated cardiac injury. J Am Soc Echocardiogr: Off Publ Am Soc Echocardiogr. 2000;13:774–9.
Banki NM, Kopelnik A, Dae MW, et al. Acute neurocardiogenic injury after subarachnoid hemorrhage. Circulation. 2005;112:3314–9.
Sugimoto K, Hirose Y, Inamasu J, et al. Association between elevated plasma norepinephrine levels and cardiac wall motion abnormality in poor-grade subarachnoid hemorrhage patients. Neurosurg Rev. 2013;36:259–66.
Shao Y, Redfors B, Scharin Tang M, et al. Novel rat model reveals important roles of beta-adrenoreceptors in stress-induced cardiomyopathy. Int J Cardiol. 2013;168:1943–50.
Kurisu S, Sato H, Kawagoe T, et al. Tako-tsubo-like left ventricular dysfunction with ST-segment elevation: a novel cardiac syndrome mimicking acute myocardial infarction. Am Heart J. 2002;143:448–55.
Sugimoto K, Watanabe E, Yamada A, et al. Prognostic implications of left ventricular wall motion abnormalities associated with subarachnoid hemorrhage. Int Heart J. 2008;49:75–85.
Temes RE, Badjatia N, Connolly ES, et al. Left ventricular dysfunction and cerebral infarction from vasospasm after subarachnoid hemorrhage. Neurocrit Care. 2010;13:359–65.
van der Bilt I, Hasan D, van den Brink R, et al. Cardiac dysfunction after aneurysmal subarachnoid hemorrhage: relationship with outcome. Neurology. 2014;82:351–8.
Akashi YJ, Musha H, Nakazawa K, Miyake F. Plasma brain natriuretic peptide in takotsubo cardiomyopathy. QJM: Mon J Assoc Physicians. 2004;97:599–607.
Rudiger A, Gasser S, Fischler M, Hornemann T, von Eckardstein A, Maggiorini M. Comparable increase of B-type natriuretic peptide and amino-terminal pro-B-type natriuretic peptide levels in patients with severe sepsis, septic shock, and acute heart failure. Crit Care Med. 2006;34:2140–4.
Shah KB, Nolan MM, Rao K, et al. The characteristics and prognostic importance of NT-ProBNP concentrations in critically ill patients. Am J Med. 2007;120:1071–7.
Lindner G, Pfortmueller CA, Braun CT, Exadaktylos AK. Non-acute myocardial infarction-related causes of elevated high-sensitive troponin T in the emergency room: a cross-sectional analysis. Intern Emerg Med. 2014;9:335–9.
Knudsen CW, Clopton P, Westheim A, et al. Predictors of elevated B-type natriuretic peptide concentrations in dyspneic patients without heart failure: an analysis from the breathing not properly multinational study. Ann Emerg Med. 2005;45:573–80.
Nyberg C, Karlsson T, Ronne-Engström E. Predictors of increased cumulative serum levels of the N-terminal prohormone of brain natriuretic peptide 4 days after acute spontaneous subarachnoid hemorrhage. J Neurosurg. 2014;120:599.
Tung PP, Olmsted E, Kopelnik A, et al. Plasma B-type natriuretic peptide levels are associated with early cardiac dysfunction after subarachnoid hemorrhage. Stroke J Cerebr Circ. 2005;36:1567–9.
Parekh N, Venkatesh B, Cross D, et al. Cardiac troponin I predicts myocardial dysfunction in aneurysmal subarachnoid hemorrhage. J Am Coll Cardiol. 2000;36:1328–35.
Deibert E, Barzilai B, Braverman AC, et al. Clinical significance of elevated troponin I levels in patients with nontraumatic subarachnoid hemorrhage. J Neurosurg. 2003;98:741–6.
Kothavale A, Banki NM, Kopelnik A, et al. Predictors of left ventricular regional wall motion abnormalities after subarachnoid hemorrhage. Neurocrit Care. 2006;4:199–205.
Reichlin T, Hochholzer W, Bassetti S, et al. Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. N Engl J Med. 2009;361:858–67.
Diringer MN, Bleck TP, Claude Hemphill J 3rd, et al. Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocrit Care. 2011;15:211–40.
Report of World Federation of Neurological Surgeons Committee on a Universal Subarachnoid Hemorrhage Grading Scale. J Neurosurg 1988;68: 985–6.
American College of Chest Physicians/Society of Critical Care. Medicine consensus conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20:864–74.
Davison DL, Terek M, Chawla LS. Neurogenic pulmonary edema. Crit Care. 2012;16:212.
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Inter, Suppl. 2012;2:1–138.
Schultz T, Omerovic E, Redfors B, et al. Stress-induced cardiomyopathy in Sweden: evidence for different ethnic predisposition and altered cardio-circulatory status. Cardiology 2012;122:180–86.
Redfors B, Shao Y, Omerovic E. Stress-induced cardiomyopathy (Takotsubo)—broken heart and mind? Vasc Health Risk Manag. 2013;9:149–54.
Hammarsten O, Jacobsson CE, Widegren M, Danylchenko T, Jaffe AS. Long-time quality assessment of the Elecsys Troponin T hs assay. Clin Biochem. 2013;46:1055–7.
Schiller NB, Shah PM, Crawford M, et al. Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. American Society of Echocardiography Committee on Standards, Subcommittee on Quantitation of Two-Dimensional Echocardiograms. J Am Soc Echocardiogr: Off Publ Am Soc Echocardiogr. 1989;2:358–67.
Liang CW, Chen R, Macri E, Naval N. Preadmission beta-blockers are associated with decreased incidence of neurogenic stunned myocardium in aneurysmal subarachnoid hemorrhage. J Stroke Cerebrovasc Dis: Off J Natl Stroke Assoc. 2013;22:601.
Boden H, Ahmed TA, Velders MA, et al. Peak and fixed-time high-sensitive troponin for prediction of infarct size, impaired left ventricular function, and adverse outcomes in patients with first ST-segment elevation myocardial infarction receiving percutaneous coronary intervention. Am J Cardiol. 2013;111:1387–93.
Acknowledgments
This study was supported by grants from the Swedish State Support for Clinical Research (ALFGBG-70000) and Gothenburg Medical Society.
Conflict of interest
Jonatan Oras, Christina Grivans, Keti Dalla, Elmir Omerovic, Bertil Rydenhag, Sven-Erik. Ricksten and Helene Seeman-Lodding declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oras, J., Grivans, C., Dalla, K. et al. High-Sensitive Troponin T and N-Terminal Pro B-Type Natriuretic Peptide for Early Detection of Stress-Induced Cardiomyopathy in Patients with Subarachnoid Hemorrhage. Neurocrit Care 23, 233–242 (2015). https://doi.org/10.1007/s12028-015-0108-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-015-0108-y